Clear Sky Science · en
Identification of key genes in the pathogenesis of hepatic ischemia-reperfusion injury based on bioinformatics and experimental verification
Why protecting the liver during surgery matters
When surgeons temporarily cut off blood flow to the liver during major operations or transplantation, restoring that flow can paradoxically injure the very organ they are trying to save. This phenomenon, called ischemia-reperfusion injury, can lead to liver dysfunction, complications, and longer hospital stays. The study summarized here uses modern gene-analysis tools and animal experiments to uncover which genes and immune responses are most critical in this process, and points to a potential drug strategy that could one day help shield the liver during surgery.
How the liver gets hurt when blood returns
During surgery, surgeons may briefly clamp blood vessels to the liver, depriving liver cells of oxygen and energy. In this “ischemia” phase, cells begin to struggle, their energy factories (mitochondria) falter, and chemical balance is disrupted. When blood flow is restored, fresh oxygen suddenly rushes in. Instead of being purely helpful, this surge reacts with built-up chemicals and generates large amounts of reactive molecules that can damage cell membranes, proteins, and DNA. At the same time, injured cells release distress signals that summon immune cells, including neutrophils and other white blood cells. This creates a powerful inflammatory wave that can cause more harm than the initial lack of blood, turning a short interruption into a serious injury.
Mapping gene activity across the injury timeline
To understand which genes drive this damage, the researchers used a well-established mouse model that closely mimics human liver surgery. Mice were grouped into four stages: healthy controls, after one hour of blocked blood flow with no reperfusion, after one hour of blockage plus six hours of reperfusion (the peak of injury), and after one hour of blockage plus 24 hours of reperfusion (a recovery phase). The team measured the activity of all genes in liver tissue at these stages. By comparing the peak-injury stage to the others, they identified 115 genes whose activity changed consistently during the damaging reperfusion period. These genes clustered in processes related to energy use, small-molecule metabolism, and blood and immune functions, hinting that disturbed metabolism and runaway inflammation lie at the heart of the problem.
Three standout genes and what they may be doing
From this large list, the scientists then used network analysis and several machine-learning methods to narrow down the most influential players. They converged on three key genes: Adh4, Akr1c14, and Cxcl1. Adh4 and Akr1c14, both normally active in liver metabolism, dipped to their lowest levels at the time of greatest injury, suggesting that the liver’s ability to handle fats, detoxify harmful by-products, and maintain steady energy supply is sharply weakened during reperfusion. In contrast, Cxcl1 spiked at the peak of injury. This gene encodes a powerful chemical signal that attracts neutrophils and other immune cells into tissues. Further analysis linked all three genes to pathways involving energy production in mitochondria and the handling of small molecules, while Cxcl1 was especially tied to pathways involved in building and processing RNA and proteins, consistent with a highly activated inflammatory state.
Immune cell traffic and signals that steer it
The study also examined how different immune cell types changed between the pre-reperfusion and peak-injury stages. Thirteen kinds of immune cells, including B cells, T cells, granulocytes, neutrophils, and mast cells, shifted markedly. Notably, Cxcl1 levels closely tracked the presence of granulocytes and neutrophils—cells known to release enzymes and reactive molecules that worsen tissue injury. The authors then built a regulatory map showing how small regulatory RNAs, long non-coding RNAs, and transcription factors may control these key genes. For Cxcl1, they identified both gene-switching proteins and small RNAs that together may ramp up its production during reperfusion, further fueling immune cell recruitment.
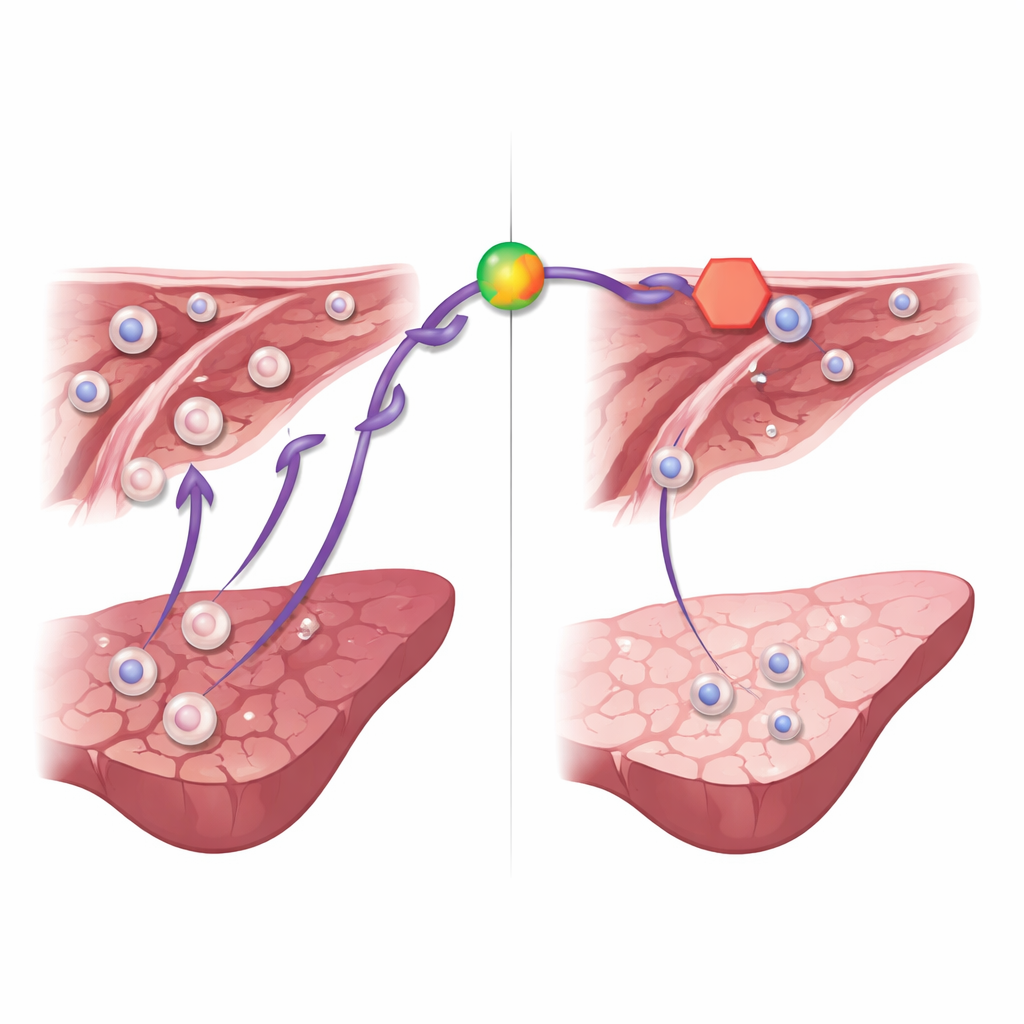
A drug hint: blocking a key signal softens the blow
Beyond computer analysis, the researchers tested whether dampening the Cxcl1 pathway could protect the liver. They used ladarixin, an experimental drug that blocks the receptors sensing Cxcl1-like signals on immune cells. Mice given ladarixin before and during reperfusion had lower levels of liver enzymes in their blood, indicating less cell damage, and showed reduced inflammatory molecules and neutrophil buildup in liver tissue. Under the microscope, their livers displayed smaller areas of dead tissue and healthier structure. These findings suggest that while many genes and cells participate in ischemia-reperfusion injury, the Cxcl1 signaling route is a particularly promising target for therapy.
What this means for future liver care
In simple terms, this work shows that when blood flow is restored to the liver after a brief shutdown, a combination of disrupted energy metabolism and excessive immune cell invasion drives much of the damage. Three genes—Adh4, Akr1c14, and especially Cxcl1—sit at key points in this process. By carefully charting when these genes turn up or down and how immune cells respond, the study offers a roadmap for future treatments. While the findings come mainly from mice and need confirmation in patients, they point toward strategies that either stabilize liver metabolism or selectively block harmful immune signals, with drugs like ladarixin offering an early glimpse of how we might better protect the liver during complex surgeries and transplants.
Citation: Zhang, T., Jiang, Z., Zhao, Q. et al. Identification of key genes in the pathogenesis of hepatic ischemia-reperfusion injury based on bioinformatics and experimental verification. Sci Rep 16, 14535 (2026). https://doi.org/10.1038/s41598-026-45495-0
Keywords: liver surgery, ischemia reperfusion, inflammation, gene regulation, neutrophils