Clear Sky Science · en
The RNA-binding protein Squid regulates embryonic midgut development via Axin alternative splicing in Bombyx mori
Why tiny silkworm guts matter
Every animal embryo, from insects to humans, must build a working digestive system before it can grow and survive. Insects are especially interesting because their guts are both highly efficient and prime targets for pest control. This study uses the domestic silkworm, Bombyx mori, to reveal how a single RNA‑binding protein called Squid helps shape the embryonic midgut—the key region for breaking down food and absorbing nutrients. By uncovering how Squid fine‑tunes gene messages, the work sheds light on basic principles of organ formation and suggests new angles for controlling agricultural pests without heavy chemical use.
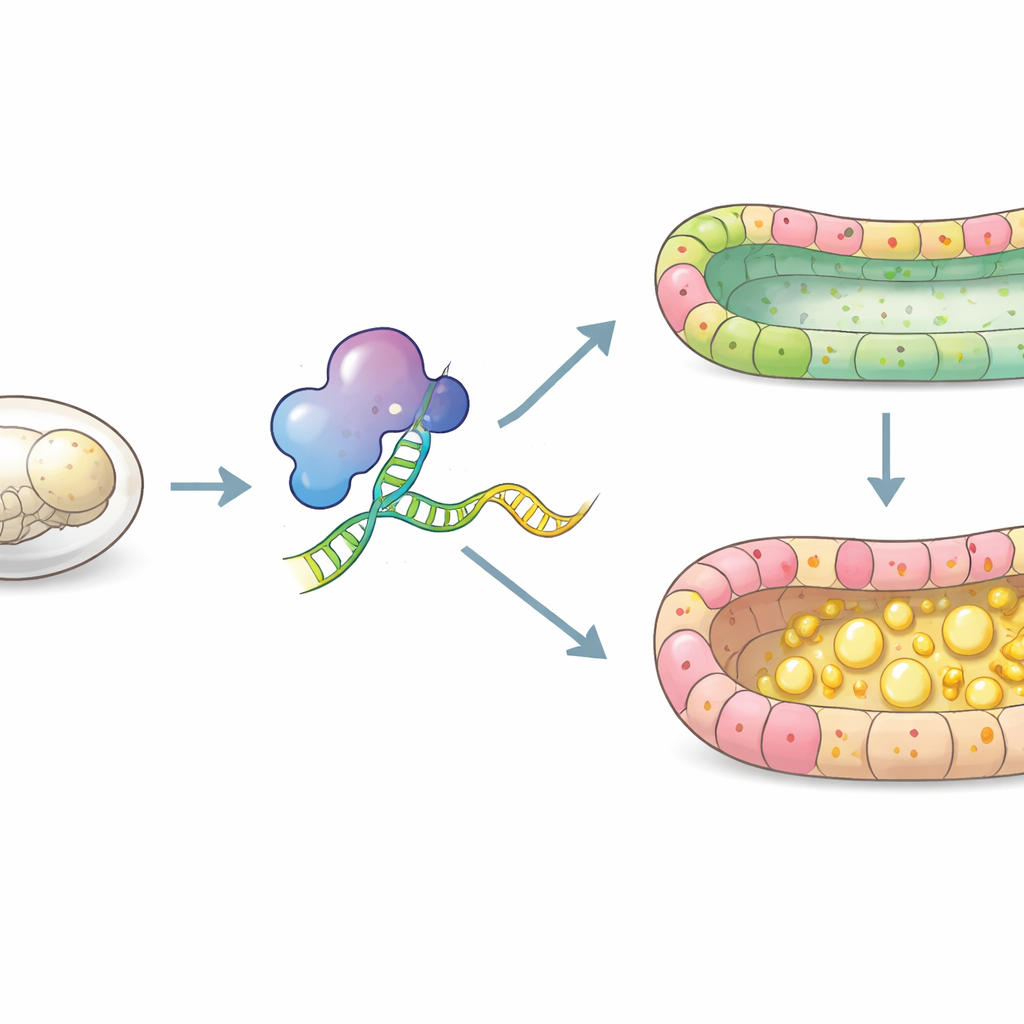
Building a functional insect gut
The insect midgut is a simple tube lined by a single layer of epithelial cells, yet it carries out digestion, nutrient uptake, immune defense, and tissue repair. In silkworm embryos, this layer is normally organized into three main cell types: intestinal stem cells that sit at the base, columnar cells that secrete digestive enzymes and absorb nutrients, and goblet cells that help manage the gut’s internal environment. The authors focused on Squid, a member of a large family of proteins that bind RNA and influence how genetic messages are edited and used in cells. Earlier work hinted that Squid was active during silkworm embryogenesis, but its precise role in gut development was unknown.
When Squid is missing, the gut falls apart
Using CRISPR/Cas9 gene editing, the researchers knocked out the Squid gene in silkworm embryos. Many embryos lacking Squid died before hatching and showed delayed development compared with normal siblings. Microscopic examination revealed that, instead of a neat midgut wall, Squid‑deficient embryos had a disorganized epithelium in which the usual cell types were hard to distinguish. The gut lumen—the central cavity where food would pass—was crowded with large lipid droplets, indicating that fats were not being processed or absorbed properly. At earlier stages, cell division appeared to stop on schedule, but the cells failed to mature into the correct specialized types, suggesting that Squid is crucial for differentiation rather than simple growth.
Energy supply and digestion stall without proper midgut
Because insect embryos rely heavily on stored egg lipids as fuel, the team asked whether the faulty midgut in Squid mutants disrupted this energy pipeline. Staining for lipids showed large amounts of fat lingering in the gut lumen of mutant embryos but not in normal ones. A broad survey of gene activity revealed that hundreds of genes involved in core metabolism—such as sugar breakdown, the citric acid cycle, and amino acid production—were dialed down in mutants. More targeted tests confirmed that transcripts encoding digestive and absorption‑related enzymes, including key steps in breaking down fats, were significantly reduced. Taken together, these findings indicate that without Squid, the embryonic midgut cannot efficiently convert stored nutrients into usable energy, contributing to embryonic death.
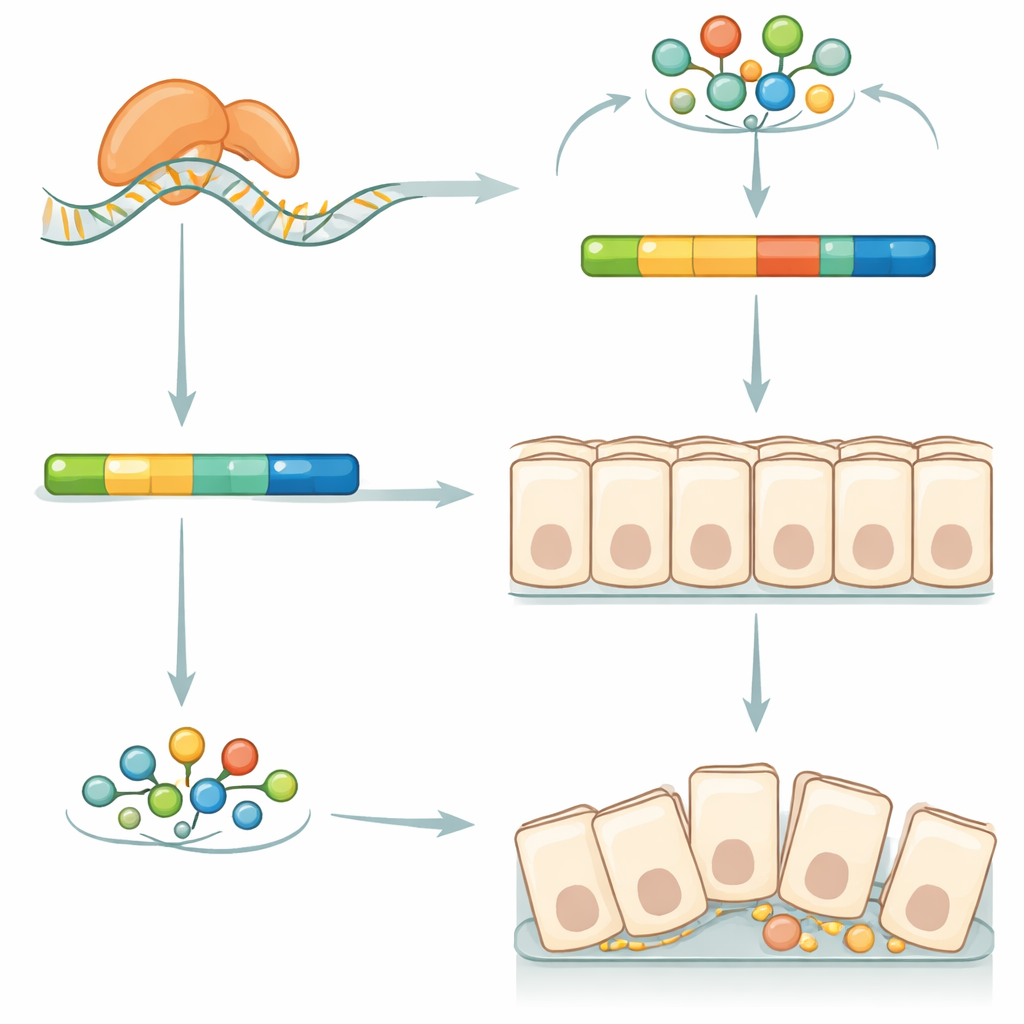
A fine control knob on a classic signaling pathway
To understand how Squid exerts such wide‑ranging effects, the authors looked for RNA molecules it physically binds. They pulled down Squid from embryo extracts and sequenced the attached RNAs, identifying over two thousand potential targets. Comparing normal and Squid‑deficient embryos showed that dozens of these targets changed how their RNA messages were spliced—the process of cutting and rejoining segments to make different protein versions. Many of the affected genes belonged to the well‑known Wnt/β‑catenin signaling pathway, which is central to gut development in many animals. One standout target was Axin, a scaffold protein that helps regulate the stability of β‑catenin, a key signal and structural component in epithelial tissues.
How changing one splice choice reshapes the gut
Axin exists in two forms in silkworms: a long version and a shorter version missing a small low‑complexity segment. In Squid‑deficient embryos, overall Axin levels stayed the same, but the balance shifted away from the long variant toward the short one. This shift coincided with a sharp drop in β‑catenin protein in the midgut epithelium. When the team overexpressed the long Axin form in cultured silkworm cells, β‑catenin levels rose; the short form had little effect. These experiments suggest that Squid normally promotes production of the long Axin variant, which in turn helps maintain appropriate β‑catenin levels to support cell‑cell junctions and proper organization of the midgut layer. Disrupting this delicate splicing control appears to undermine the stem cell niche, block normal differentiation, and ultimately compromise the entire organ.
What this means beyond silkworms
For non‑specialists, the central message is that the way cells edit their RNA messages can determine whether an organ forms correctly. In silkworm embryos, the RNA‑binding protein Squid ensures that a key signaling component, Axin, is spliced into the version that keeps β‑catenin at healthy levels in the midgut. When Squid is removed, this balance is lost, the gut epithelium becomes disordered, nutrients are not absorbed, and the embryo dies. Because similar RNA‑binding proteins and Wnt/β‑catenin signaling operate in many animals, including humans, this work highlights how subtle changes in RNA processing can have profound effects on tissue development, health, and potentially on strategies to manage insect pests.
Citation: Tong, C., Mo, W., Cai, M. et al. The RNA-binding protein Squid regulates embryonic midgut development via Axin alternative splicing in Bombyx mori. Commun Biol 9, 449 (2026). https://doi.org/10.1038/s42003-026-09692-x
Keywords: embryonic midgut development, RNA binding proteins, alternative splicing, Wnt beta-catenin signaling, silkworm Bombyx mori