Clear Sky Science · en
Spatiotemporal brain transcriptomics reveal risk gene hot-spots in major neuropsychiatric disorders
Why timing in the brain matters
Many mental and neurological disorders first appear at particular ages, from childhood autism to late life Alzheimer’s disease. This study asks a simple but powerful question: do the genes that raise risk for these conditions tend to be most active in the brain at the same times and places when symptoms usually emerge? By tracing when and where risk genes switch on during human brain development, the authors map “hot spots” of vulnerability that may help explain who gets sick, when, and why certain brain circuits are involved.
Following risk genes across a lifetime
To build this map, the researchers combined several large public datasets that measure which genes are active in different parts of the human brain, from early pregnancy through adulthood. They focused on risk genes identified by large genetic studies for 15 traits, including intelligence, autism, attention deficit hyperactivity disorder, Tourette’s syndrome, obsessive compulsive disorder, mood and anxiety conditions, epilepsy, and degenerative diseases such as Alzheimer’s and Parkinson’s. For each trait, they asked whether its risk genes were used more than expected in particular brain regions, ages, or cell types, using both bulk tissue and single cell data and treating enrichment as a sign of increased vulnerability.
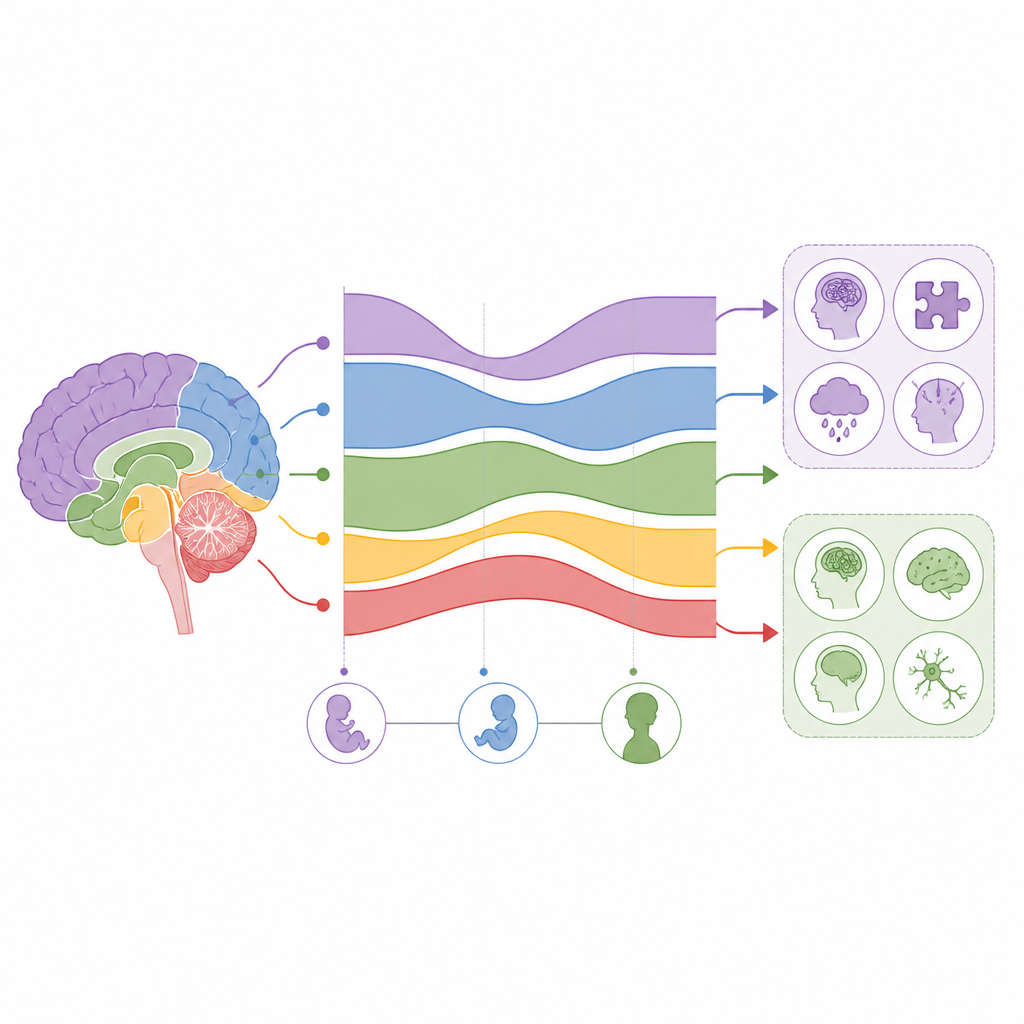
Early life versus later life vulnerability
A clear pattern emerged: traits that tend to start in childhood, such as autism, ADHD, Tourette’s, and higher or lower intelligence, showed their strongest risk gene activity before birth. In contrast, conditions that usually appear in adulthood, including epilepsy, Alzheimer’s disease, and Parkinson’s disease, showed peak activity of their risk genes after birth, often in midlife. Some disorders, notably major depression, obsessive compulsive disorder, and schizophrenia, had more complex profiles, with risk genes turning up both around birth and again in adolescence or young adulthood. Overall, the timing of risk gene activity roughly tracked the typical age when symptoms peak, supporting the idea that many of these conditions grow from long running developmental processes rather than sudden events.
Brain circuits as hot spots for risk
The study also pinpointed where in the brain these risk genes are most active. Many psychiatric traits shared broad involvement of the cerebral cortex, which supports thinking, planning, and social behavior. But there were notable differences in deeper structures. Risk genes for ADHD were especially enriched in the thalamus and striatum, key hubs for attention and movement control, while genes linked to panic disorder and neuroticism lit up in the amygdala, a center for emotion and fear. Obsessive compulsive disorder showed strong enrichment in the striatum, thalamus, and cerebellum, matching long standing circuit models of the disorder. By contrast, risk genes for autism, Tourette’s, and schizophrenia were relatively depleted in many subcortical regions, hinting at a more strongly cortical origin.
Zooming in to cells and molecular pathways
To understand mechanisms, the authors grouped genes that tend to turn on together into co expression “modules” and asked which modules were enriched for risk genes and when and where they were active. Some modules were mainly used by glial cells, such as microglia, astrocytes, and cells that form myelin, and were linked to Alzheimer’s disease, panic disorder, and ADHD. Others were used by excitatory and inhibitory neurons and were enriched for synapse formation, signaling, and forebrain development, connecting them to intelligence, autism, depression, schizophrenia, and epilepsy. Still other modules were most active very early in fetal life and involved chromatin and cell cycle control, suggesting that disruptions in how brain cells are born and programmed may set the stage for later mood and cognitive problems.
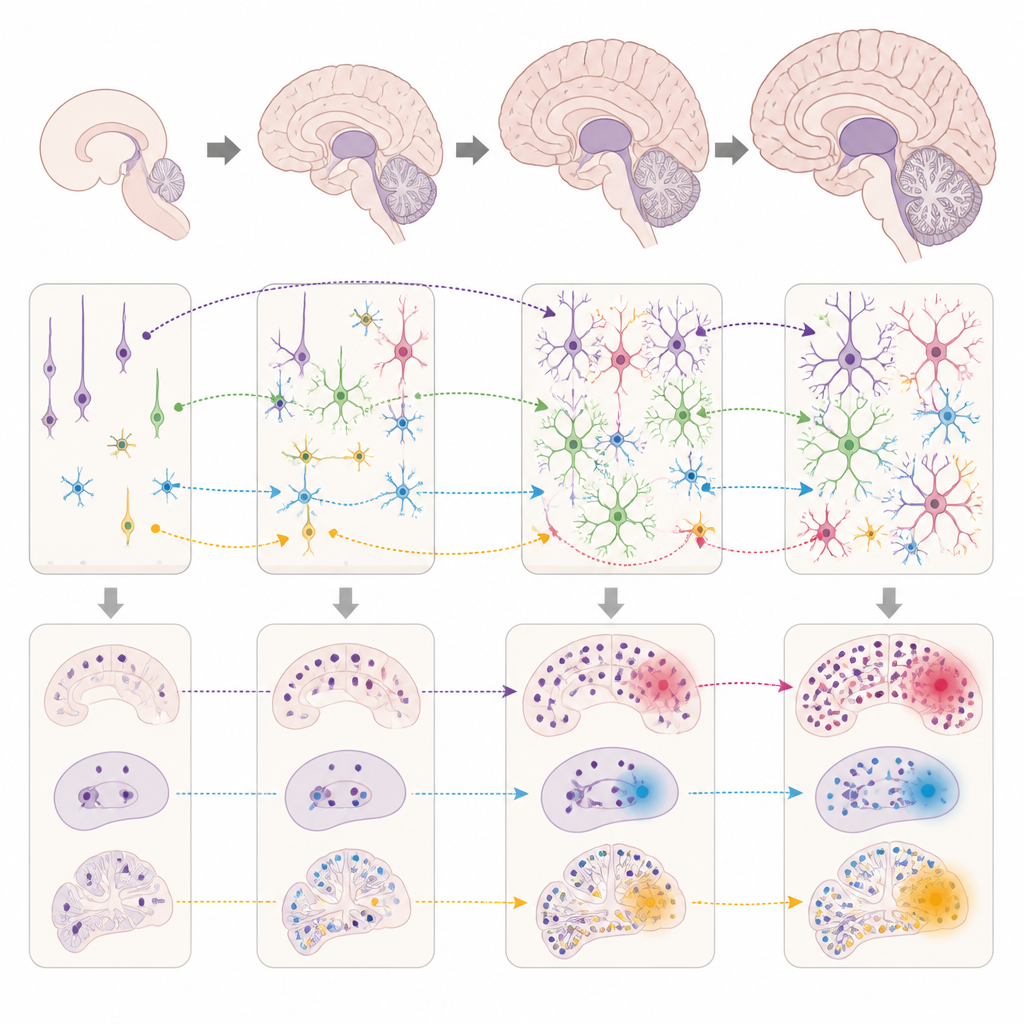
What this means for understanding brain disorders
In everyday terms, this work suggests that the same genes do not increase risk everywhere and at all times; instead, they act in specific brain regions, cell types, and developmental windows. When those windows line up with the circuits that support attention, emotion, or memory, the stage is set for particular disorders to emerge. By charting these patterns across many conditions at once, the study offers a systems level view that connects genetic findings to brain development and anatomy. In the long run, such maps could guide more precise research on when and where to look for early warning signs, and which cellular processes might be the most promising targets for treatment.
Citation: Liu, W., Shimogori, T. Spatiotemporal brain transcriptomics reveal risk gene hot-spots in major neuropsychiatric disorders. Commun Biol 9, 634 (2026). https://doi.org/10.1038/s42003-026-10045-x
Keywords: brain development, neuropsychiatric disorders, gene expression, risk genes, brain circuits