Clear Sky Science · en
Predictive simulations of postural control: exploring the role of signal noise and neural delays in Parkinson’s disease
Why Balance Problems in Parkinson’s Matter
Many people with Parkinson’s disease worry most about something very down‑to‑earth: staying upright. As the disease progresses, even quiet standing can become unsteady, raising the risk of falls and injury. Yet doctors can only see the outward wobbling, not the inner workings of the nervous system that cause it. This study uses computer simulations of the body and brain working together to explore two hidden suspects—noisy movement signals and slowed nerve processing—and how they might make balance worse in Parkinson’s disease.
Looking Inside the Balance System
To stay balanced, our bodies constantly keep the body’s center of mass above the feet. Sensors in the skin, muscles, inner ear, and eyes send information to the brain, which then sends commands to the leg and trunk muscles to make tiny corrections. This cycle runs many times per second and is always influenced by background "chatter" from breathing, heartbeat, and random fluctuations in nerve signals. In Parkinson’s disease, changes in deep brain regions called the basal ganglia are known to slow movement and alter rhythmic brain activity, but how exactly this affects the standing balance loop is hard to measure directly in real people.
Building a Virtual Person
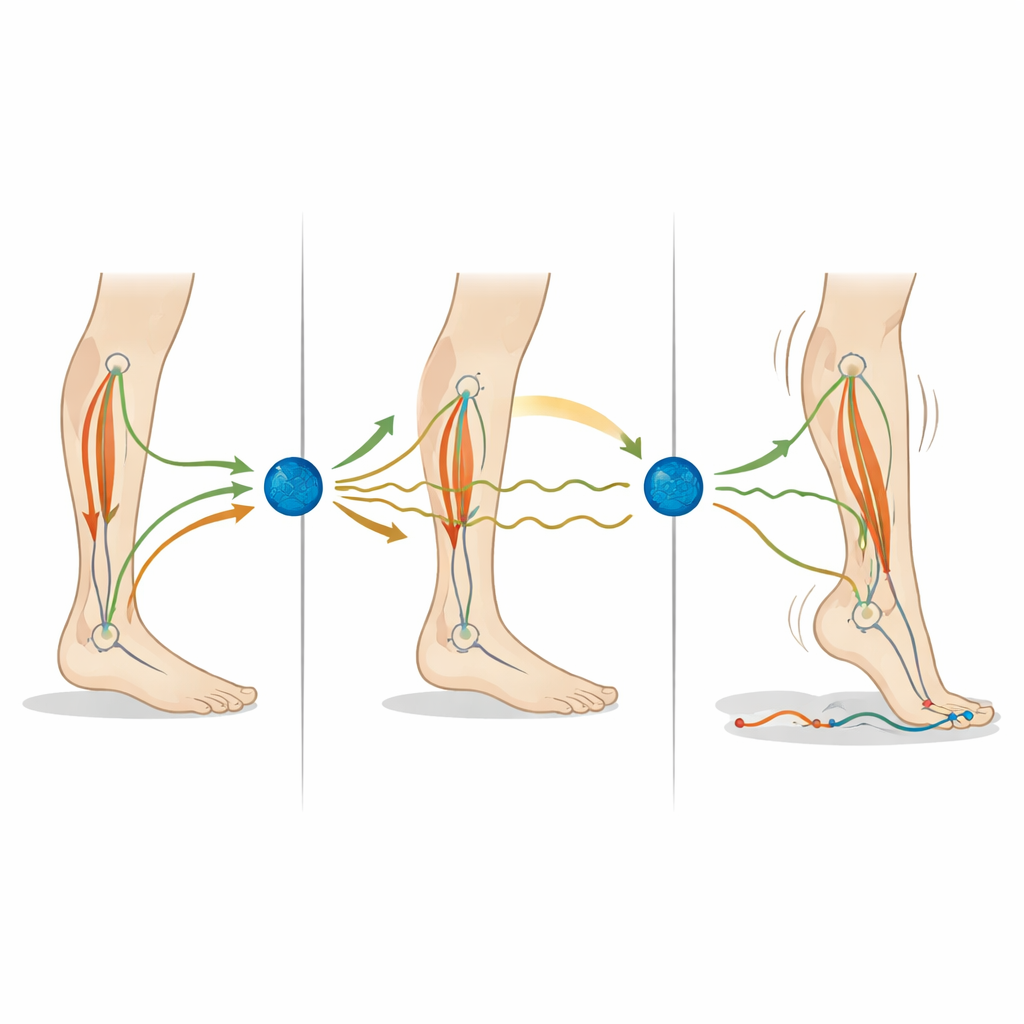
The researchers built on an existing digital model that links a simplified human skeleton and leg muscles to a control system representing the nervous system. In this virtual person, sensory signals report the body’s position, a controller compares this to an ideal upright stance, and then sends commands to muscles, which generate forces and move the joints. The model also includes realistic delays as signals travel along nerves and through brain pathways, plus random internal noise added to the outgoing motor commands. By adjusting the amount of noise or the length of delay, the team could see how the simulated body swayed forward and backward during 75 seconds of quiet standing, and then compare these results with motion‑capture data from 31 people with Parkinson’s and 31 healthy peers.
How Noisy Signals Change Sway
In the first set of simulations, the team increased two types of noise in the outgoing muscle commands: a constant background noise and a signal‑dependent noise that grew with the strength of the command. As either type of noise increased, the virtual person swayed more. The path traced by the pressure under the feet became longer and wider, and the joints in the pelvis, hips, knees, and ankles moved through larger angles. Muscle activity also rose, reflecting the extra effort needed to control the wobblier body. These patterns closely matched the differences seen between the healthy volunteers and those with Parkinson’s, especially for the constant background noise, suggesting that less precise motor signals could be a key contributor to real‑world instability.
What Slower Nerves Do to Standing
Next, the researchers lengthened the total time it took for signals to travel through the balance loop, mimicking slower neural processing. With added delay, most measures of sway again increased: the simulated center of pressure wandered further, and joint movements grew larger. These changes appeared whether the model started with low or high noise, though some sway features, such as the exact front‑back position of the pressure under the feet and the average sway frequency, changed little—mirroring the experimental data. The results suggest that both noisier signals and longer processing times can nudge the nervous system into a less stable way of standing that resembles Parkinson’s disease.
What This Means for People with Parkinson’s
For non‑experts, the key message is that balance problems in Parkinson’s may arise not just from weak muscles or stiff joints, but from hidden changes in how cleanly and how quickly the brain and nerves communicate. By tuning these internal factors in a virtual patient and matching the outcomes to real motion data, the study shows that increased noise and delayed processing can together recreate Parkinson‑like sway. In the future, similar models could help doctors estimate a person’s internal control settings from simple balance tests, track how these change over time or with treatment, and ultimately design more targeted therapies and rehabilitation programs to keep people steadier on their feet.
Citation: Shanbhag, J., Wechsler, I., Fleischmann, S. et al. Predictive simulations of postural control: exploring the role of signal noise and neural delays in Parkinson’s disease. Sci Rep 16, 9849 (2026). https://doi.org/10.1038/s41598-026-45161-5
Keywords: Parkinson’s disease, postural control, balance, neuromusculoskeletal simulation, falls