Clear Sky Science · en
A lanmodulin homologous protein, from Mesorhizobium qingshengii J19, involved in yttrium immobilization
Why this matters for everyday tech
Rare earth elements hide inside many of the devices and green technologies we rely on, from phones and wind turbines to electric cars and medical scanners. Mining and refining these metals is costly and polluting, so scientists are searching for gentler ways to pull them from waste streams and contaminated water. This study explores how a protein from a soil bacterium can latch onto one of these metals, yttrium, and help trap and recover it in a cleaner way.
Metals that power modern life
Rare earth elements form a family of metals that behave in similar ways in water, which makes them hard to separate from one another and from other ions. Yttrium and its cousins are essential for strong magnets, batteries, screens and lasers. In recent years, researchers discovered that some bacteria actually need rare earths to run key enzymes that help them use simple carbon compounds as food. That means nature already contains molecules that can pick out these metals with surprising precision, offering clues for new recycling tools.
A bacterial protein with a special grip
The team focused on a bacterium called Mesorhizobium qingshengii J19, previously shown to tolerate and immobilize high levels of yttrium. By scanning its genome, they found a gene for a small metal-binding protein related to lanmodulin, a well known rare earth catcher from another bacterium. This new protein, built from so-called EF-hand motifs that normally bind calcium, differs at key positions in its sequence, replacing a commonly found building block (proline) with another (threonine). The researchers suspected that this change might alter which metals it prefers and how tightly it grips them. 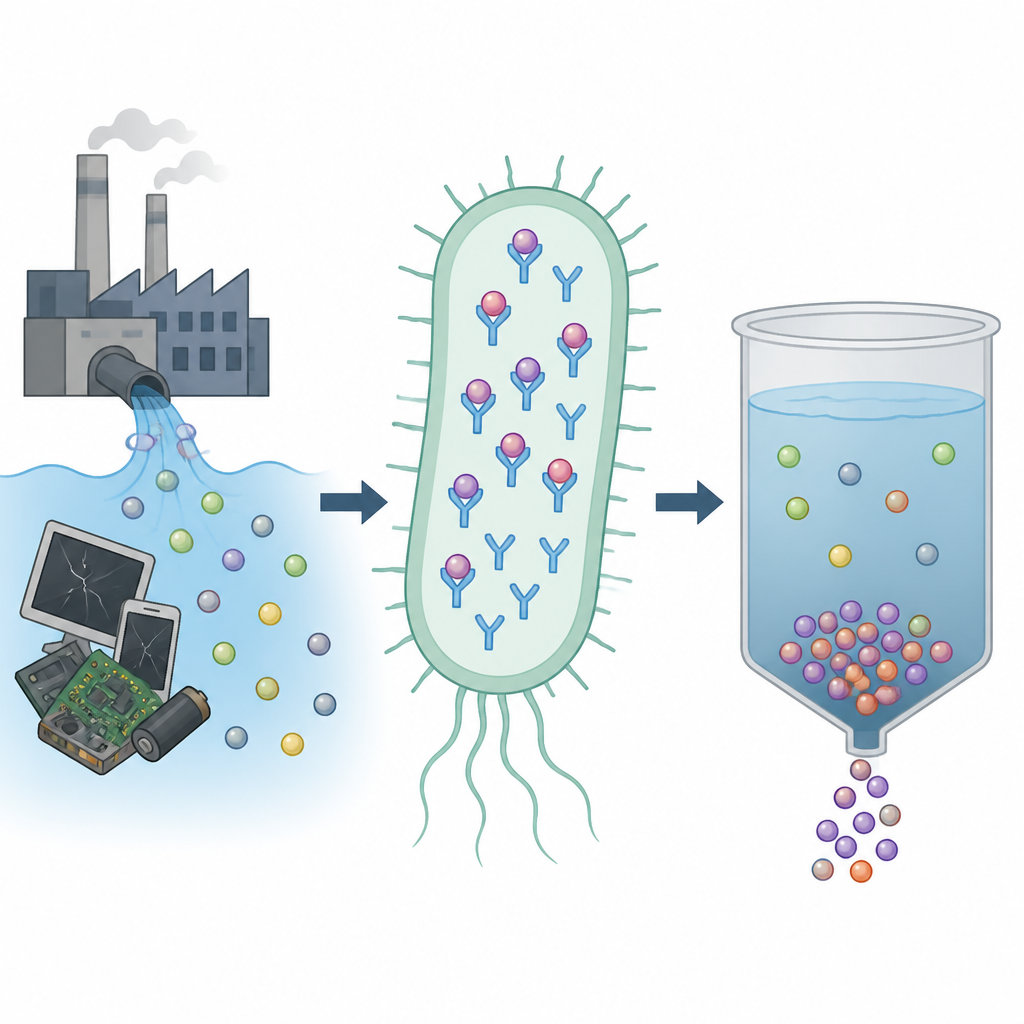
Turning common lab bacteria into metal sponges
To test the protein, the scientists inserted its gene into Escherichia coli, a standard workhorse bacterium used in labs, and coaxed the cells to make large amounts of the protein in the space between their inner and outer membranes. Compared to normal E. coli, the engineered cells accumulated much more yttrium and also more neodymium when both metals were present together, while they did not gain extra ability to accumulate scandium. This pattern hints that the protein has a sweet spot for metal ions of certain sizes, favoring yttrium and neodymium over both smaller and slightly larger relatives.
Measuring the protein’s taste for metals
After purifying the protein, the team ran a series of binding tests in which they mixed it with known amounts of yttrium and then separated protein-bound metal from free metal. They found that at moderate concentrations, the protein bound large amounts of yttrium, especially at slightly alkaline pH and room temperature. Under the best conditions, its apparent capacity to hold yttrium was far higher than values previously reported for lanmodulin binding another rare earth, lanthanum, although the authors stress that methods and metals differ. Additional tests showed that the protein can also bind neodymium, but when yttrium and neodymium were offered together, yttrium won out, suggesting a modest preference for that metal. 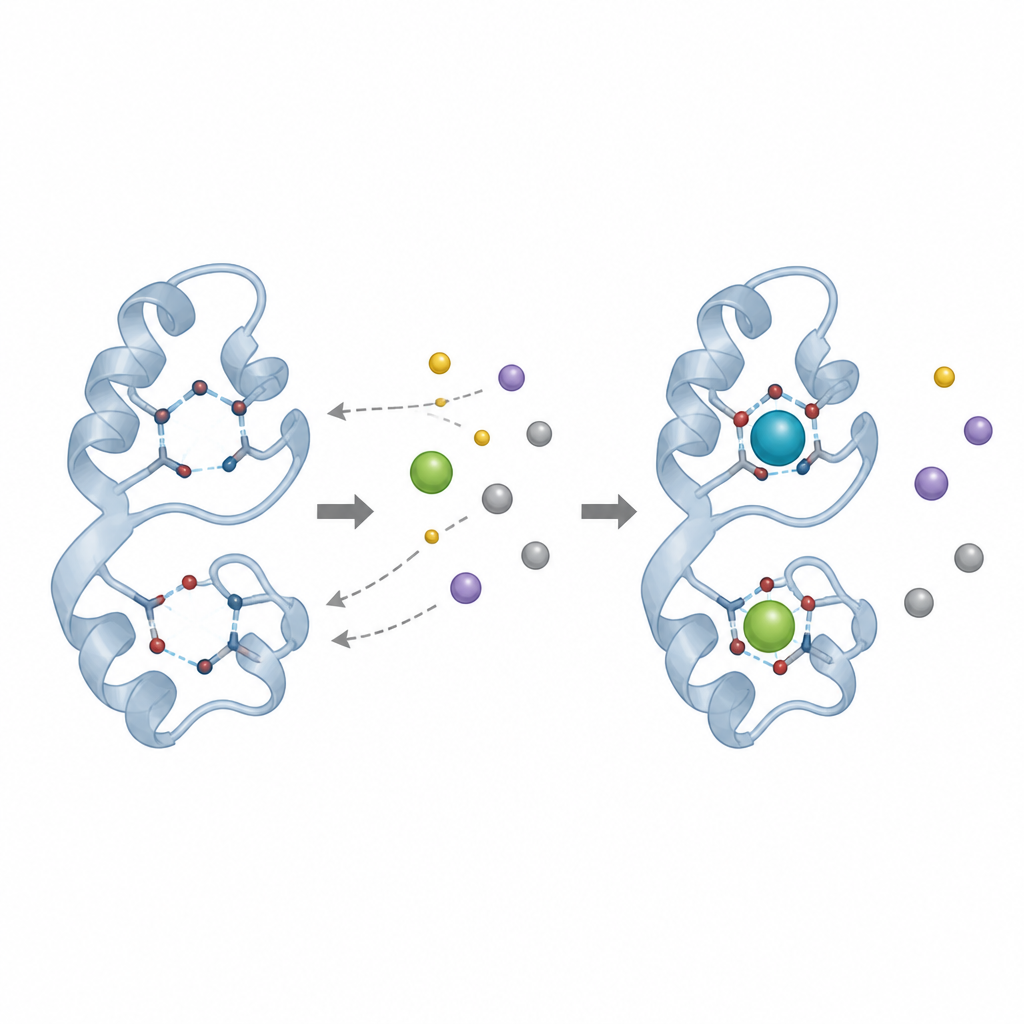
From lab assays to cleaner recovery methods
Because M. qingshengii J19 cells can trap yttrium on their surfaces and even form yttrium-rich mineral particles, and because its EF-hand protein binds yttrium so well in solution, the authors see several possible uses. Engineered E. coli cells carrying the protein could sit behind selective membranes and act like living filters that grab rare earths from dilute, mixed-metal streams such as mining effluents or electronic waste leachates. Alternatively, the purified protein could be fixed onto solid supports or membranes and reused in controlled cycles of metal capture and release, using mild changes in acidity or salt levels to strip the metals off for recycling.
What this means in simple terms
In essence, the study shows that a protein borrowed from a soil microbe can work as a tiny claw that prefers to hold onto yttrium and, to a lesser extent, neodymium. By understanding when this claw works best and how selective it is, researchers move closer to designing bio-based filters that can clean up and recover valuable rare earth metals from watery wastes. Such approaches could one day complement or partially replace harsh chemical processes, helping to secure critical materials for modern technology while lowering the environmental cost of getting them.
Citation: Coimbra, C., Morais, P.V. & Branco, R. A lanmodulin homologous protein, from Mesorhizobium qingshengii J19, involved in yttrium immobilization. Sci Rep 16, 15015 (2026). https://doi.org/10.1038/s41598-026-45294-7
Keywords: rare earth elements, yttrium, lanmodulin homolog, bioremediation, metal-binding protein