Clear Sky Science · en
SOX9 represses the human galectin-3 promoter in SW1353 cells: potential implications for osteoarthritis
Why this matters for aching joints
Osteoarthritis, the most common form of arthritis, slowly wears away the cushioning cartilage in our joints and leaves millions of people in daily pain. One protein, galectin-3, has emerged as a key troublemaker that stirs up inflammation and speeds cartilage breakdown. This study asks a deceptively simple question: what inside our cells turns the galectin-3 gene up or down? By uncovering how a cartilage "identity" factor called SOX9 reins in galectin-3, the work points to a deeper wiring diagram of osteoarthritis and hints at new ways to calm the disease at its source. 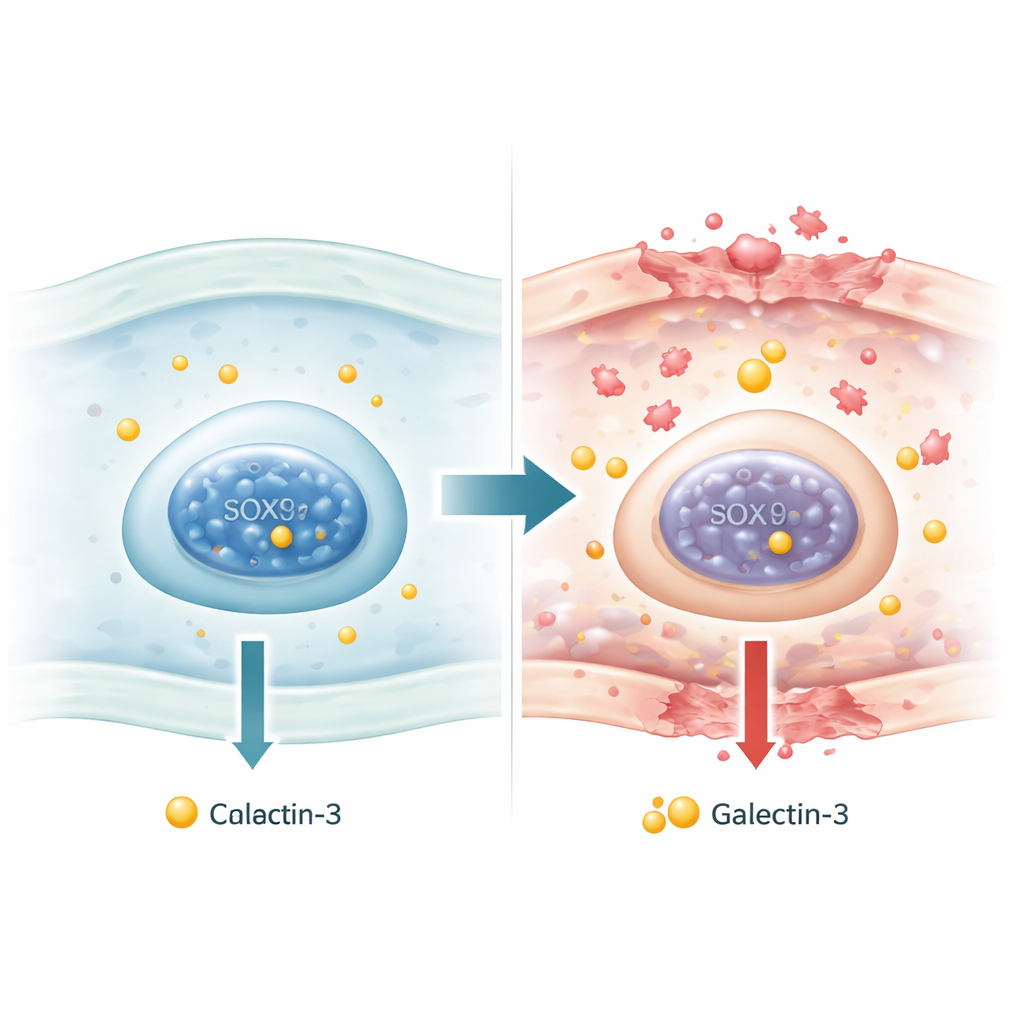
A troublemaking protein in worn-out cartilage
Galectin-3 is a sugar-binding protein found in many tissues and species. In people with osteoarthritis and early rheumatoid arthritis, its levels in blood and joint cartilage are often elevated. In damaged knee cartilage, cells that produce more galectin-3 tend to sit in areas with the worst tissue destruction. Experiments have shown that galectin-3 outside the cell can latch onto cartilage cells, trigger inflammatory signals, and switch on enzymes that chew up the surrounding matrix. Yet, despite this central role, it has not been clear why the galectin-3 gene (called LGALS3) becomes overactive in diseased cartilage in the first place.
Cartilage’s identity keeper steps into the picture
Cartilage cells, or chondrocytes, are guided by a family of DNA-binding proteins known as SOX factors. Among them, SOX9 is often described as a master regulator: it helps chondrocytes make and maintain the collagen and gel-like molecules that give cartilage its spring. In healthy cartilage, SOX9 works together with its partners SOX5 and SOX6 to uphold this program. In osteoarthritis, however, this network becomes unbalanced: SOX5 and SOX6, and especially SOX9, are reduced, while another family member, SOX4, is increased. Because SOX proteins can bend DNA and remodel the local landscape of genes, the authors reasoned that shifts in SOX activity might directly alter how strongly chondrocytes express the galectin-3 gene.
Zooming in on the gene’s on–off switch
To test this idea, the researchers used a human chondrocyte-like cell line called SW1353 and built a series of DNA fragments covering the region in front of the LGALS3 gene, where the cell’s transcription machinery docks. They fused these fragments to a luciferase reporter, a light-producing gene that glows in proportion to promoter activity. By trimming away pieces from the far upstream region toward the gene, they discovered that a short stretch of DNA spanning just 149 base pairs around the start site (from −97 to +52) was sufficient to drive strong activity. Removing smaller portions within this zone caused stepwise drops in signal, revealing that key control elements, packed with GC-rich motifs that attract other transcription factors, lie in this compact segment.
How SOX9 pushes the brakes on galectin-3
Next, the team asked how different SOX proteins affect this promoter. When they artificially increased SOX2 or SOX9, the LGALS3 promoter activity fell sharply in a dose-dependent manner, while SOX4, SOX5, and SOX6 showed no consistent effect. Further tests suggested that SOX2 acts in a non-specific way and is not relevant for cartilage biology, whereas SOX9 shows targeted control. SOX9 reduced activity most strongly when the full-length promoter was present, but repression remained clear even in the minimal −97/+52 fragment. Using a chromatin pull-down method, the authors demonstrated that SOX9 physically binds within a −93/+49 region of the LGALS3 promoter. Mutating a predicted SOX9 recognition motif in this segment lowered the baseline activity of the promoter and slightly weakened SOX9’s repressive effect, indicating that this site is important but not the sole contributor. Importantly, when SOX9 was overexpressed in the cells, endogenous LGALS3 messenger RNA levels dropped by about 30%, confirming that this wiring operates on the native gene, not just an artificial reporter. 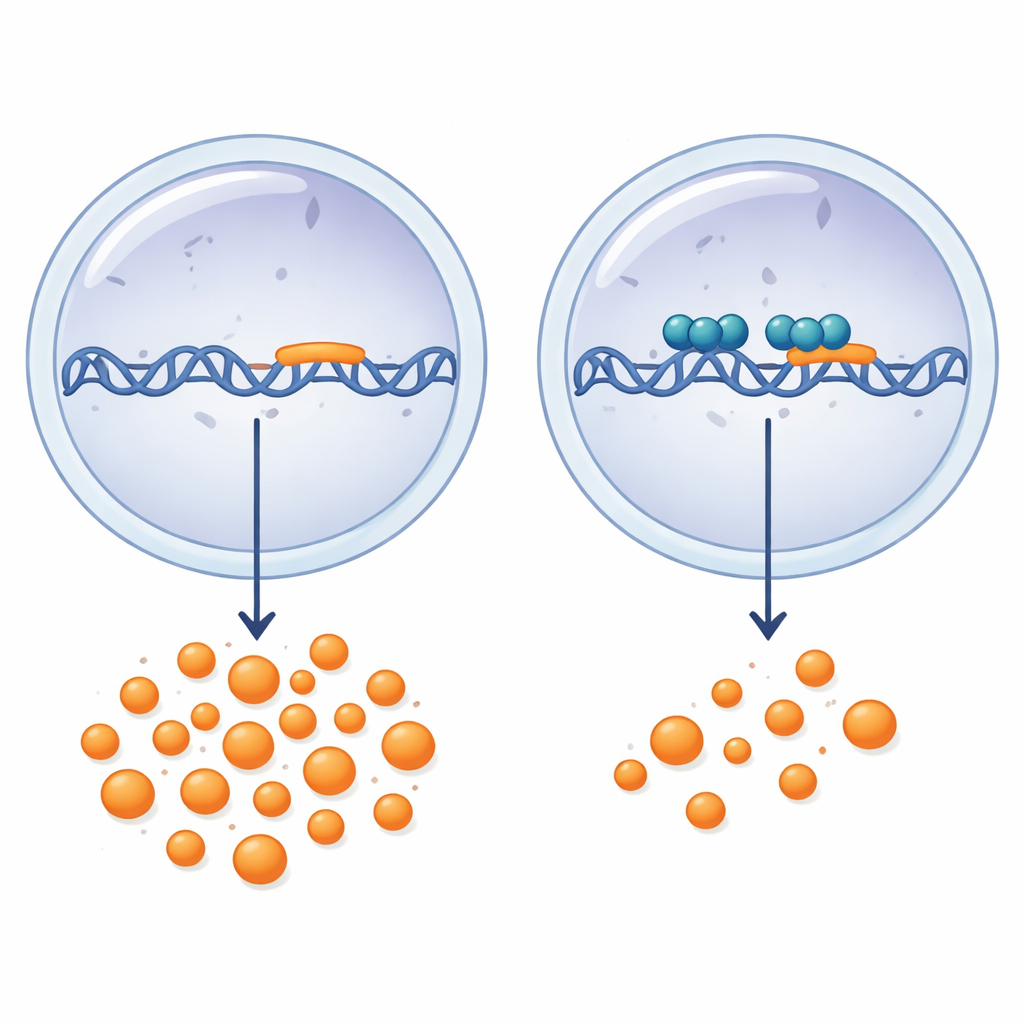
What this means for understanding and treating osteoarthritis
Taken together, the findings reveal a compact core promoter that controls the human galectin-3 gene and identify SOX9 as a direct brake on this switch in cartilage-like cells. In osteoarthritis, where SOX9 levels are typically reduced, the loss of this brake could help explain why galectin-3 becomes overabundant and drives inflammation and matrix degradation. While this study was performed in a cancer-derived cell line and needs validation in primary cartilage cells and animal models, it maps a crucial link between a cartilage identity factor and a damaging inflammatory mediator. In the longer term, strategies that restore or mimic SOX9’s restraining influence on LGALS3—whether via gene regulation, epigenetic drugs, or RNA-based therapies—might help lower galectin-3 levels and slow the joint damage that underlies osteoarthritis pain.
Citation: Alba, B., Buddiga, S., Kaltner, H. et al. SOX9 represses the human galectin-3 promoter in SW1353 cells: potential implications for osteoarthritis. Sci Rep 16, 14284 (2026). https://doi.org/10.1038/s41598-026-50507-0
Keywords: osteoarthritis, galectin-3, SOX9, cartilage, gene regulation