Clear Sky Science · en
Host proteins associated with strong neutralizing SARS-CoV-2 antibody responses in a South African cohort
Why some people make stronger COVID-fighting antibodies
When people catch the virus that causes COVID-19, their bodies make antibodies that can block the virus from infecting cells. But not everyone produces these powerful “neutralizing” antibodies to the same degree. This study asked a simple but important question: can we read clues in the blood soon after infection that tell us who will later develop especially strong virus-blocking antibodies, and what does that reveal about how our bodies fight SARS-CoV-2?
Following patients from illness to recovery
Researchers in Durban, South Africa followed 71 adults hospitalized during the first wave of COVID-19, before vaccines and major variants appeared. All were infected with the original form of SARS-CoV-2 and had never been vaccinated. Blood was taken twice: first, about six days after diagnosis, when patients were still acutely ill, and again about a month later, during recovery. Early blood samples were analyzed using a high‑throughput technique that can measure roughly 5,000 different proteins in blood plasma at once. Later samples were tested to see how well each person’s antibodies could neutralize live virus and how strongly their antibodies bound to the viral spike protein.
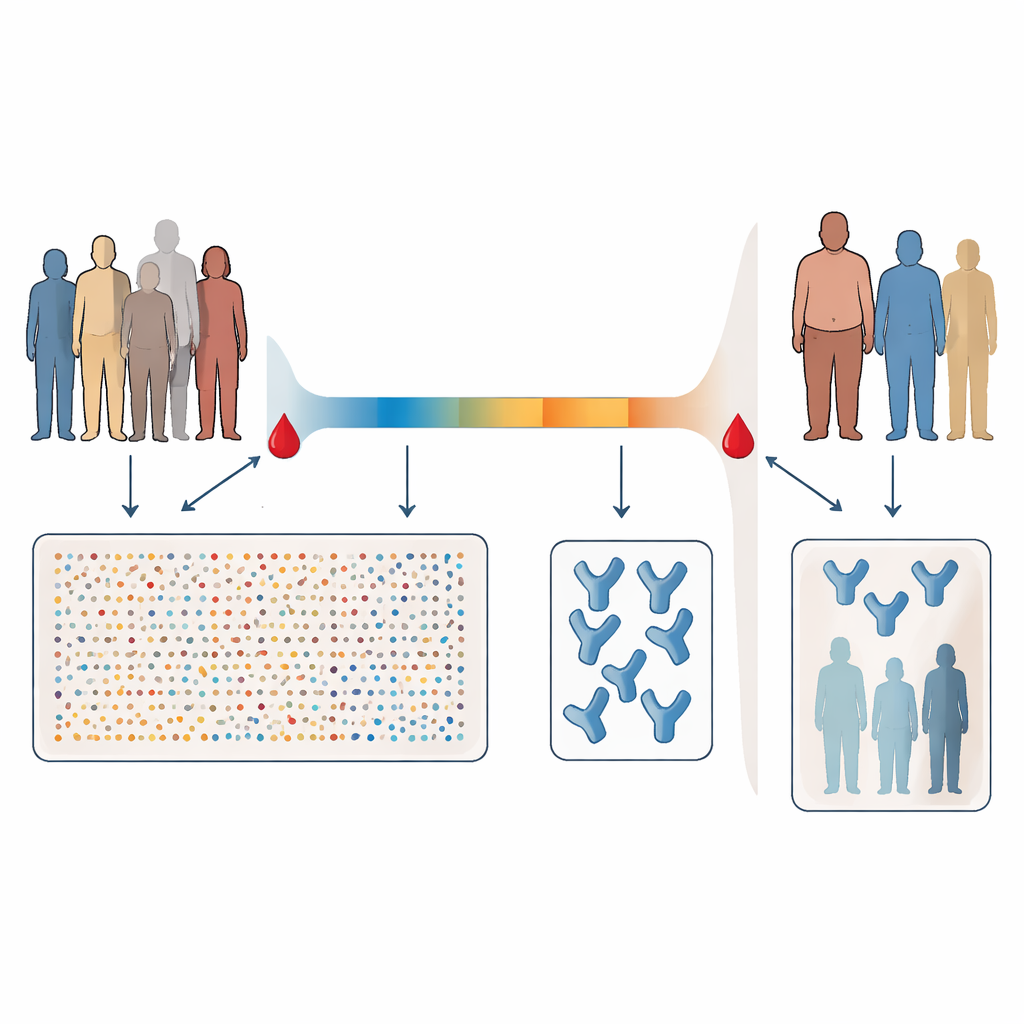
Illness level, risk factors, and antibody strength
As seen in many other studies, people who were sicker tended to have stronger neutralizing antibody responses. Participants were classified by whether they ever needed supplemental oxygen, a standard marker of more serious COVID-19. Those who needed oxygen, and those who were older, male, or had conditions like high blood pressure or diabetes, were more likely to fall into the high‑neutralizer group. Even among patients who never needed oxygen, subtler markers of worse illness—such as a higher ratio of neutrophils to lymphocytes in the blood and the same set of risk factors—were more common in people with strong neutralizing responses. This suggests that, even without intensive care or ventilation, somewhat more severe disease often goes hand‑in‑hand with more potent antibodies.
Early protein signals that predict strong antibodies
The central focus of the work was the pattern of proteins circulating in blood early in infection. By comparing people with high and low neutralizing activity, and also by treating neutralization as a continuous scale, the team identified hundreds of proteins that differed between groups. Many of these overlapped with proteins associated with simple spike‑binding antibodies, but a subset appeared more closely tied to the quality of the neutralizing response. One standout protein was HSPA8, a member of the heat shock protein family that helps fold other proteins and plays a key role in how immune cells display pieces of virus to helper T cells. Higher levels of HSPA8 early in infection were strongly linked to better neutralizing activity later, and this single protein could, on its own, distinguish high from low neutralizers reasonably well. When combined with two other proteins, FAP and MLN, prediction improved further.
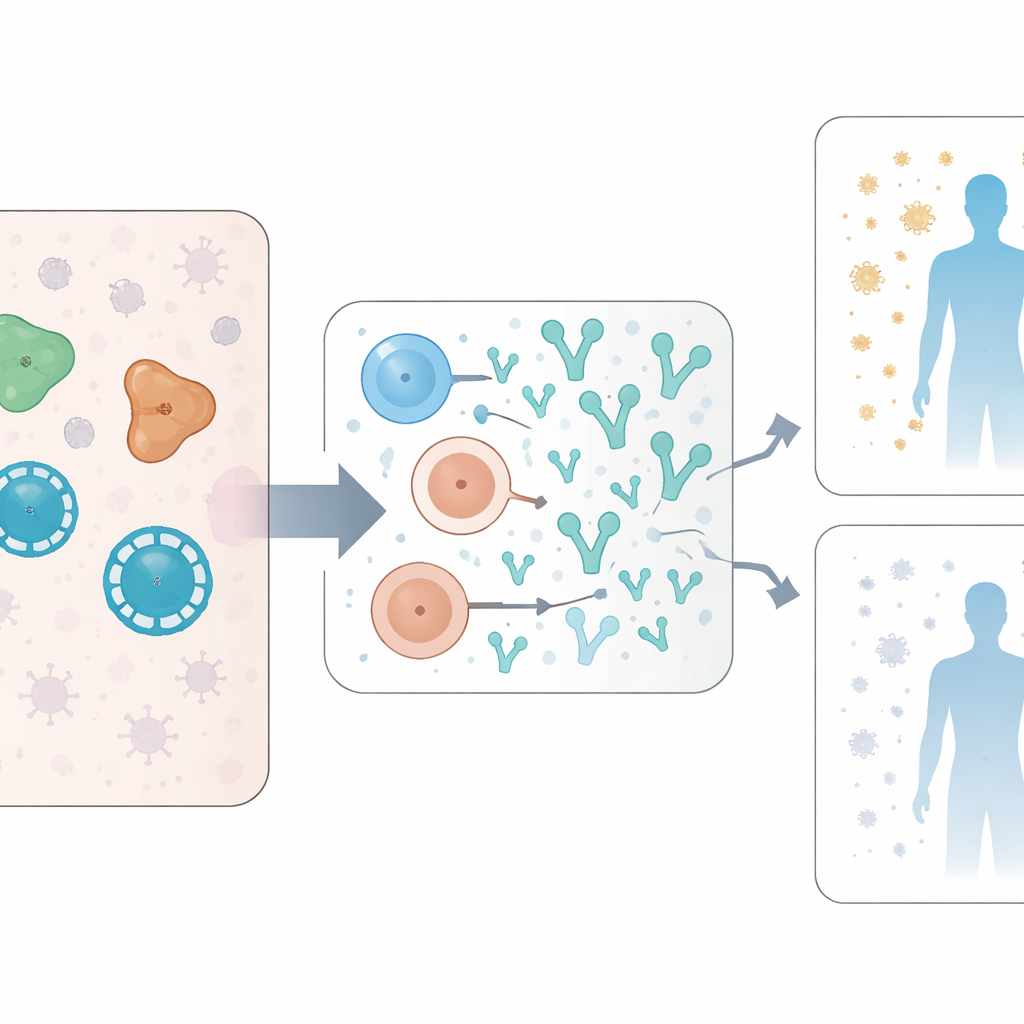
Pathways that connect metabolism, infection, and immunity
Looking beyond individual proteins, the researchers examined whole biological pathways that were more active in people who later developed strong neutralizing antibodies. Several of these pathways are involved in how cells handle energy and fats, including fatty acid metabolism and oxidative phosphorylation, as well as signals like PI3K/Akt/mTOR. These same pathways are known to be exploited by SARS‑CoV‑2 to copy itself, and they also support energy‑hungry immune cells such as activated B cells. The study found that blocking fatty acid metabolism, in other experimental work cited by the authors, can dampen viral replication. Together, this hints that high viral activity and a revved‑up cellular metabolism may help drive a more powerful neutralizing antibody response, even as they contribute to worse disease.
How disease severity, proteins, and antibodies intertwine
The proteins tied to disease severity overlapped only partly with those linked to strong neutralization, suggesting related but not identical processes. For example, the chemokine CXCL13—previously described as a marker of severe COVID‑19—was higher in patients needing oxygen. Yet when the researchers removed all oxygen‑requiring patients from the analysis, almost all of the protein differences between high and low neutralizers disappeared. This implies that the biology of more serious illness, including higher viral loads and stronger inflammation, is deeply intertwined with the development of robust neutralizing antibodies, even among patients who never come close to intensive care.
What this means for understanding COVID-19 immunity
To a lay reader, the key takeaway is that our early blood chemistry carries a fingerprint of how powerfully we will later be protected by neutralizing antibodies after COVID‑19. Proteins involved in showing viral fragments to the immune system, reshaping cell metabolism, and responding to inflammation all appear to influence this outcome. While severe illness is dangerous and never desirable, the same processes that make some people sicker may also help them mount stronger, longer‑lasting protection. Mapping these host protein and pathway signatures could eventually guide vaccines or treatments that coax the immune system into producing strong neutralizing antibodies without requiring people to endure severe disease.
Citation: Khairallah, A., Jule, Z., Piller, A. et al. Host proteins associated with strong neutralizing SARS-CoV-2 antibody responses in a South African cohort. Commun Med 6, 203 (2026). https://doi.org/10.1038/s43856-026-01427-7
Keywords: neutralizing antibodies, SARS-CoV-2, proteomics, host immune response, COVID-19 severity