Clear Sky Science · en
Solvatochromic, spectroscopic, DFT calculations, antimicrobial and docking studies of new Fe(III), Co(II), and Ni(II) chelates containing 1,2,4-triazine
New Molecules That Fight Germs and Glow
Chemists have created a family of tiny metal-based molecules that can both light up under certain conditions and slow the growth of harmful microbes. These dual-purpose materials are interesting because they hint at future drugs that can be tracked inside the body by their glow, or smart coatings that both sense and kill bacteria and fungi on medical devices.
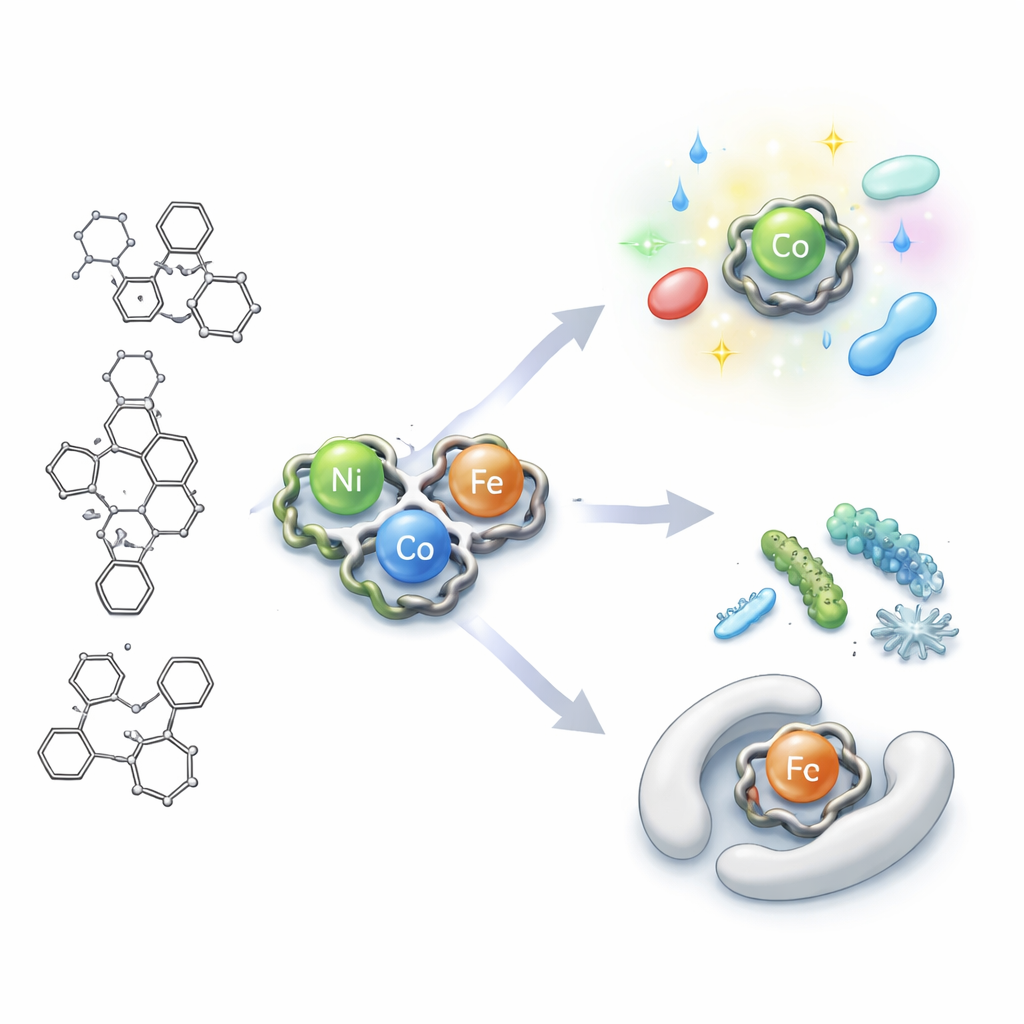
Building Tiny Metal Cages
The team started from a carbon-based framework known for its rich biological activity and ability to grab onto metals. They modified it to make a claw-like unit (a hydrazone–triazine ligand) that can attach to metal ions at three points, using two nitrogen atoms and one oxygen atom. When they mixed this claw with nickel, cobalt, or iron salts, the pieces snapped together into three new metal “chelates”: one with nickel, one with cobalt, and one with iron. A battery of tests—including infrared and visible-light spectroscopy, magnetism, and thermal analysis—showed how the atoms are arranged. The nickel version adopts a roughly four-cornered (tetrahedral) shape, while the cobalt and iron versions prefer a six-cornered (octahedral) arrangement around the metal centers.
From Nanoparticles to Light-Responsive Materials
Using X-ray diffraction and transmission electron microscopy, the researchers found that the complexes form particles just a few billionths of a meter across, with the nickel compound appearing as tiny spheres and cubes. All three complexes emit light and change their color response depending on the surrounding liquid. By recording how their absorption and fluorescence signals shift in solvents of different polarity, the authors could estimate how the internal charge distribution changes between the ground and excited states. The data show that when these molecules are excited, their internal charge moves significantly, making them more polar and more easily influenced by their environment. This behavior is valuable for sensors and optical devices, because it means the complexes can translate subtle environmental changes into visible color or brightness shifts.
Probing Behavior with Computer Models
To complement the experiments, the team turned to quantum-chemical calculations. They used density functional theory to optimize the shapes of the complexes and to examine the highest occupied and lowest unoccupied molecular orbitals, which control how electrons move during reactions and light absorption. The iron complex showed the smallest energy gap between these orbitals, suggesting it is the most chemically reactive of the three. The models also mapped regions of negative and positive charge across each molecule, highlighting the same nitrogen and oxygen atoms identified experimentally as key contact points. Importantly, calculated values of polarizability and hyperpolarizability indicate that these complexes respond strongly to electric fields, far more than a standard reference compound. That means they are promising candidates for nonlinear optical applications such as advanced photonic switches and signal-processing components.
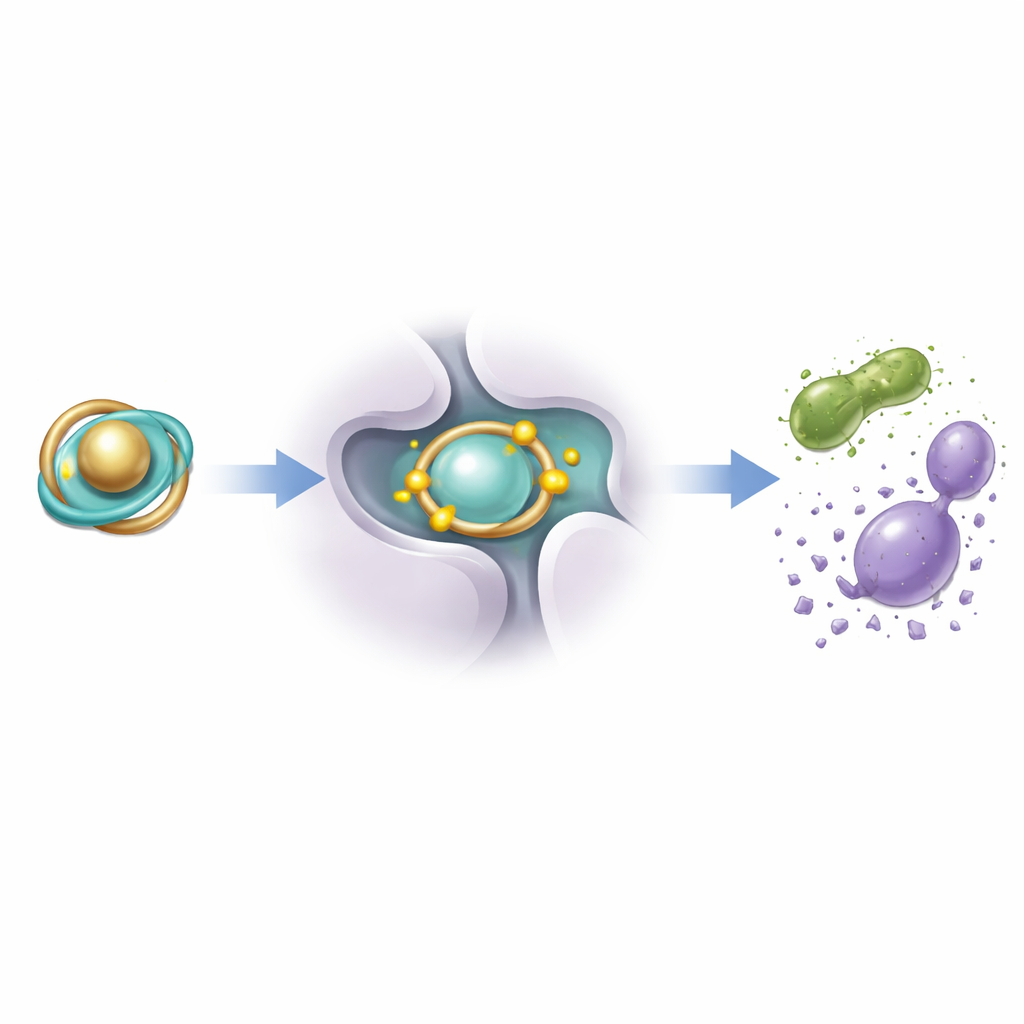
Challenging Bacteria and Fungi
The researchers then tested how well the new complexes could slow or stop the growth of common disease-causing microbes: the bacteria Staphylococcus aureus and Escherichia coli, and the fungus Candida albicans. The free ligand alone showed only weak effects, but its metal chelates were much more potent. The iron complex in particular produced large clear zones where microbes could not grow and worked at relatively low concentrations, with strong activity against both bacteria and fungi. To understand why, the team used molecular docking simulations, virtually placing each complex into the active pocket of a bacterial enzyme crucial for fatty-acid synthesis. The iron complex nestled most snugly into this pocket and formed the strongest simulated interactions, mirroring its superior antimicrobial performance in the lab.
What These Findings May Mean
Taken together, the work introduces three new nanometer-scale metal complexes that combine strong antimicrobial activity with sensitive light-based responses and robust thermal stability. For a non-specialist, the key message is that small changes in how organic claws grip metal ions can tune not only how these particles glow, but also how effectively they interfere with vital processes in bacteria and fungi. The iron-containing complex stands out as the best all-around performer, pointing toward future materials that can simultaneously detect their surroundings, carry out optical functions, and act as targeted antimicrobial agents in medicine or smart coatings.
Citation: Abdelrhman, E.M., Samy, F., Adly, O.M. et al. Solvatochromic, spectroscopic, DFT calculations, antimicrobial and docking studies of new Fe(III), Co(II), and Ni(II) chelates containing 1,2,4-triazine. Sci Rep 16, 13406 (2026). https://doi.org/10.1038/s41598-026-48416-3
Keywords: metal chelates, luminescent antimicrobials, triazine hydrazone, nonlinear optical materials, molecular docking