Clear Sky Science · en
N2-(1-carboxyethyl)-2′-deoxyguanosine as a genotoxic glycation marker associated with chromatin architectural alterations in oral squamous cell carcinoma
Why this matters for everyday tobacco users
Oral cancer is a major and growing health problem in South Asia, especially among people who use smokeless tobacco products like gutka, naswar, paan, and khaini. This study looks beneath the microscope to ask a simple but urgent question: what exactly are these products doing to the DNA inside the cells of the mouth? The researchers focus on a special kind of chemical scar on DNA, called CEdG, and show how it links everyday tobacco habits to deep changes in the way our genetic material is packed and organized inside cells.
A hidden chemical in common chewing products
Smokeless tobacco is made through drying, curing, and fermenting leaves. These steps do more than add flavor: they generate reactive chemicals known as glycotoxins. One of them, methylglyoxal, can latch onto DNA without any help from enzymes. When it reacts with a DNA building block called guanine, it forms a stable lesion called CEdG. Earlier work in other diseases showed that CEdG can damage DNA and promote genetic errors, but its role in oral cancer had not been explored. In this study, the authors first measured methylglyoxal in popular smokeless tobacco brands and found high levels in widely used products such as gutka, naswar, khaini, and zarda, with lower levels in paan and paan masala. This supports the idea that users are repeatedly bathing the cells of their mouths in methylglyoxal every time they keep tobacco in contact with the cheek or gums.
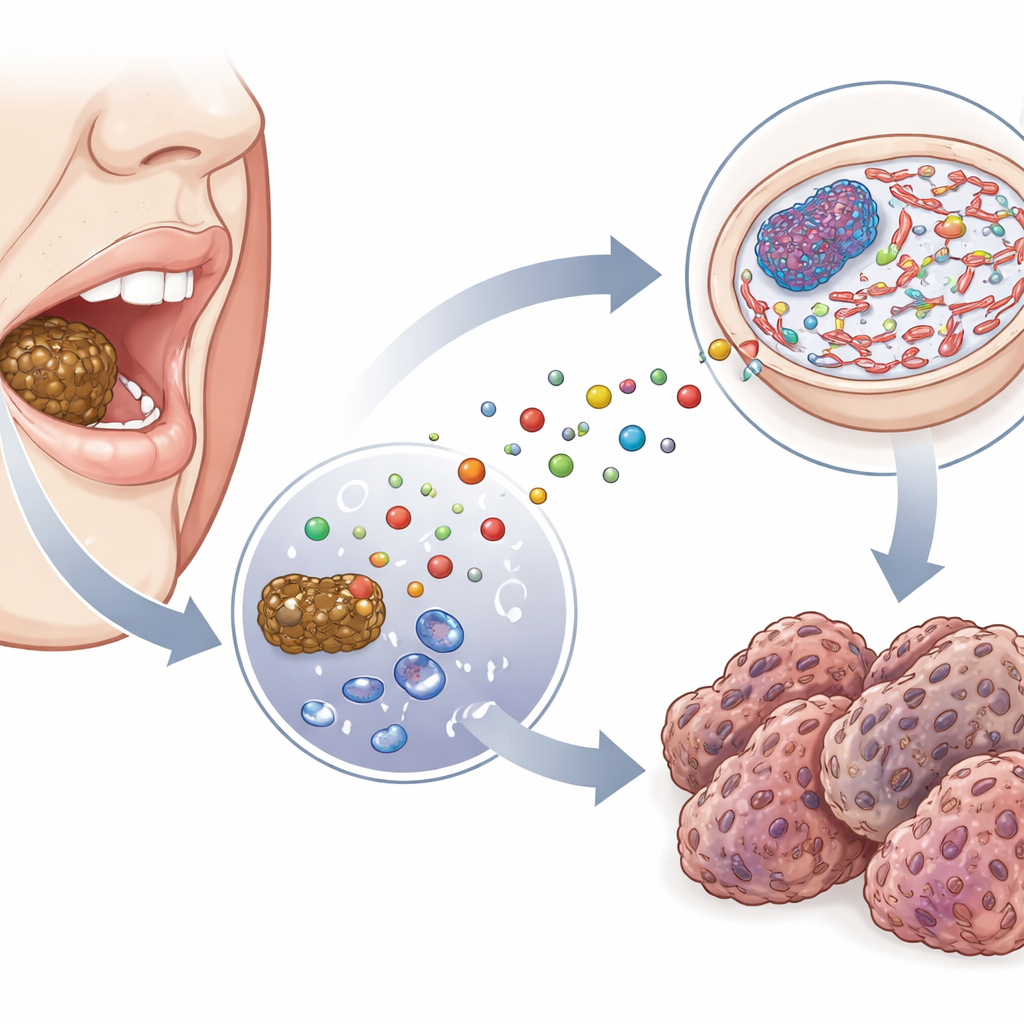
Tracking DNA scars inside mouth tumors
To see whether this exposure leaves a mark on DNA, the team examined tissue samples from patients with oral squamous cell carcinoma and compared them with normal tissue taken from the same mouths, a short distance away from the tumor. Using a fluorescent antibody that specifically recognizes CEdG, they visualized where this DNA lesion appears inside cells. Cancerous tissue showed many more brightly stained nuclei, higher overall signal, and greater variation from cell to cell than nearby healthy tissue. Importantly, the glow was concentrated in the nucleus, where the cell’s DNA is stored, rather than in the surrounding cytoplasm. These patterns indicate that CEdG is not only present but enriched in the genetic material of tumor cells from people who use smokeless tobacco.
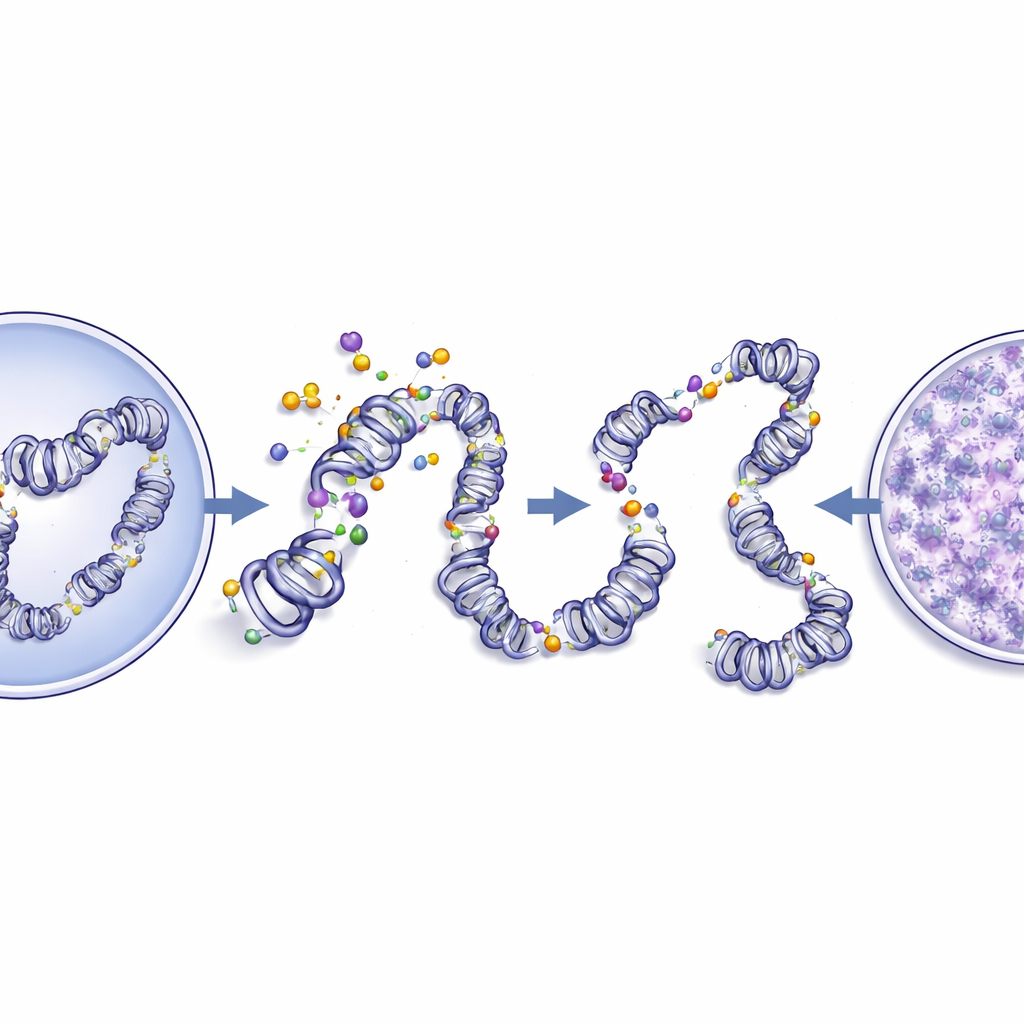
When DNA packing turns from orderly to chaotic
DNA in healthy cells is not just a loose string; it is folded into a carefully layered structure called chromatin. The way this material is packed affects which genes are turned on or off. The researchers used high-resolution images of standard stained slides to measure the size and shape of nuclei and to analyze the fine texture of chromatin. Compared with normal tissue, cancer cells had larger, more irregular nuclei and chromatin that looked more disorganized and “grainy.” Digital analysis showed higher entropy, contrast, and fractal dimension—features that signal a more chaotic, less compact arrangement—and reduced measures of order and smoothness. When these image-based measurements were compared directly with CEdG levels, stronger CEdG staining went hand in hand with more disturbed chromatin texture. This tight correlation suggests that DNA glycation and the architectural remodeling of chromatin are closely linked in oral tumors.
Connecting tobacco exposure, DNA damage, and tumor behavior
By combining chemical measurements in tobacco products, precise imaging of DNA lesions, and computational analysis of nuclear structure, the study outlines a chain of events: methylglyoxal-rich smokeless tobacco can generate CEdG in DNA; CEdG is abundant and unevenly distributed within the nuclei of oral cancer cells; and higher CEdG levels track with nuclei whose chromatin has lost its normal organization. Because chromatin structure helps control which genes are active, such disorganization may contribute to the unstable gene activity patterns that drive cancer growth and spread. The work also highlights CEdG as a distinct kind of marker—one that reflects direct damage to DNA from a known environmental exposure, rather than a downstream response molecule.
What this could mean for prevention and care
For lay readers, the key message is that smokeless tobacco does not merely irritate the mouth’s surface; it can leave lasting chemical scars on DNA that are tied to the internal architecture of our cells’ genetic material. While this study cannot yet prove that CEdG causes oral cancer, it shows that higher levels of this lesion accompany both heavy methylglyoxal exposure and profound disruption of chromatin in tumors. With further research in larger groups and more precise measurements, CEdG could become a tissue-based sign of tobacco-driven DNA damage and possibly a tool for tracking risk or treatment response in people who use smokeless tobacco.
Citation: Khan, G., Waraich, R.S., Khan, H. et al. N2-(1-carboxyethyl)-2′-deoxyguanosine as a genotoxic glycation marker associated with chromatin architectural alterations in oral squamous cell carcinoma. Sci Rep 16, 12715 (2026). https://doi.org/10.1038/s41598-026-45839-w
Keywords: oral cancer, smokeless tobacco, DNA damage, chromatin structure, methylglyoxal