Clear Sky Science · en
PRC2 loss impairs small cell lung cancer tumorigenesis and enhances sensitivity to G9a/GLP inhibition
Why this research matters for people with lung cancer
Small cell lung cancer is one of the deadliest forms of lung cancer, often spreading quickly and resisting current treatments such as chemotherapy and immunotherapy. This study asks a simple but powerful question: instead of only attacking the cancer’s DNA, what if we could also rewire the switches that tell cancer cells which genes to turn on or off? By doing so, the researchers uncovered a new weakness in small cell lung cancer that may make existing drugs more effective and open the door to smarter combination therapies.
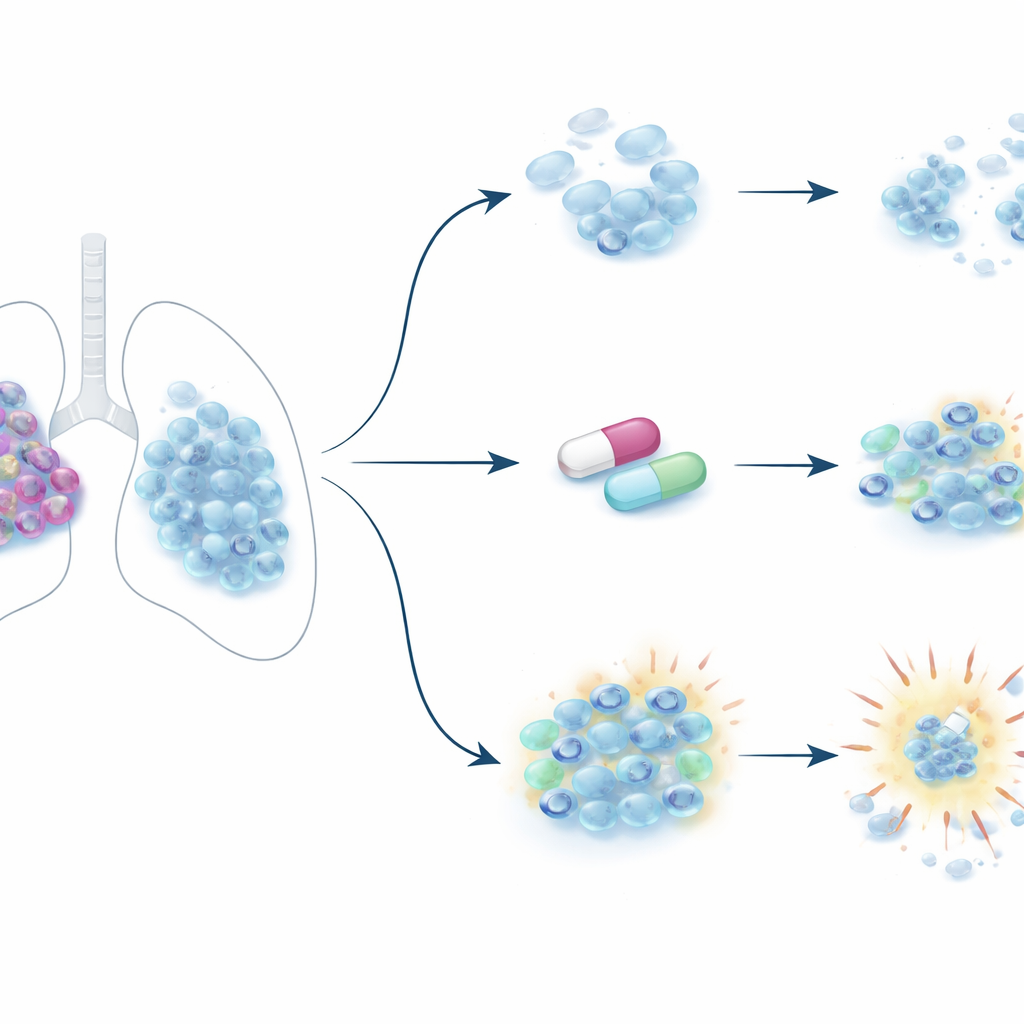
A deadly cancer that hides from the immune system
Small cell lung cancer is usually diagnosed late and has a five-year survival rate below 7 percent. Many tumors keep a “neuroendocrine” identity, meaning they resemble hormone-producing nerve cells. These tumors tend to grow as loose, floating clusters, hide from the immune system, and quickly become resistant to chemotherapy. Only a small fraction of patients have tumors that are inflamed and visible to immune cells, which is why immune checkpoint drugs help so few people. Because of this, scientists are turning their attention to the cancer’s epigenome—the chemical tags on DNA and its packaging proteins that act like software for the genome—to see whether resetting these tags can make tumors both less aggressive and easier to kill.
A master gene-silencing complex with two faces
The study focuses on a protein machine called PRC2, which normally shuts down sets of genes by placing chemical marks on the proteins around which DNA is wrapped. PRC2 has two key aspects: its physical structure, which holds the complex together, and its enzyme activity, which actually adds the silencing marks. Using mouse models engineered to develop small cell lung cancer, the researchers removed a core structural part of PRC2 called EED right at the start of tumor formation. Without this component, the expected lung tumors almost never formed, and the rare lesions that did appear grew poorly and lacked typical neuroendocrine features. Cancer cells that lost EED died off, and even a drug designed to break down EED selectively killed both mouse and human small cell lung cancer cells while sparing lung adenocarcinoma cells. This showed that the structural integrity of PRC2 is essential for both starting and maintaining this cancer.
Enzyme blocking alone is not enough
The team then asked whether drugs that block PRC2’s enzyme activity—specifically EZH2 inhibitors that are already approved for some blood cancers—could mimic the effect of removing EED. In cultured small cell lung cancer cells, these drugs efficiently erased the chemical marks written by PRC2 but had only modest effects on cell survival over a few days. In mouse tumor grafts, long-term treatment lowered these marks in the tumors but did not help the animals live longer. However, when the drugs were given for an extended period in cell culture, they caused much broader shifts in gene activity, including increased expression of genes involved in cell-surface markers and immune recognition. The cancer cells also began to lose their neuroendocrine traits and adopt a more adherent, less floating growth style, suggesting that prolonged enzyme inhibition slowly pushes the cells into a different, more vulnerable state rather than killing them outright.
Uncovering a drug-sensitive weak spot
To exploit this new state, the researchers performed a focused screen combining EZH2 inhibitors with a panel of other drugs. They found that after pretreatment with an EZH2 inhibitor, neuroendocrine small cell lung cancer cells became highly sensitive to blocking another pair of enzymes, G9a and GLP, which also control gene silencing through different chemical marks. This hypersensitivity was seen only in neuroendocrine small cell lung cancer lines, not in related non-neuroendocrine lines or in mesothelioma cells. In mice carrying human small cell lung cancer grafts, the G9a/GLP inhibitor alone had a modest survival benefit, the EZH2 inhibitor alone did not, but the combination extended survival more substantially. Molecular analyses showed that EZH2 inhibition rewired the network of proteins interacting with PRC2 and, together with G9a/GLP inhibition, strongly boosted genes involved in oxidative stress and cellular oxidases, including enzymes that drive lipid oxidation.
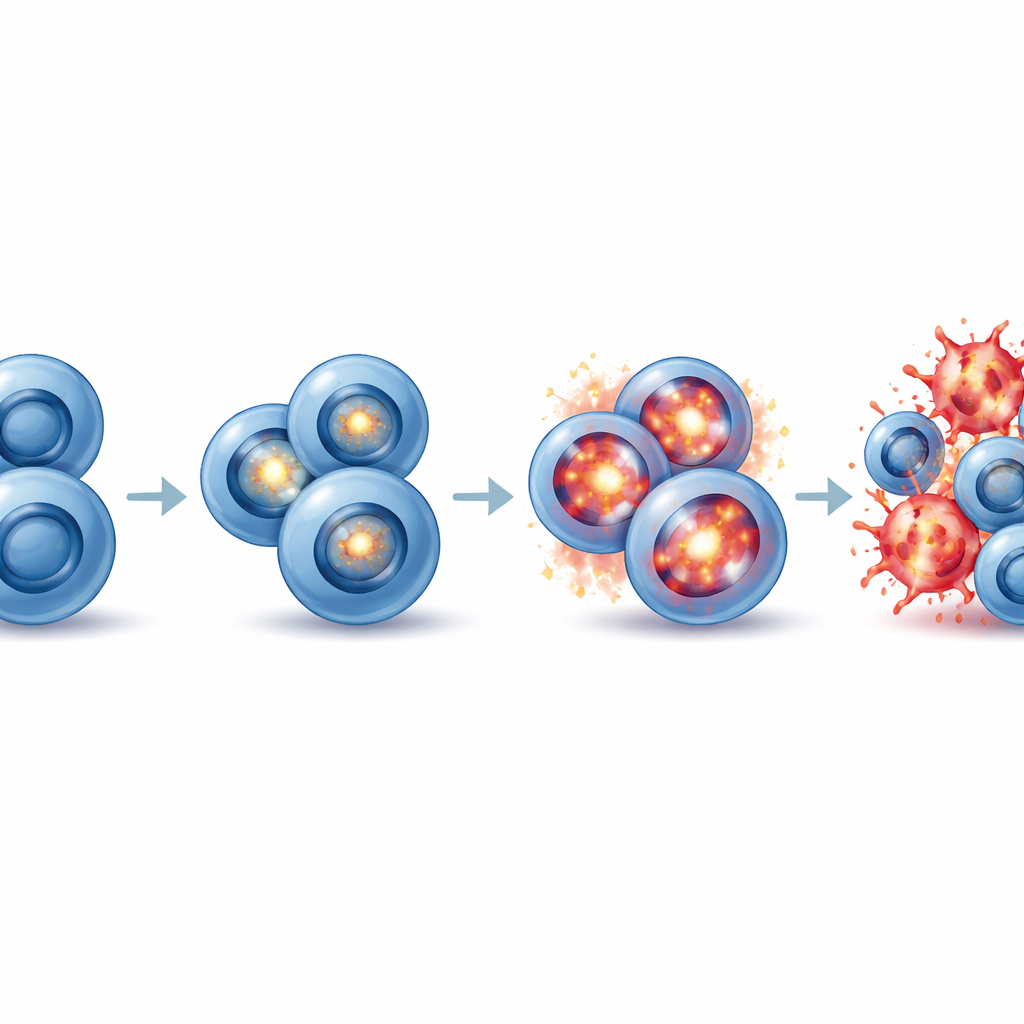
Forcing cancer cells into a fatal oxidative overload
Oxidative stress arises when reactive oxygen molecules build up faster than cells can neutralize them, damaging fats, proteins, and DNA. The study showed that the combination of EZH2 and G9a/GLP inhibition triggers a sharp rise in oxidative stress specifically in neuroendocrine small cell lung cancer cells. Markers of oxidative damage increased, and direct measurements with a fluorescent probe confirmed higher levels of reactive oxygen species only in the combination-treated neuroendocrine cells. When the researchers added a common antioxidant, N-acetyl-L-cysteine, it rescued the cells from death, proving that oxidative stress was the main cause of their demise. Non-neuroendocrine cells did not experience this surge, explaining why they were spared. In essence, prolonged EZH2 inhibition “primes” neuroendocrine small cell lung cancer cells so that a follow-up hit to G9a/GLP overwhelms their stress defenses and pushes them over an oxidative cliff.
What this means for future treatments
For a layperson, the key message is that this work separates two roles of the same gene-silencing machine in small cell lung cancer. The structural core of PRC2, including EED, is absolutely required for this cancer to exist at all, making it an attractive target for new degrader drugs that physically dismantle the complex. In contrast, simply turning off the enzyme portion with existing inhibitors does not stop tumors by itself, but it does reshape the cancer cells into a state that exposes a hidden weakness: a dependence on tightly controlled oxidative stress. By pairing prolonged EZH2 inhibition with G9a/GLP inhibitors, the researchers were able to exploit this weakness and selectively kill hard-to-treat neuroendocrine tumors through oxidative overload. This concept of first nudging cancer cells into a more vulnerable identity and then striking their new Achilles’ heel could help design smarter, more durable treatment strategies for patients with this aggressive disease.
Citation: Kopparam, J., Chandrasekaran, G., Hulsman, D. et al. PRC2 loss impairs small cell lung cancer tumorigenesis and enhances sensitivity to G9a/GLP inhibition. Commun Biol 9, 469 (2026). https://doi.org/10.1038/s42003-026-09677-w
Keywords: small cell lung cancer, epigenetic therapy, PRC2, EZH2 inhibitor, oxidative stress