Clear Sky Science · en
Candidozyma auris-infected Galleria mellonella larvae: the effect of the humanized monoclonal antibody Dia-T51 and its synergy with amphotericin B
Why a New Tool Against Fungal Infections Matters
Deadly fungal infections are a growing problem in hospitals worldwide, especially for patients whose immune systems are already under stress. One of the most worrying culprits is Candida auris, a yeast that resists many standard medicines and can spread quickly in intensive care units. This study explores a new biological therapy, a human-made antibody called Dia-T51, and tests whether it can protect living organisms from C. auris and make an existing antifungal drug work better, all using an insect larva as a stand-in for human infection.
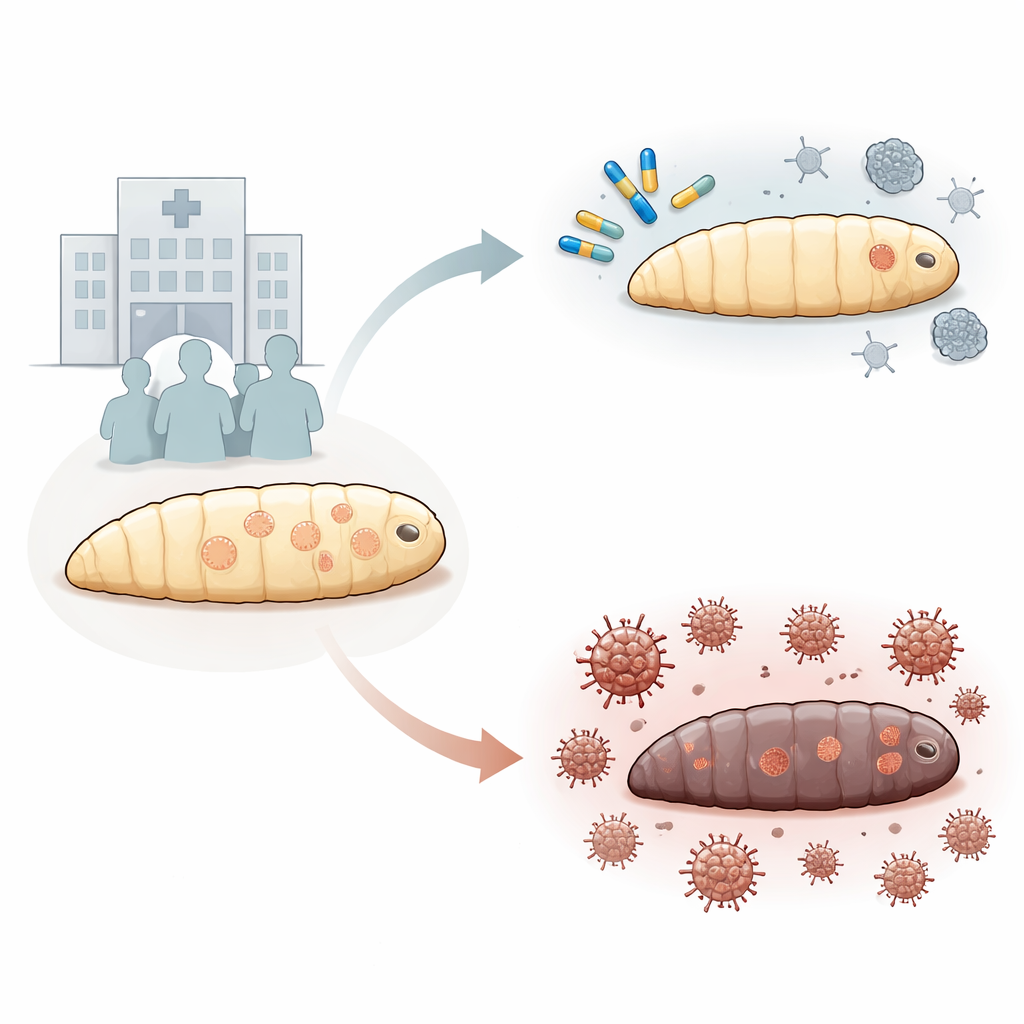
A Dangerous Fungus on the Rise
Candida auris has rapidly become a high-priority global health threat because it is hard to detect, difficult to treat, and can survive on hospital surfaces. Many patients with bloodstream infections caused by this fungus die, in part because the organism often resists several drug classes at once. At the same time, our antifungal arsenal is small, and high doses of powerful drugs such as amphotericin B can damage organs, especially the kidneys. This situation has pushed researchers to search for smarter therapies that do not simply rely on ever-increasing drug doses.
A Designer Antibody With a Clear Target
The researchers focused on a structure that is common to many fungi but absent in humans: a sugar-based building block called beta-1,3-glucan, which is part of the fungal cell wall. They had previously created Dia-T51, a “humanized” monoclonal antibody that binds tightly to this structure, latching onto the fungal surface without reacting to human cells. Earlier laboratory work showed that Dia-T51 could slow fungal growth and help immune cells engulf C. auris. It also hinted that pairing the antibody with amphotericin B might be more effective than either treatment alone, but the best dosing strategy and real-world impact still needed to be tested in living organisms.
Testing in a Simple Living Model
To explore safety and effectiveness, the team used larvae of the wax moth Galleria mellonella, a widely accepted model for human infection studies. These larvae have an innate immune system that behaves in many ways like our first line of defense, including cells that swallow microbes and a dark pigment response that marks inflammation. First, the scientists confirmed that Dia-T51 and amphotericin B were not toxic to the larvae at the tested doses. Then they infected the larvae with C. auris and treated them with the antibody alone, the drug alone, or both. Dia-T51 on its own significantly increased survival after a lethal infection and worked especially well when given before exposure to the fungus, protecting more than 90% of larvae and preventing the heavy darkening associated with severe inflammation.
Stronger Together: Antibody and Drug Synergy
When the team examined the combination of Dia-T51 and amphotericin B, the results were striking. In test-tube experiments, adding Dia-T51 allowed the same level of fungal killing with four- to eight-fold lower doses of amphotericin B. In infected larvae, a dose of amphotericin B that was useless on its own suddenly became highly protective when paired with Dia-T51, achieving survival rates that exceeded those seen with much higher, potentially harmful doses of the drug alone. Measurements of fungal cells in the larvae’s blood-like fluid showed that the combination cleared the fungus faster and more thoroughly than either treatment alone, and that inflammation resolved more quickly. These findings suggest that Dia-T51 helps expose or weaken the fungal cell wall in a way that makes amphotericin B more deadly, while also shaping the immune response so that it is effective but not overly damaging.
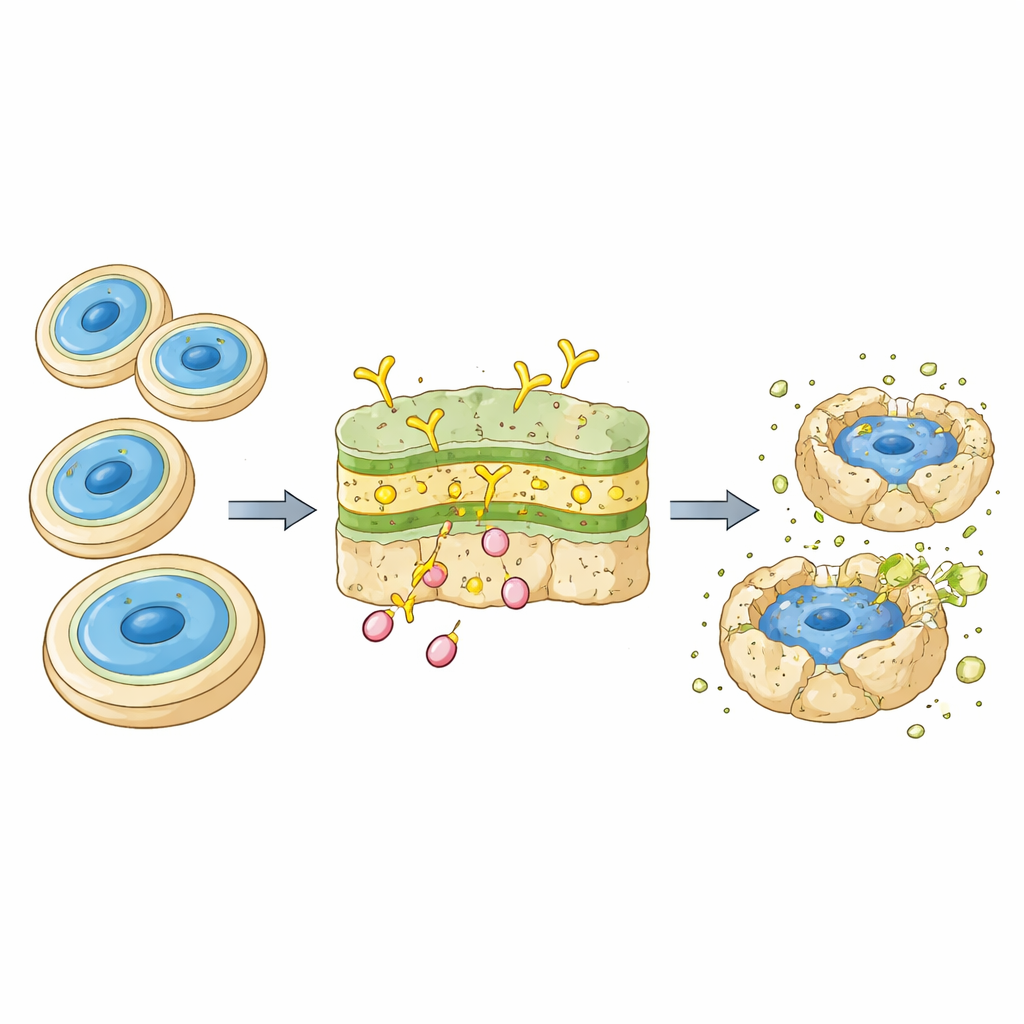
What This Could Mean for Patients
Although insect larvae are not humans and cannot reproduce the full complexity of human disease, they provide a powerful first test of how new therapies behave in a living system. This study shows that Dia-T51 is safe in vivo, can directly protect against deadly C. auris infection, and, crucially, can turn a low, otherwise ineffective dose of amphotericin B into a potent, long-lasting treatment. For patients, this points toward a future in which antibodies like Dia-T51 might be used to rescue failing antifungal drugs, allowing clinicians to use lower doses with fewer side effects while still overcoming resistant fungi. If confirmed in mammalian models and clinical trials, such antibody–drug partnerships could become an important new line of defense against hard-to-treat fungal infections in hospitals.
Citation: Vanzolini, T., Fiori, V. & Magnani, M. Candidozyma auris-infected Galleria mellonella larvae: the effect of the humanized monoclonal antibody Dia-T51 and its synergy with amphotericin B. npj Antimicrob Resist 4, 25 (2026). https://doi.org/10.1038/s44259-026-00198-4
Keywords: Candida auris, antifungal resistance, monoclonal antibody, amphotericin B synergy, Galleria mellonella model