Clear Sky Science · en
Antibiotic-induced gut microbiome remodeling reduces neuroinflammation in traumatic brain injury
Why the Gut Matters After a Head Injury
When we think about concussions or traumatic brain injuries, we usually picture damage inside the skull. But this study shows that what happens in the gut can powerfully shape how the brain heals. By briefly reshaping the community of microbes living in the intestines with antibiotics, the researchers found they could actually reduce brain damage and inflammation after head injury in mice, revealing an unexpected route to protect the brain.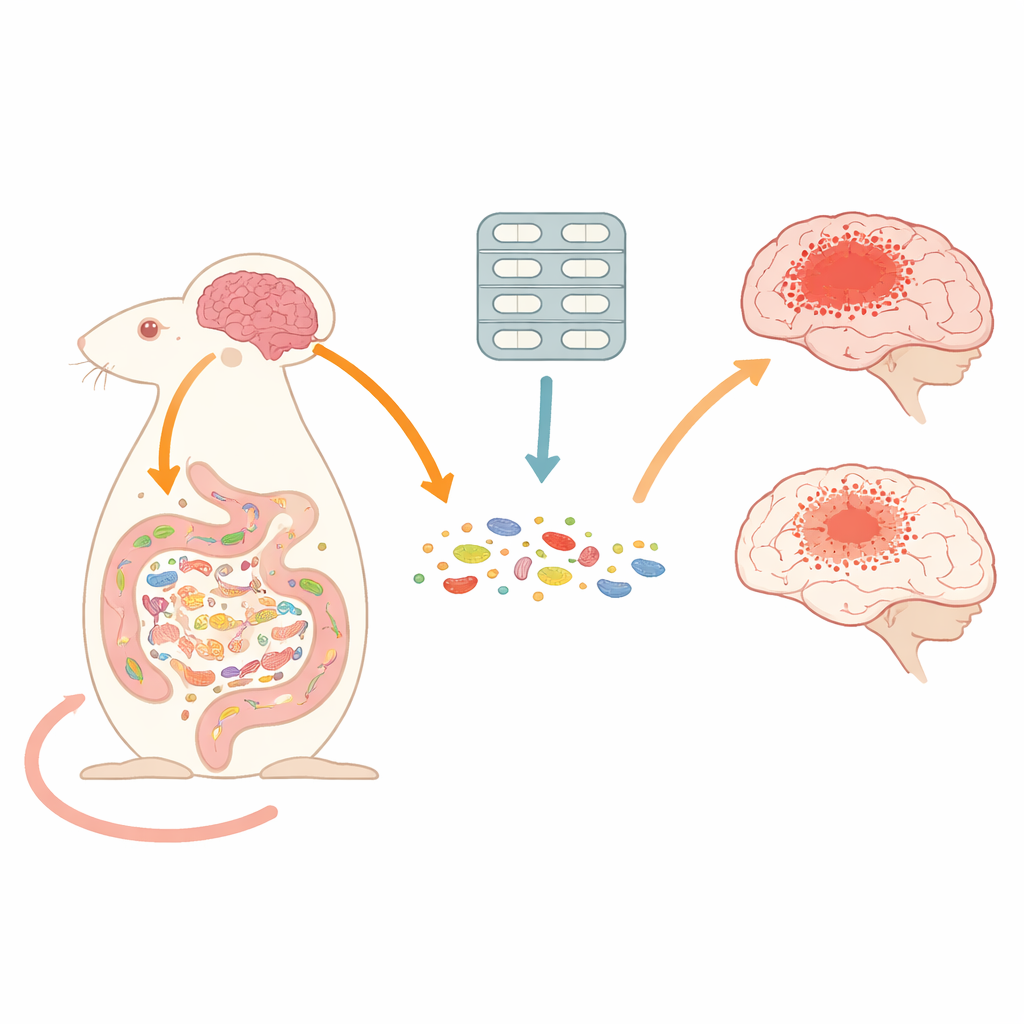
Recreating Head Injury and Gut Disruption in Mice
The team used a well-established model of traumatic brain injury in male mice, giving either a single controlled impact or two impacts separated by more than a month to mimic repeated injuries. Some mice received only water, while others were given a three-day cocktail of broad-spectrum antibiotics by mouth, similar to drugs used in intensive care units. The scientists then measured brain damage, motor function, immune cell activity in the brain, and detailed changes in gut microbes and their chemical products. They also studied germ-free mice raised entirely without microbes to see how a complete lack of a microbiome affects brain recovery.
Antibiotics Shrink Brain Damage but Come With Trade-Offs
Despite strongly disturbing the gut microbiome, short-term antibiotic treatment made the brain injuries less severe. Mice that had two head injuries and no antibiotics developed the largest brain lesions, the most cell death, and strong activation of immune cells called microglia, astrocytes, and infiltrating macrophages. In contrast, injured mice that received antibiotics had smaller lesion volumes, fewer dying cells in both the cortex and thalamus, and milder activation of these brain immune cells. They also showed less invasion of peripheral immune cells into the brain. Motor performance still worsened after repeated injury, but brain tissue damage and inflammatory signals were clearly reduced by the antibiotic course.
How the Microbiome and Its Chemicals Are Remodeled
The gut microbes did not escape unscathed. Antibiotics sharply reduced bacterial DNA in feces and reshaped microbial diversity, especially after repeated injury. Both traumatic brain injury and antibiotics on their own shifted the mix of species, and together they produced the largest change. Levels of short-chain fatty acids—small molecules made by gut microbes that often support anti-inflammatory and protective functions—fell in the bloodstream of antibiotic-treated mice, particularly the fatty acid butyrate. Long-read DNA sequencing revealed that some species, such as Parasutterella excrementihominis and Lactobacillus johnsonii, persisted or even expanded despite treatment, hinting that antibiotic-resistant or resilient microbes with special immune-calming properties might help counterbalance the loss of others.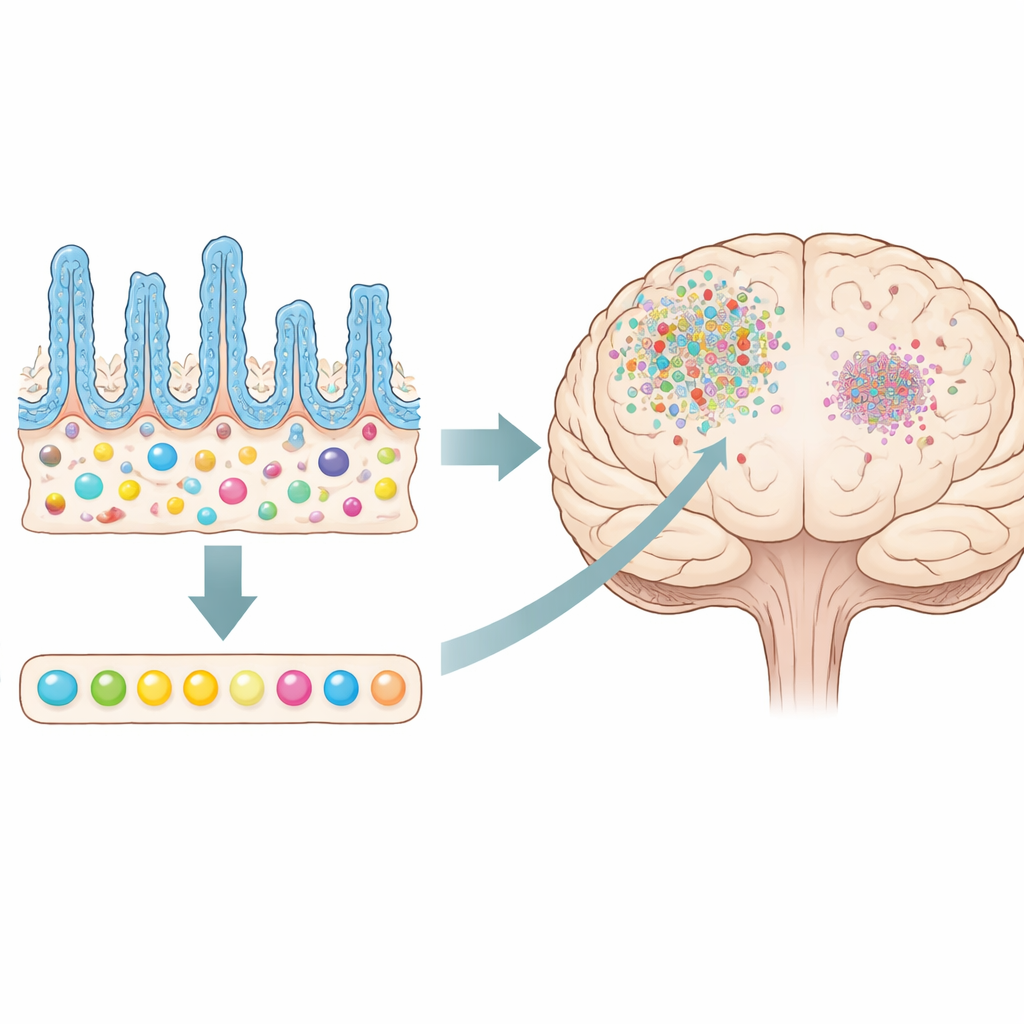
Gut Structure Suffers, Yet the Brain Benefits
Looking directly at the intestine, the researchers saw that worse brain injury was linked with shorter, disorganized finger-like villi and a loss of goblet cells that produce protective mucus. Short-term antibiotics further altered this already stressed gut lining, leading to shorter villi, narrower crypts, and fewer mucus-producing cells in the most severely injured, antibiotic-treated animals. In other words, the gut barrier itself appeared more fragile, even as inflammation in the brain went down. This contrast highlights that antibiotic-driven microbiome changes can help the brain while simultaneously imposing costs on intestinal health.
What Happens When There Are No Microbes at All?
To separate the impact of disrupting the microbiome from entirely lacking one, the team examined germ-free mice that had never been colonized by microbes. After brain injury, these animals fared worse than normal mice: their lesions were larger, and both microglia and astrocytes in the brain were more strongly activated. This suggests that a lifelong absence of microbes deprives the immune system and brain of important “training” signals needed for resilience. In contrast, briefly disturbing an already established microbiome with antibiotics seems to dial down certain inflammatory pathways without erasing all microbial functions.
What This Could Mean for Future Treatments
Overall, the study shows that traumatic brain injury not only harms the brain but also disrupts the gut and its microbial residents, and that the gut community in turn feeds back onto the injured brain. A short course of broad antibiotics in mice reshaped the microbiome, reduced key inflammatory responses, and limited brain tissue loss, even though it lowered helpful microbial metabolites and worsened some gut features. Because long-term or repeated antibiotic use in people carries serious risks, the authors do not propose antibiotics themselves as a cure. Instead, they argue that understanding which resilient bacteria and microbial pathways calm inflammation could inspire safer, targeted microbiome-based strategies—such as tailored probiotics or precision antimicrobials—to improve recovery after brain trauma.
Citation: Flinn, H., Marshall, A., Holcomb, M. et al. Antibiotic-induced gut microbiome remodeling reduces neuroinflammation in traumatic brain injury. Commun Biol 9, 481 (2026). https://doi.org/10.1038/s42003-026-09737-1
Keywords: traumatic brain injury, gut microbiome, antibiotics, neuroinflammation, short-chain fatty acids