Clear Sky Science · en
Iron ion enables photocatalytic hydrogen evolution from methanol
A Simple Way to Make Clean Fuel
Hydrogen is often hailed as a clean fuel of the future because it releases only water when used, not carbon dioxide. But today most hydrogen is still made from fossil fuels, which undermines its climate benefits. This study explores a surprisingly simple way to make hydrogen from everyday alcohols such as methanol, using ordinary iron salts and light instead of expensive precious metals and high temperatures. 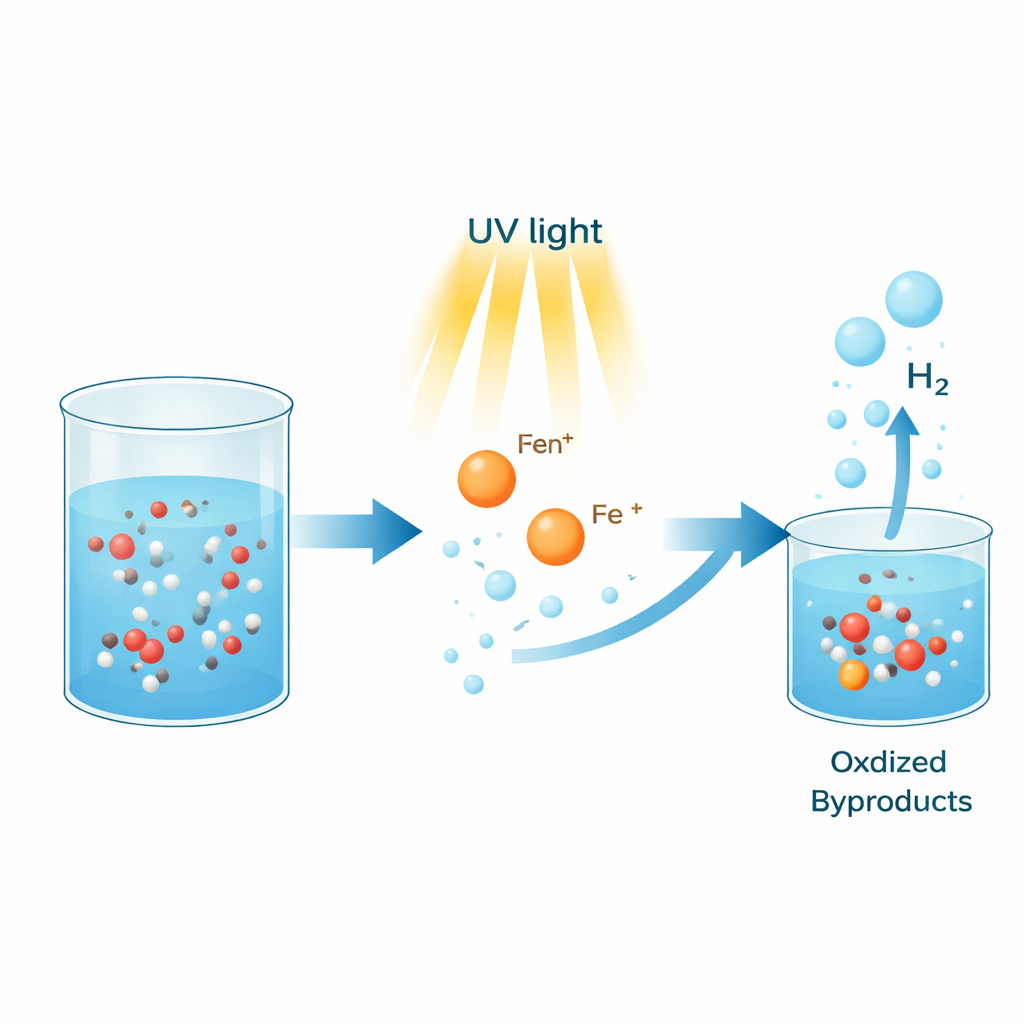
Turning Liquid Alcohol into Useful Gas
The researchers focused on methanol, a common liquid that already stores a lot of hydrogen in a compact form. If you can strip hydrogen out of methanol efficiently, the liquid can act as an easy-to-handle hydrogen carrier for fuel cells and other devices. Traditional methods rely on complex metal compounds or solid catalysts that contain rare elements like platinum, ruthenium, or iridium, and often demand high heat. In contrast, this work shows that a simple iron salt, dissolved in methanol and helped by a small amount of base, can release hydrogen when the mixture is shined with ultraviolet light.
How Light and Iron Work Together
In the new system, iron ions in solution grab onto nearby methanol molecules. Ultraviolet light is absorbed by these iron–alcohol pairs, causing an internal charge shift: electrons move from the alcohol side toward the iron center. This light-driven step turns iron(III) into iron(II) and creates very reactive alcohol fragments called radicals. These short-lived fragments help break apart methanol and pair up hydrogen atoms into hydrogen gas, while the carbon part of methanol is left in a more oxidized form such as formaldehyde or formic acid. The team’s experiments also showed that the reaction needs both light and oxygen from the air to keep running; under nitrogen, no hydrogen is formed, indicating that oxygen quietly participates in resetting parts of the system. 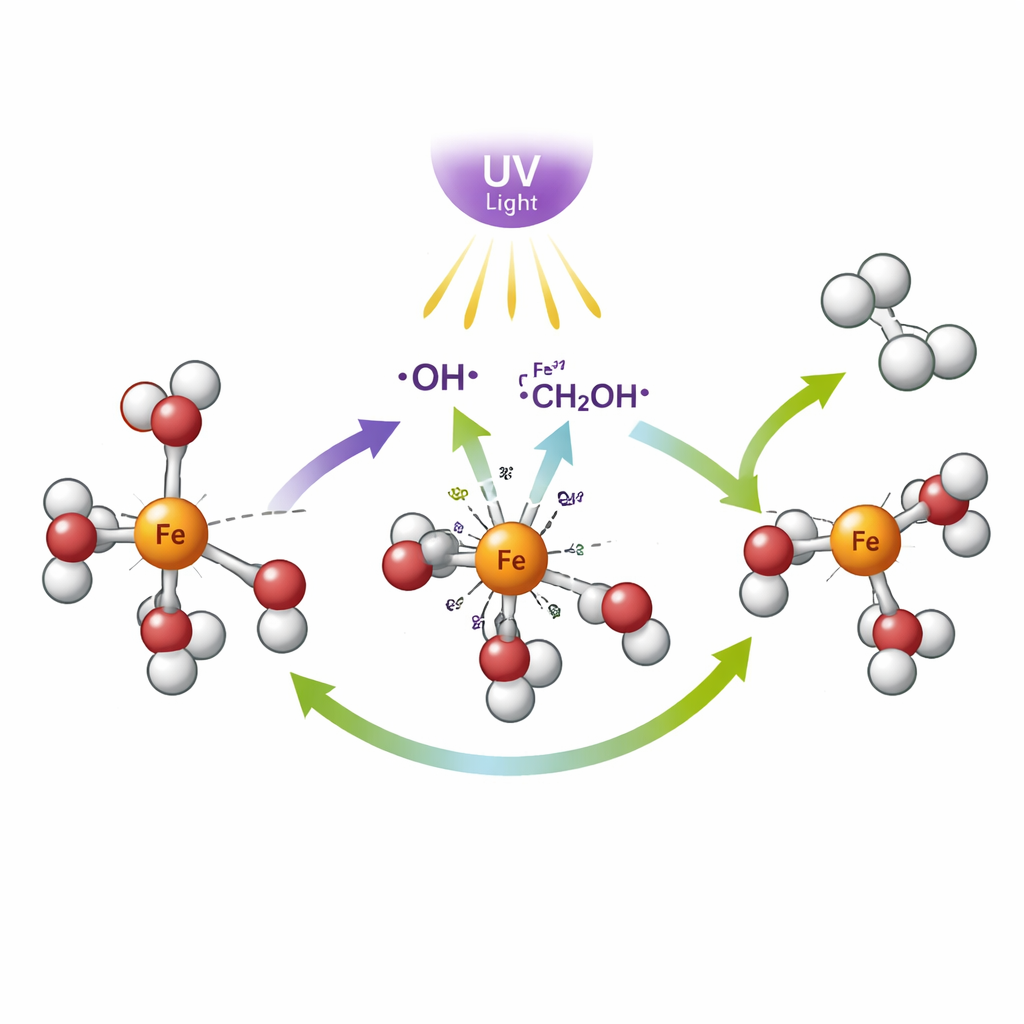
Tuning the Reaction and Testing Its Limits
Because the setup is so simple, the authors could easily probe what makes it work best. Adding sodium hydroxide, a common base, greatly boosts the reaction by helping convert methanol into a more reactive form that binds iron more strongly. Measuring how reaction speed changes with base concentration revealed a saturation behavior similar to the way enzymes operate, hinting at a fast initial adjustment step followed by a slower light-driven step. The amount of hydrogen produced also depends on how much iron is present and how intense the light is: lower iron concentrations and stronger ultraviolet illumination give higher efficiency per iron ion. The reaction continues steadily for days, can be modestly scaled up, and the same iron solution can be reused several times without a major loss in activity.
Beyond Methanol: Other Alcohols and Biomass
The team explored whether this light-and-iron approach could handle other feeds. They found that simple alcohols like ethanol, 1-propanol, and 2-propanol also produced hydrogen, though less efficiently than methanol, likely because bulkier molecules are harder to dehydrogenate. When water was added, the reaction slowed but still produced hydrogen at rates similar to some solid photocatalysts reported in the literature. Most strikingly, the system could even generate hydrogen from more complex, renewable sources such as glucose, cellobiose, starch, cellulose, and wood powder, though here the rates were much lower and the solid materials were only lightly altered.
What This Means for Future Energy
Overall, the study shows that something as basic as a dissolved iron salt can rival far more elaborate catalysts in turning methanol into hydrogen under light. The process still relies on ultraviolet light and works best without much water, so it is not yet ready to replace existing industrial methods. However, its low cost, simplicity, and ability to handle a range of alcohols and biomass suggest a fresh direction for clean-fuel research. By proving that bare iron ions in solution can act as efficient light-driven catalysts, the work opens the door to new, stripped-down designs for producing hydrogen from renewable feedstocks.
Citation: Sakurai, M., Kawasaki, Y., Itabashi, Y. et al. Iron ion enables photocatalytic hydrogen evolution from methanol. Commun Chem 9, 151 (2026). https://doi.org/10.1038/s42004-026-02009-3
Keywords: photocatalytic hydrogen production, methanol dehydrogenation, iron catalysts, solar fuels, liquid hydrogen carriers