Clear Sky Science · en
Dopamine neuron specific RNA-sequencing reveals Neprilysin 1 acts downstream of the cohesin complex to suppress learning
Why this matters for everyday memory
We usually think of better memory as something to be trained or drugged into existence. This study in fruit flies suggests another possibility: the brain may come with built-in brakes that deliberately hold learning back, and some of those brakes are set during early development yet can be adjusted later in life. By uncovering one such brake, the authors provide clues to how normal memory is tuned and why certain genetic disorders cause intellectual disability.
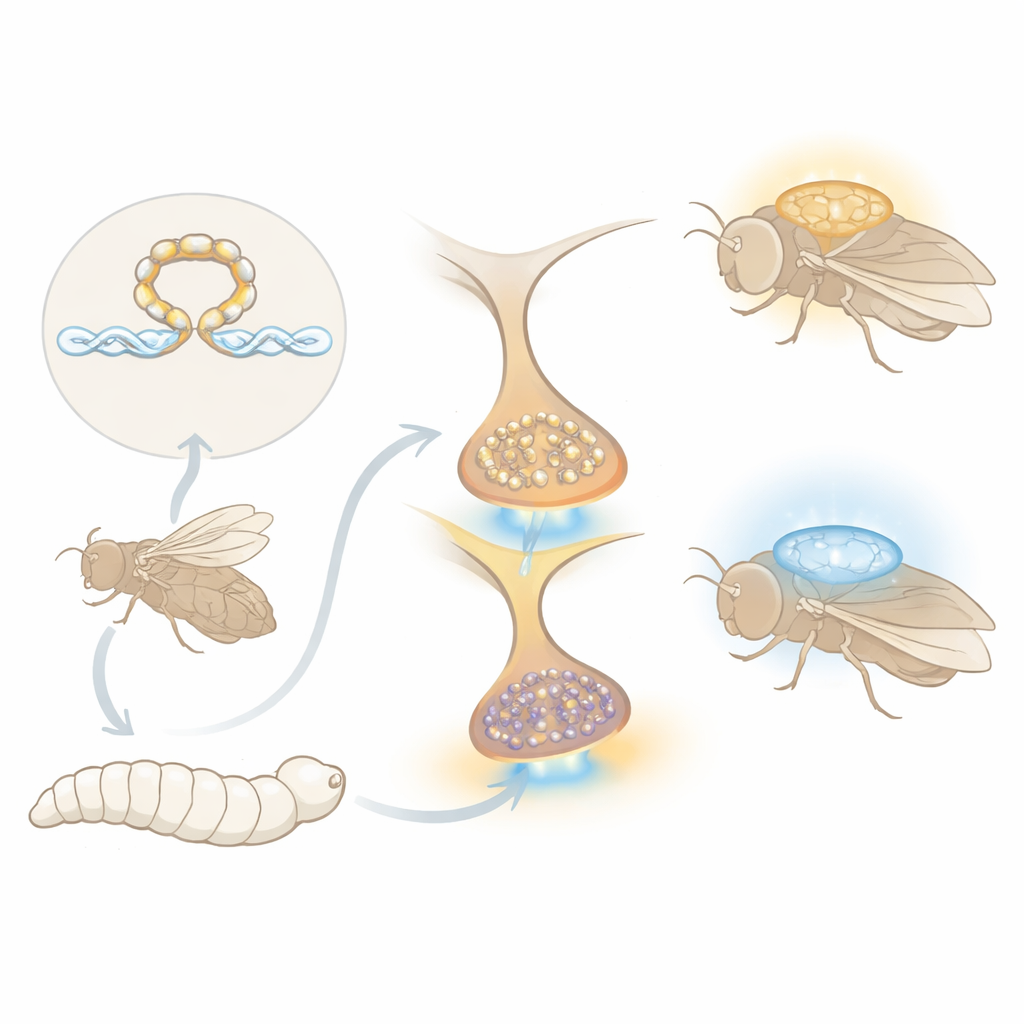
A molecular brake on learning
Previous work from this group identified a protein called Stromalin as a surprising memory suppressor. Stromalin is part of the cohesin complex, best known for holding sister chromosomes together during cell division, but it also helps control which genes are turned on or off. In fruit flies, reducing Stromalin in a small set of dopamine-producing neurons roughly doubled the number of tiny chemical packets, called synaptic vesicles, at their connections, leading to stronger dopamine release and better learning in an odor–shock task. What remained mysterious was how a gene-regulation complex that acts in the cell nucleus could dictate how many vesicles are built for future communication.
Reading the message in dopamine cells
To bridge this gap, the researchers isolated just 25 dopamine neurons from developing fly larvae and sequenced their RNA, a snapshot of which genes are active. Comparing normal cells with those lacking Stromalin, they found 160 genes whose activity changed. They then used a large-scale genetic screen to switch off each of these candidate genes specifically in dopamine neurons and asked two questions: did flies learn better, and did the neurons show more synaptic vesicle marker at their terminals? This two-step filter narrowed the field to a handful of genes whose loss mimicked Stromalin’s effects on memory and synaptic markers.
Spotlight on Neprilysin 1
Among the candidates, one gene stood out: Neprilysin 1 (Nep1), which encodes a membrane enzyme that cuts small signaling peptides outside cells. Using an independent method to measure gene activity across the whole brain, the team confirmed that reducing either Stromalin or another cohesin subunit, SMC1, consistently lowered Nep1 levels. When they knocked down Nep1 only in dopamine neurons, flies learned faster and remembered better, and their dopamine terminals carried more synaptic vesicle marker both during late larval stages and in adults. Direct imaging of dopamine release showed that these neurons kept releasing strong pulses of dopamine through repeated shocks, instead of adapting and weakening as in normal flies. Crucially, tamping down vesicle transport with a motor-protein mutation erased the Nep1-induced boost in learning and synaptic markers, implying that Nep1 normally acts to limit the available vesicle pool.
Resetting the brake downstream of cohesin
To test whether Nep1 truly lies downstream of Stromalin, the authors overproduced Nep1 while also reducing Stromalin. In dopamine neurons, this combination restored both synaptic vesicle markers and memory performance back toward normal, counteracting the enhancement seen with Stromalin loss alone. Similar rescues appeared when these manipulations were extended across the entire brain. Interestingly, while cohesin’s influence on Nep1 levels seems to be set during a critical larval window, reducing Nep1 activity only in adulthood was still enough to enhance learning, showing that the brake can be adjusted after development. At the same time, cutting Nep1 or SMC1 across all neurons impaired memory, echoing cognitive problems seen in people with cohesin-related syndromes.
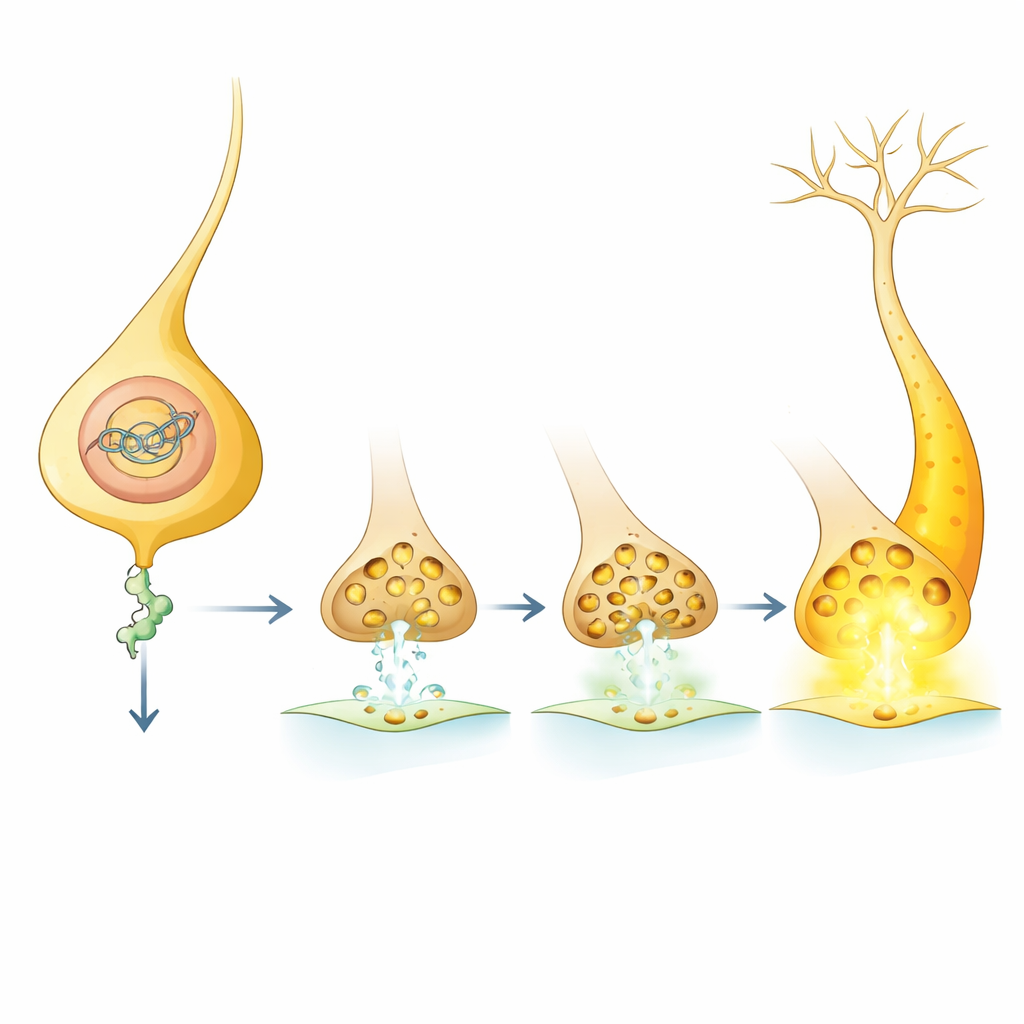
What this means for understanding and treating memory problems
In everyday terms, cohesin acts like a developmental dial that sets how strongly certain dopamine pathways can talk to downstream brain centers by tuning levels of Nep1. When cohesin function drops, Nep1 levels fall, more synaptic vesicles accumulate, and dopamine signals become stronger, making flies better learners in some circuits but harming others when changes are widespread. Because Nep1 can still shape learning when manipulated only in adults, the work suggests that some consequences of early gene-regulation defects might be softened later by targeting downstream players like Nep1. While these results come from fruit flies, they resonate with findings in mouse models and human patients, hinting that finely adjusting similar molecular brakes could one day help rebalance learning and memory in developmental brain disorders.
Citation: Pimenov, I., MacMullen, C.M., Ezeh, C. et al. Dopamine neuron specific RNA-sequencing reveals Neprilysin 1 acts downstream of the cohesin complex to suppress learning. Commun Biol 9, 441 (2026). https://doi.org/10.1038/s42003-026-09690-z
Keywords: memory suppressor genes, dopamine neurons, synaptic vesicles, cohesin complex, neprilysin