Clear Sky Science · en
Milk fat globule EGF and factor V/VIII domain containing (MFGE8) as a novel player in equine endometrial fibrosis
Why this matters for horse owners and breeders
Many mares become less fertile as they age, often for reasons that remain hidden until pregnancy repeatedly fails. This study looks inside the uterus of older mares to understand one common culprit: scarring of the uterine lining, known as endometrial fibrosis. The authors focus on two messenger proteins that seem to pull this process in opposite directions—one that drives scar formation and another that may help to hold it back—offering clues toward future treatments that could preserve fertility in aging horses.
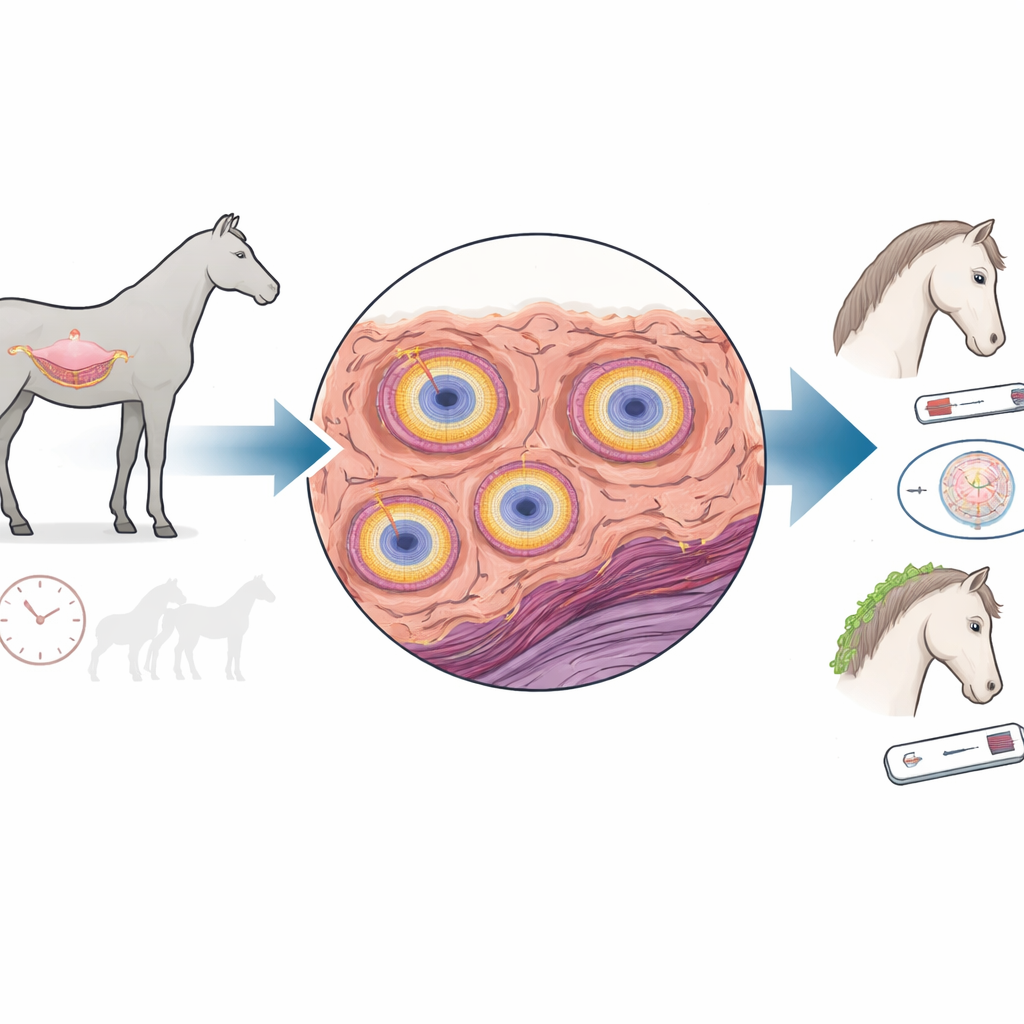
Scars in the womb and lost fertility
In a healthy mare, tiny glands in the uterine lining release nourishing fluids that support the earliest stages of pregnancy. With age, these glands in many mares become wrapped in layers of scar tissue and stiff material between cells. This process, called fibrosis or endometrosis, disrupts gland function and is strongly linked to infertility. The scarring arises when normal wound-healing runs out of balance: repair cells called fibroblasts transform into more forceful “myofibroblasts” that lay down excess collagen and other building blocks of scar tissue, and then fail to switch off.
Two molecular signals with opposite tendencies
The team focused on transforming growth factor beta 1, a well-known driver of scarring in many organs, and MFGE8, a lesser-known protein that in other tissues often acts as a brake on fibrosis. Using a technique that highlights where specific genes are active in tissue slices, they mapped four key players in mare uteruses: the scarring signal, its helpers CCN2 and TAGLN, and MFGE8. They found that when the scarring signal was strongly active in gland areas, its helpers were almost always active as well, and the surrounding tissue showed many layers of fibroblast-like cells and heavy scarring. In contrast, MFGE8 was most strongly seen around glands that were already fibrotic, marking it as closely tied to the disease, though its exact role was not immediately clear from location alone.
Zooming in on individual cells
To see how these signals change cell behavior, the researchers grew uterine fibroblasts from several mares on soft, collagen-coated gels that better mimic real tissue than standard plastic lab dishes. They then exposed the cells either to the scarring signal or to MFGE8 and examined gene activity cell by cell using single-cell sequencing. The scarring signal caused sweeping changes across nearly all fibroblast groups: genes for collagen and other matrix components surged, factors that promote growth and survival increased, and protective immune-style signals known as interferons were dampened. Together, these shifts paint the picture of a tissue environment primed to stiffen and scar.
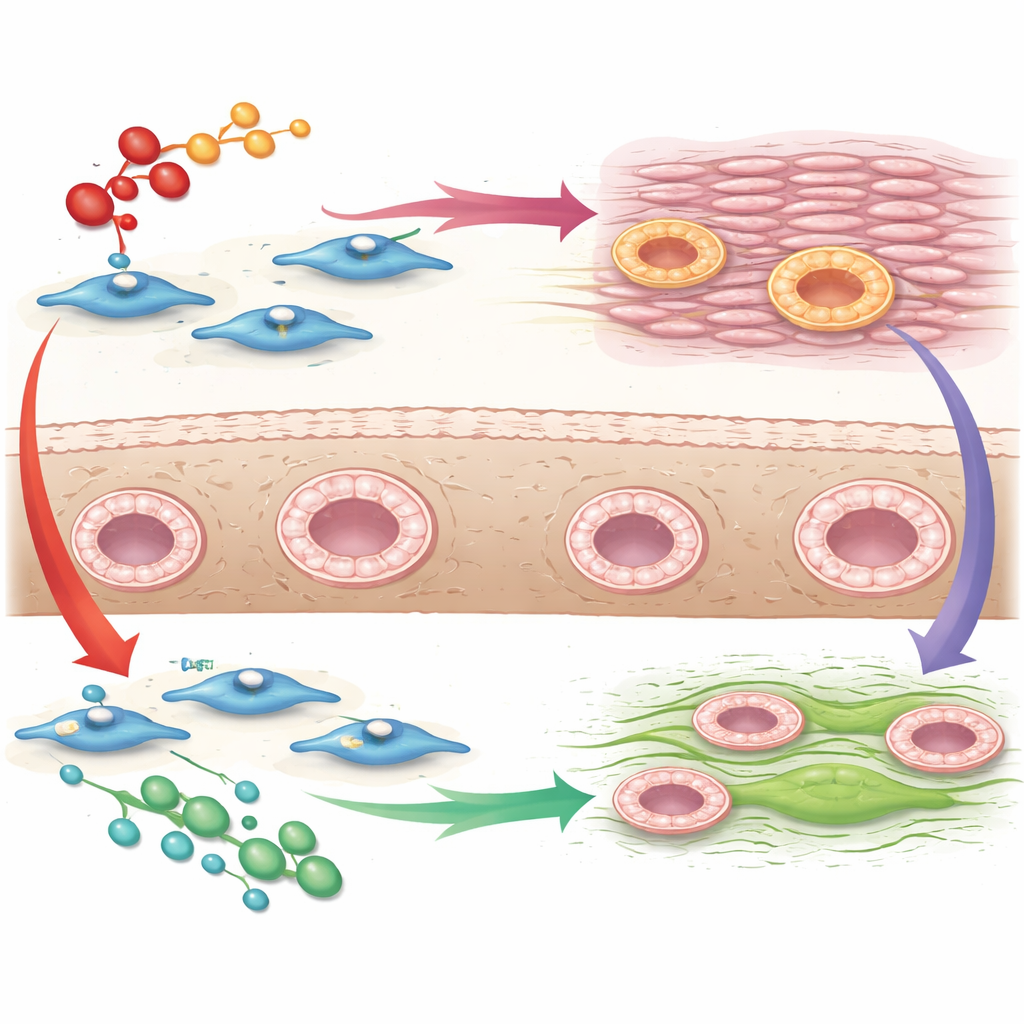
A subtle but promising counterbalance
The influence of MFGE8 was more modest and appeared strongest in a distinct subgroup of fibroblasts. In these cells, gene patterns pointed toward slowed growth and movement, less activation of pathways linked to unchecked scarring, and more activity in a regulatory route centered on a protein called PTEN, which in other organs helps limit scar-forming behavior. MFGE8 also nudged some fibroblast groups to increase type I interferon signals—pathways that other studies have shown can restrain fibrosis—and to decrease levels of LIF, a molecule tied to harmful scarring in kidneys and lungs. Not every change pointed cleanly in an anti-scarring direction, but the overall picture suggested that MFGE8 tends to counter, rather than amplify, the runaway repair triggered by the scarring signal.
What this means for future mare care
Taken together, the work reinforces the scarring signal as a major driver of uterine damage in aging mares and highlights TAGLN as a practical marker of actively scar-forming cells. At the same time, the tight link between MFGE8 and fibrotic areas, plus its cell-level effects that favor restrained growth and more balanced signaling, identify it as a new and intriguing player that may help limit fibrosis rather than cause it. While these findings are early and based on laboratory models, they lay the groundwork for future strategies that might tip the balance in the mare’s uterus away from scarring and toward healthy repair, potentially extending the fertile years of valuable broodmares.
Citation: zu Klampen, E., Neufeld, G. & Klein, C. Milk fat globule EGF and factor V/VIII domain containing (MFGE8) as a novel player in equine endometrial fibrosis. Sci Rep 16, 12045 (2026). https://doi.org/10.1038/s41598-026-46595-7
Keywords: mare fertility, uterine scarring, fibrosis, growth factors, single-cell analysis