Clear Sky Science · en
Brain-wide mapping reveals temporal and sexually dimorphic opioid actions
Why this matters for the opioid crisis
Opioid medicines like morphine are powerful painkillers, but they also fuel a devastating addiction epidemic. To develop better treatments, scientists need to know exactly how these drugs alter activity across the entire brain, and how those effects differ between men and women and over time. This study used cutting‑edge 3D brain imaging in mice to build a brain‑wide map of morphine’s impact on nerve cells, revealing when, where, and in whom the drug most strongly reshapes brain activity.
Seeing the whole brain at once
Instead of focusing on a few well‑known hotspots, the researchers set out to scan the entire mouse brain at single‑cell detail. After giving male and female mice an injection of either morphine or salt solution, they removed the brains one or four hours later. The brains were made optically transparent and stained for c‑Fos, a protein that lights up in recently active neurons. Using high‑speed confocal microscopes, they captured enormous 3D image files for each whole brain. A custom computing pipeline then automatically detected individual glowing cells and aligned their positions to a standard mouse brain atlas, allowing the team to count active cells in hundreds of named regions simultaneously. 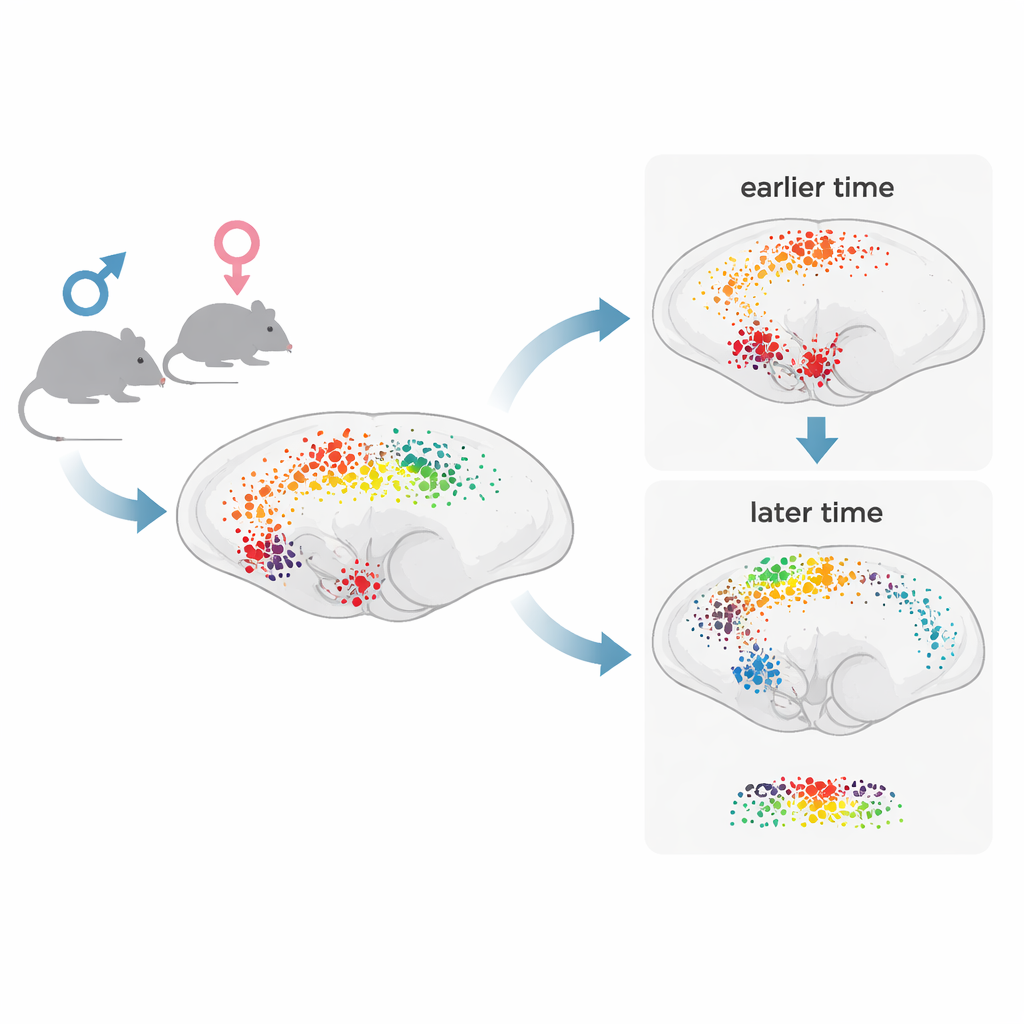
How morphine’s effects spread over time
The maps showed that a single dose of morphine broadly boosts neuronal activity compared with controls, but not all regions respond the same way or at the same moment. About two‑thirds of major brain structures showed more active cells after morphine, with especially strong responses in deep regions such as the brainstem, pons, and hypothalamus, and dense activation in certain layers of the cortex. At one hour, many of the most responsive areas were classic components of pain relief and reward circuits, such as the periaqueductal gray, nucleus accumbens, ventral tegmental area, thalamic relay nuclei, and brainstem sites involved in pain modulation and bodily functions. By four hours, fewer regions showed large changes overall, but the activity pattern had shifted: signals spread more prominently into cortical and hippocampal areas linked to learning, memory, and long‑term adaptation. This supports a “two‑wave” model in which an early surge in subcortical circuits is followed by a slower wave of cortical plasticity.
Different brain responses in males and females
The study also uncovered striking sex differences. In general, males showed stronger morphine‑induced activation than females in many key reward and stress‑related regions. These included the nucleus accumbens, lateral septum, ventral pallidum, several amygdala nuclei, bed nucleus of the stria terminalis, and parts of the hippocampus and cingulate cortex. In males, morphine mostly increased activity at both time points. In females, the picture was more complex: levels were often suppressed at one hour and higher by four hours, likely reflecting stronger stress responses to injection in the female control group and differences in how opioid receptors couple to brain cells. The data highlight that the same drug dose can engage partially different networks in male and female brains, especially in regions tied to craving, withdrawal, mood, and sleep. 
A new roadmap for studying addictive drugs
Beyond the familiar addiction hubs, the whole‑brain survey revealed many regions not usually associated with opioids, such as certain sensory and association cortices, specific thalamic and hypothalamic nuclei, brainstem structures, and even parts of the cerebellum. Many of these areas express opioid receptors but had rarely been studied in this context. By combining large‑scale imaging with machine‑learning models, the authors showed that accurately predicting whether a mouse received morphine requires information from many regions at once, underscoring how distributed the drug’s impact really is. For a layperson, the take‑home message is that morphine does not just “hit” one pleasure center—it sends ripples across vast brain networks, with timing and sex shaping which circuits are most affected. This new mapping approach offers a powerful tool for pinpointing brain targets for safer pain treatments and more effective therapies for opioid use disorder.
Citation: Vasylieva, I., Smith, R., Aravind, E. et al. Brain-wide mapping reveals temporal and sexually dimorphic opioid actions. Commun Biol 9, 466 (2026). https://doi.org/10.1038/s42003-026-09730-8
Keywords: opioid addiction, brain mapping, sex differences, morphine, neuronal activity