Clear Sky Science · en
AI-assisted quantitative deciphering of molecular configuration and steric effects for high-performance zinc battery electrolytes
Smarter Batteries for a Renewable Future
As solar panels and wind farms multiply, we need affordable, safe batteries that can store huge amounts of energy for hours or days. Aqueous zinc-ion batteries are especially attractive because zinc is cheap, abundant, and works in water-based electrolytes instead of flammable organic liquids. But today’s zinc batteries still suffer from short lifetimes and unstable metal surfaces. This study shows how artificial intelligence can sift through thousands of candidate molecules to find a few that quietly transform how zinc ions move in the liquid, dramatically extending battery life.
Using AI to Search a Sea of Molecules
The authors start from a familiar problem in chemistry: there are far more possible organic molecules than any lab can test by trial and error. To tackle this, they built a large database of over 20,000 small molecules from public chemical archives, then coupled it to an AI system based on large language models. Using a method called retrieval-augmented generation, the AI first “read” and summarized 50 recent research papers on zinc batteries to extract what actually matters for electrolyte additives—such as how well a molecule dissolves in water, where its polar groups sit, and how it interacts with zinc ions and the metal surface. The researchers then carefully engineered prompts so that the AI could apply these literature-derived rules consistently when screening molecules against three key criteria: simple ring or short-chain structures, moderate water solubility, and the right kinds of polar chemical groups.
From Thousands of Options to Two Standout Helpers
Initial AI screening narrowed the huge molecular pool down to 18 promising candidates. To sharpen this list further, the team performed quantum mechanical calculations to quantify two basic properties: how strongly each molecule binds to a zinc ion, and how heavy it is. Plotting binding strength against molecular weight let them pick out pairs that were similar in these respects but very different in shape. From this approach, two molecules emerged as ideal “contrast partners”: 2-methylimidazole (MI), a rigid ring, and 3-aminopropanol (AP), a flexible chain. This pairing allowed the researchers to cleanly probe how molecular shape and the amount of space a molecule occupies—its so-called steric effects—govern the microscopic structure around zinc ions and, ultimately, battery performance.
How Molecular Shape Steers Ion Motion
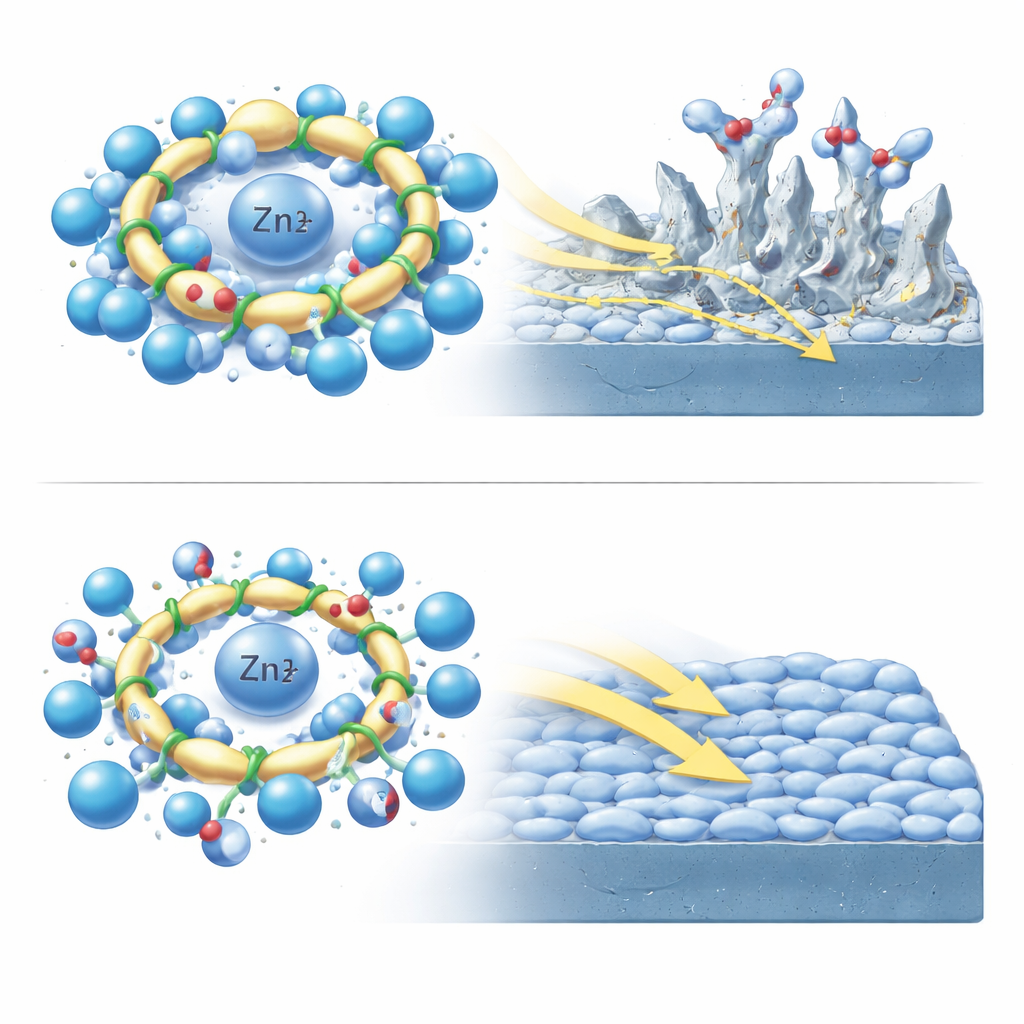
Using large-scale molecular dynamics simulations, the authors watched how MI and AP sit in the first shell of molecules surrounding a zinc ion in water. MI’s stiff ring forms a bulky, inflexible cage that forces surrounding water molecules into a crowded arrangement, raising a kind of “entry barrier” for solvent rearrangement and limiting how easily the zinc ion can move. In contrast, AP’s bendable chain shifts its conformation to fit the environment, building a more compact but less crowded shell that lets water and ions move more freely. AP also coordinates with zinc through multiple sites along its chain, including both nitrogen and oxygen atoms, meaning it can stabilize the ion while still leaving room for quick migration. These structural differences ripple outward, altering how tightly zinc and sulfate ions cluster, the electrostatic landscape the ions experience, and the way water molecules form and break hydrogen bonds.
Rebuilding the Liquid’s Hidden Network
The team dug deeper into the “invisible wiring” of the electrolyte: the hydrogen-bond network among water molecules. In plain zinc sulfate solution, water molecules are strongly linked, which helps protons hop rapidly via the Grotthuss mechanism, feeding undesirable hydrogen gas evolution at the zinc surface. Adding MI disrupts this network somewhat but leaves many pathways intact. AP, with its flexible chain and multiple bonding sites, does far more: it steals hydrogen bonds from water-water pairs, converts many long, weak links into stronger but more localized ones, and substantially lowers the overall dielectric response and dipole fluctuations of the liquid. Together with favorable solvation and desolvation energies and strong adsorption onto the zinc surface, this rewiring of the hydrogen-bond network calms down side reactions, lowers the energy barrier for zinc-ion motion, and encourages ions to plate smoothly rather than forming needle-like dendrites.
From Microscopic Tweaks to Longer-Lasting Cells
When these additives were tested in real zinc symmetric cells, the microscopic insights translated into striking gains. In plain zinc sulfate, the cells failed after about 130 hours of cycling. With MI, lifetime stretched to roughly 1500 hours; with AP it reached about 1900 hours, while corrosion currents dropped and the onset of hydrogen evolution shifted to more negative voltages. Microscopy showed that AP led to flat, compact zinc deposits instead of sharp protrusions. To a non-specialist, the takeaway is that a flexible chain molecule, chosen by an AI-guided search and validated by detailed simulations and experiments, can rearrange the liquid environment around zinc so that ions move quickly, side reactions slow down, and the battery lasts far longer. More broadly, the study showcases a blueprint for using AI not just to hunt for promising materials, but to understand why they work—offering a powerful path toward better, safer energy storage technologies.
Citation: Gao, Y., Sun, R., Shi, Y. et al. AI-assisted quantitative deciphering of molecular configuration and steric effects for high-performance zinc battery electrolytes. Commun Mater 7, 93 (2026). https://doi.org/10.1038/s43246-026-01100-5
Keywords: aqueous zinc-ion batteries, electrolyte additives, artificial intelligence in materials science, solvation structure, hydrogen bond network