Clear Sky Science · en
Astrocytic EAAT1 suppression by EV-ACLY underlies glutamate imbalance and cognitive impairment in POCD
Why surgery and memory problems matter
Many older adults notice that thinking and memory feel “off” after a major operation. This condition, called postoperative cognitive dysfunction, can slow recovery and reduce independence, yet the biological reasons behind it have remained murky. This study uncovers a chain reaction inside the brain, showing how immune cells changed by surgery can disturb brain chemistry and weaken the connections needed for learning and memory.
Tiny messengers in the blood
The researchers focused on microscopic bubbles, known as extracellular vesicles, that cells use to send cargo through the bloodstream. They isolated these vesicles from the blood of patients who developed thinking problems after surgery and from healthy controls. When vesicles from affected patients were given to aging mice, the animals developed brain inflammation, higher levels of the signaling chemical glutamate, and trouble with memory tasks such as the Morris water maze. Detailed brain recordings showed that their synapses, the contact points where nerve cells talk to each other, were less able to strengthen in response to stimulation, a key feature of healthy learning circuits. 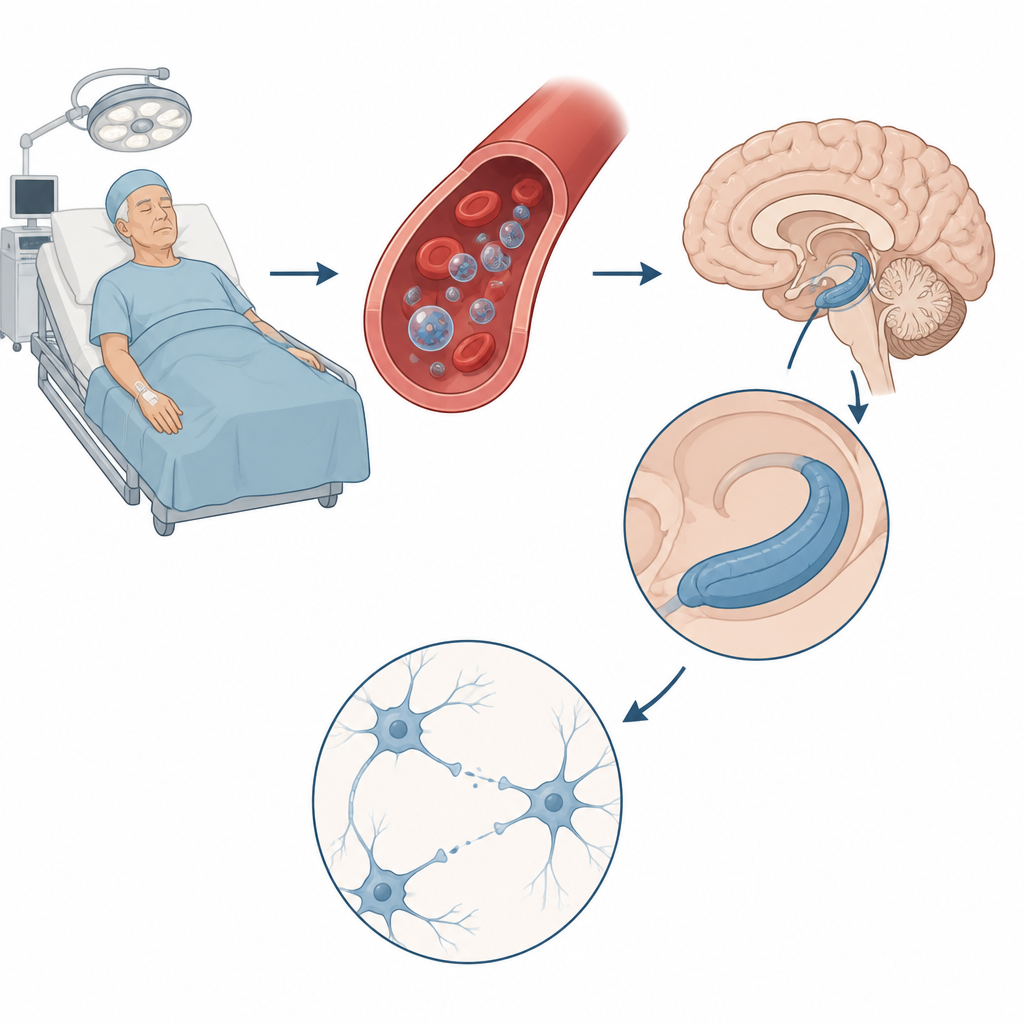
A harmful message from brain immune cells
Closer inspection pointed to microglia, the brain’s resident immune cells, as a major source of these harmful vesicles. When microglia were activated in the lab, they released vesicles enriched in an enzyme called ATP citrate lyase. These vesicles were readily taken up by neighboring astrocytes, star-shaped support cells that help keep brain chemistry in balance. In mice, blocking microglia with a drug greatly reduced brain inflammation, lowered glutamate buildup, preserved the structure of nerve cell branches and spines, and improved memory performance after surgery, suggesting that microglial signals are central to the problem.
How support cells lose control of glutamate
Astrocytes normally act as cleaners, sweeping excess glutamate out of the space between nerve cells through a transporter protein called EAAT1. The team discovered that the vesicle-delivered enzyme in astrocytes boosts the production of acetyl-CoA, a small molecule that fuels chemical tags on proteins controlling gene activity. This, in turn, increases a modified form of the switch protein NF-κB p65, which settles onto the EAAT1 gene and turns it down. With EAAT1 suppressed, astrocytes cannot clear glutamate efficiently, leading to a rise in this excitatory chemical around synapses and setting the stage for damage to neurons and their connections.
Proof from animal models
In aged mice, injecting vesicles from surgery patients or from activated microglia into the hippocampus caused microglial and astrocyte activation, loss of dendritic spines, weaker synaptic signaling, and clear memory deficits. When the researchers removed the enzyme from these vesicles using genetic tools, or blocked vesicle release or uptake, astrocytes regained EAAT1, glutamate levels normalized, and synapses and behavior improved. Time-course experiments showed that repeated vesicle exposure led to gradually worsening synaptic function and memory, highlighting that this is a cumulative process rather than a single hit. 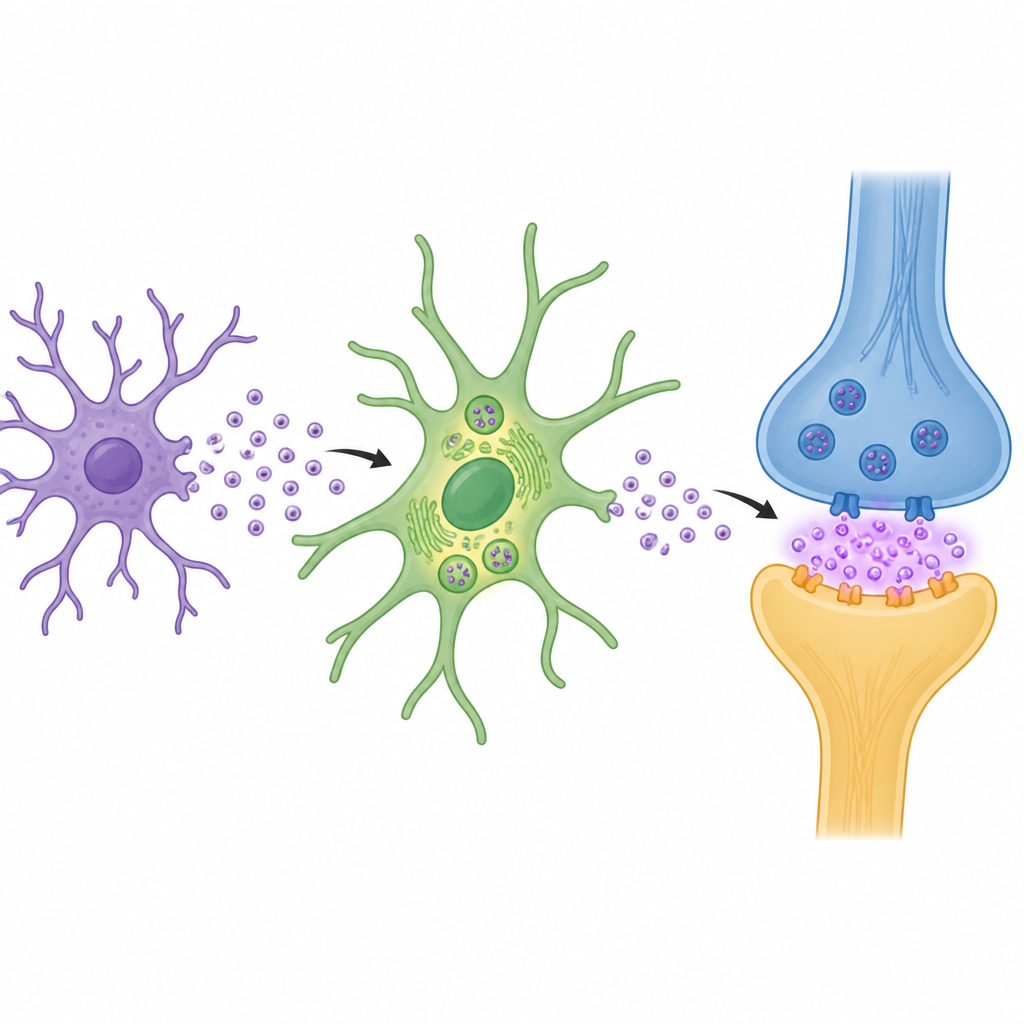
What this means for patients
In everyday terms, the study suggests that after surgery, brain immune cells can send out enzyme-loaded bubbles that “reprogram” nearby support cells, causing them to clear glutamate less effectively. The resulting chemical imbalance quietly erodes synapses and memory in vulnerable older brains. By identifying this enzyme-driven pathway as a key link between inflammation, glutamate overload, and cognitive decline, the work points to new treatment ideas, such as drugs that temper the enzyme’s activity or limit vesicle traffic during the critical period around surgery.
Citation: Qi, Z., Ding, L., Zhao, Y. et al. Astrocytic EAAT1 suppression by EV-ACLY underlies glutamate imbalance and cognitive impairment in POCD. Commun Biol 9, 636 (2026). https://doi.org/10.1038/s42003-026-09888-1
Keywords: postoperative cognitive dysfunction, microglia, astrocytes, glutamate imbalance, extracellular vesicles