Clear Sky Science · en
Integrated molecular and histological profiling of preeclampsia suggests angiogenic dysregulation associated with maternal and fetal outcomes
When Pregnancy Blood Pressure Becomes Dangerous
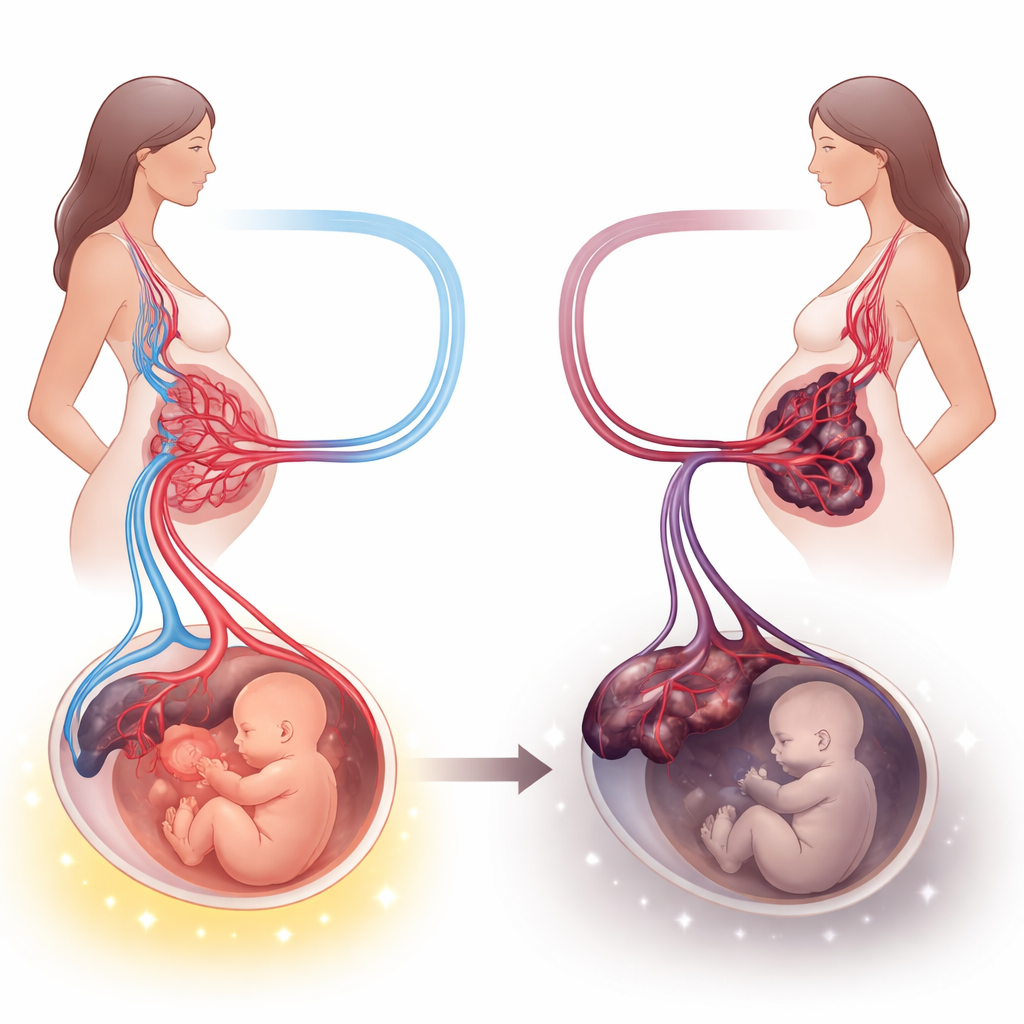
Preeclampsia is a serious form of high blood pressure that strikes during pregnancy and can endanger both mother and baby. Doctors have known for years that the placenta—the shared lifeline between the two—is central to this condition, but exactly what goes wrong inside this organ has been harder to pin down. This study takes a deep look at the placenta’s structure and chemistry in women with and without preeclampsia, aiming to connect microscopic changes with real-world outcomes like baby’s birth weight and timing of delivery.
Looking Closely at the Pregnancy Lifeline
The researchers examined placentas from 30 women with preeclampsia and 30 women with normal blood pressure. They did not just weigh and measure these organs; they also looked at thin tissue slices under the microscope and used special stains to highlight key molecules. These markers included substances that help blood vessels grow, maintain healthy vessel lining, control inflammation, and regulate cell death. By combining these laboratory findings with medical records on blood pressure, medications, delivery week, and newborn health, the team built an integrated picture of how preeclampsia reshapes the placenta.
Physical Signs of a Stressed Placenta
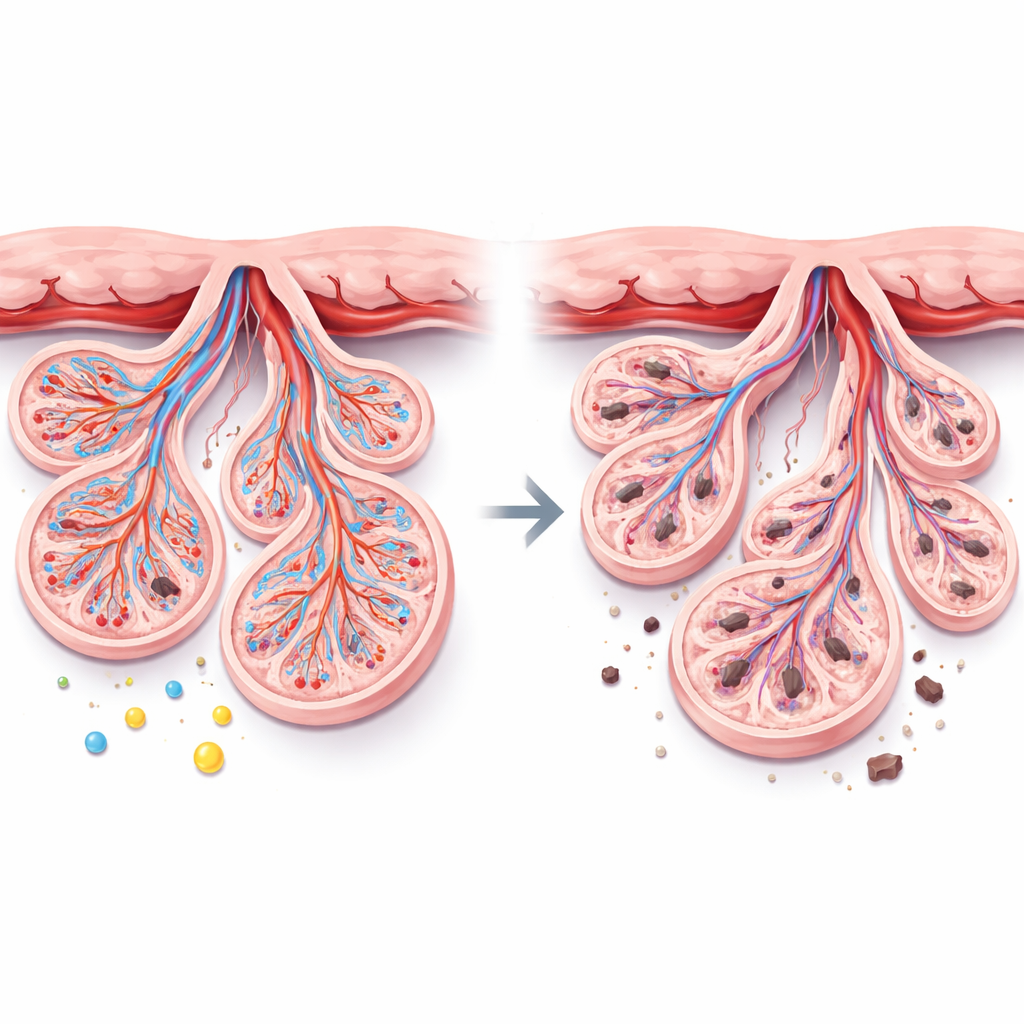
Compared with healthy pregnancies, placentas from preeclamptic mothers were smaller and lighter, and their babies tended to arrive earlier and weigh less. Under the microscope, the preeclampsia placentas showed hallmarks of chronic stress: more clumps of aging surface cells (called syncytial knots), thicker bands of clot-like material between the finger-like villi, underdeveloped tiny branches where exchange should happen, pockets of swelling, areas without blood vessels, and heavy deposits of calcium. Together, these features suggest that blood flow through the placenta is impaired and that the tissue is trying—and failing—to adapt to a low-oxygen, high-pressure environment.
Molecular Clues: Growth Signals, Protection, and Damage
On the molecular level, the study found that several protective and growth-promoting signals were dialed down in preeclampsia, while damaging signals were turned up. Levels of α-Klotho, a protein that helps guard cells against stress and supports healthy blood vessels, were markedly lower. So were markers that guide new vessel growth and function, such as VEGF, PlGF, and eNOS. In contrast, TNF-α, a powerful inflammatory molecule, was much higher. A test that detects broken DNA revealed a surge in cell death inside the placenta. Importantly, α-Klotho tended to move in step with the healthy growth markers and placental weight, and in the opposite direction of inflammation and cell death, hinting that it may be a central player in keeping the placenta balanced.
Linking Placental Changes to Mother and Baby
Using statistical models that accounted for age, smoking, and pregnancy length, the authors showed that preeclampsia itself was strongly linked to this cluster of placental changes: more inflammation and cell death, and less of the proteins that foster healthy vessels. These shifts were also tied to smaller placentas and lower birth weights, even after adjusting for how early the babies were born. Early-onset preeclampsia showed particularly marked loss of α-Klotho, with somewhat preserved vessel enzyme activity, suggesting the body may initially try to compensate for placental stress. Treatments like magnesium sulfate appeared to modestly support some vessel-growth signals, though the study was too small to draw firm conclusions.
What This Means for Future Pregnancies
In plain terms, this work supports the idea that preeclampsia is not just about high blood pressure; it is a disease of a struggling placenta whose blood vessels do not form or function properly. The organ becomes smaller, more scarred, and more prone to cell death, which in turn limits the baby’s growth and can prompt an early birth. By tying together the physical appearance of the placenta with chemical signals and clinical outcomes, the study highlights α-Klotho and related vessel-growth factors as promising targets for future tests or treatments. While the research cannot yet prove cause and effect, it points toward new ways to protect the shared lifeline between mother and baby.
Citation: Gelenli Dolanbay, E., Hocaoglu, M., Mert, T. et al. Integrated molecular and histological profiling of preeclampsia suggests angiogenic dysregulation associated with maternal and fetal outcomes. Sci Rep 16, 12874 (2026). https://doi.org/10.1038/s41598-026-45626-7
Keywords: preeclampsia, placenta, angiogenesis, pregnancy complications, birth weight