Clear Sky Science · en
Preparation of novel karaya gum derived covalent immobilizers via polyethylene-imine and glutaraldehyde processing
Turning Plant Gum into a Smart Enzyme Helper
Many people cannot digest the milk sugar lactose, and food makers are searching for gentler, greener ways to remove it from dairy streams like whey. This study shows how a natural tree gum, already used in foods and medicines, can be transformed into a sturdy "parking spot" for enzymes that cut lactose. By engineering this plant material so that enzymes stick tightly to it, the researchers created tiny reusable tools that keep working for weeks, even in the presence of heat, metals, and solvents that usually weaken proteins.
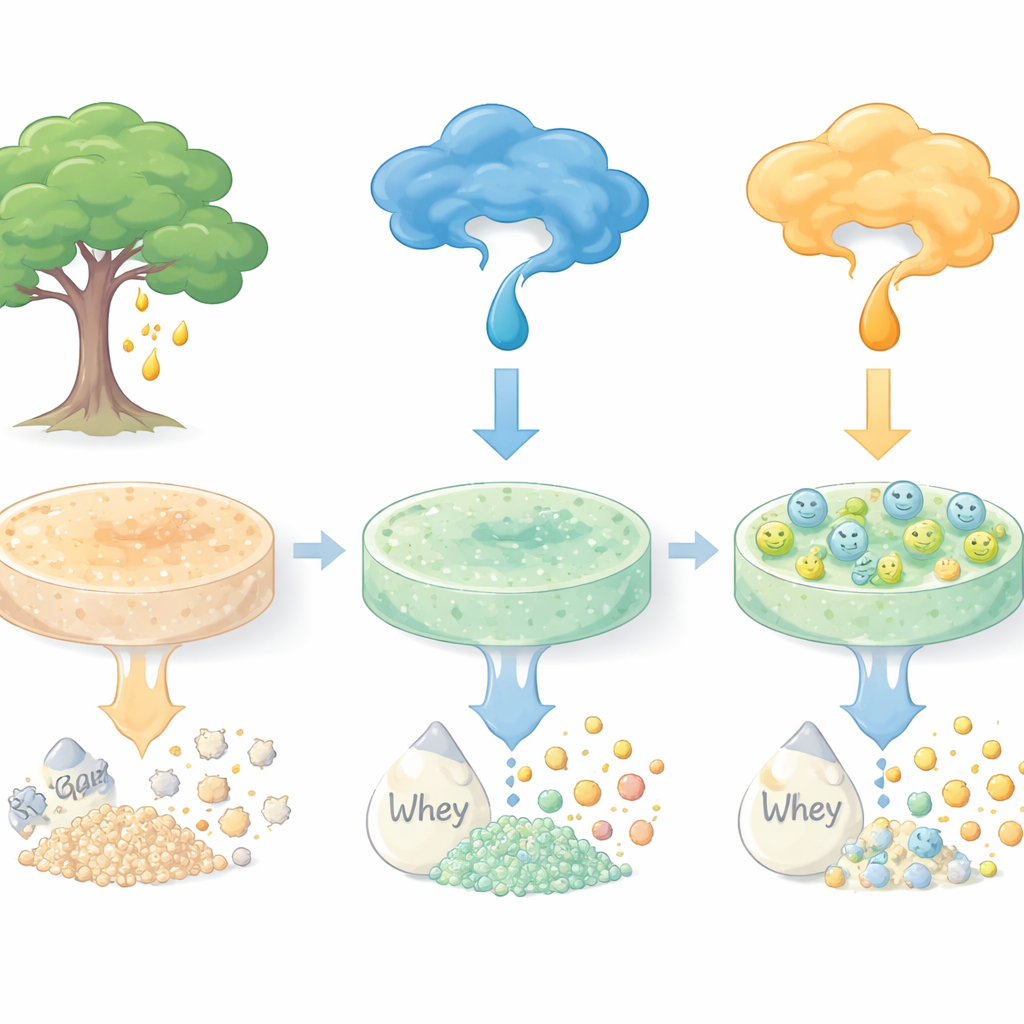
A Common Food Additive with New Possibilities
The work centers on karaya gum, a biodegradable polysaccharide obtained from the sap of Sterculia trees. Karaya gum is inexpensive, absorbs water well, and is already used as a thickener in foods, wound dressings, and drug delivery systems. However, it is usually soft and difficult to handle on its own. To make it into a solid yet gentle support for enzymes, the researchers blended karaya gum with a small amount of agar, another familiar gelling agent from seaweed. When heated and then cooled, this mixture formed firm discs that could be lifted, washed, and processed without crumbling.
Building a Sticky but Stable Enzyme Platform
Simply placing enzymes on the gum–agar discs would not be enough: they could wash away or lose activity. The team therefore used a two-step chemical treatment to convert the discs into strong "covalent immobilizers". First, they soaked the discs in a solution of polyethylene-imine, a positively charged, branched polymer that clings to the negatively charged sugars in the gum and agar, forming a dense outer coating rich in amino groups. Next, they treated this coated material with glutaraldehyde, a small linker molecule that reacts with those amino groups to create numerous anchoring points. Later, when the enzyme is added, it attaches to these points through multiple permanent bonds, greatly reducing leakage and motion that can lead to damage.
Tuning the Recipe for Maximum Enzyme Power
To find the best conditions for making this immobilizer, the researchers systematically varied the acidity (pH) and strength of the polyethylene-imine solution and the amount of glutaraldehyde. Using a statistical design approach, they discovered that a moderately alkaline polyethylene-imine solution and relatively high concentrations of both agents gave the highest activity of the attached enzyme. Microscopy showed that the treatment smoothed and reinforced the surface of the discs, while infrared spectroscopy confirmed that new chemical linkages had formed. Under these optimized conditions, the material could bind substantial amounts of the enzyme β-galactosidase, which splits lactose into the simpler sugars glucose and galactose, with immobilization efficiencies approaching 70%.
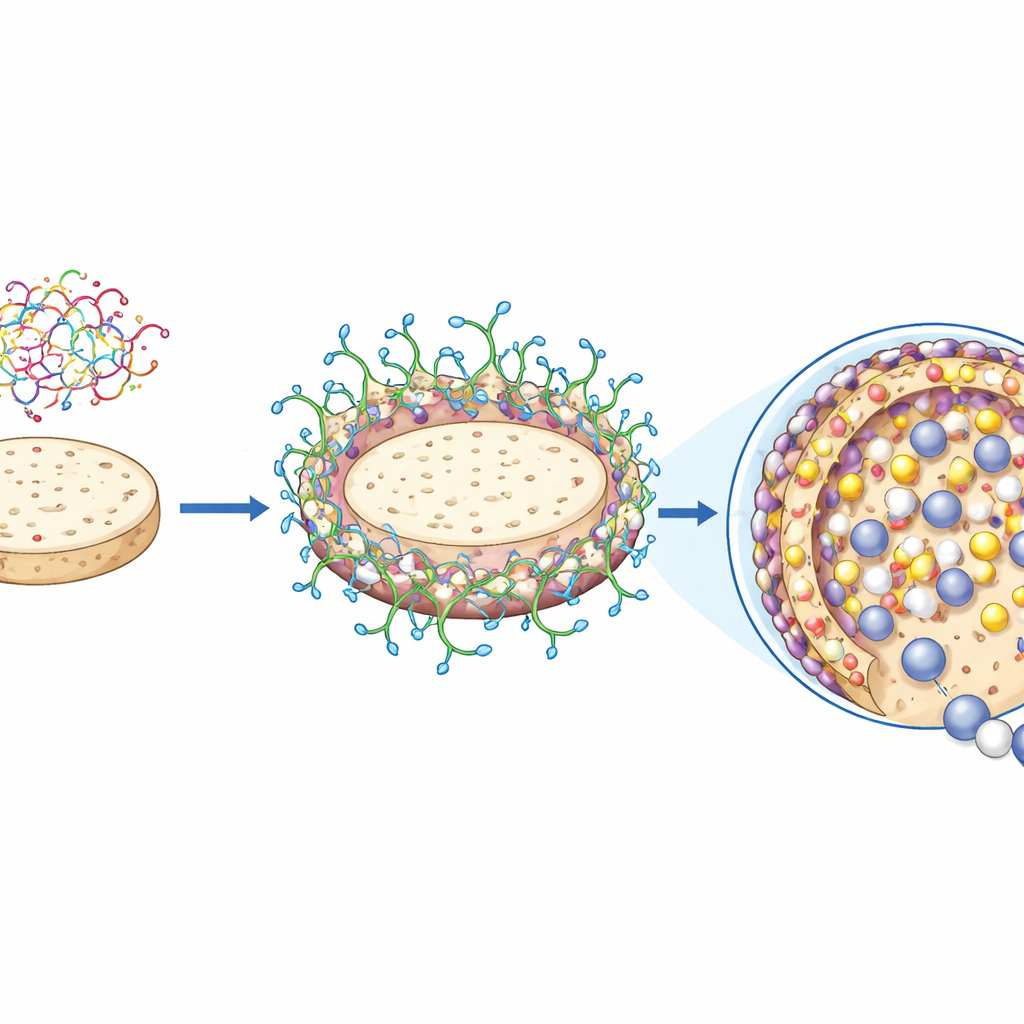
Enzymes That Resist Heat, Metals, and Solvents
Once attached to the modified gum–agar discs, β-galactosidase behaved more like a rugged industrial tool than a fragile biological molecule. The immobilized enzyme tolerated higher temperatures than the free enzyme and lost activity more slowly when held at elevated heat, as shown by longer half-lives and greater resistance to thermal breakdown. Its useful pH range became broader and shifted slightly toward more acidic conditions. Importantly, the immobilized enzyme withstood heavy metal ions such as mercury, aluminum, and iron—contaminants that can appear in milk and whey—without significant loss of activity, while the free enzyme was strongly inhibited. It also handled contact with several organic solvents much better, in part because the thick polymer coating and the hydrated gel helped shield it from harsh surroundings.
From Laboratory Discs to Cleaner Dairy Streams
For any immobilized enzyme system, long-term reuse and storage are critical. The engineered karaya gum–agar discs retained over 90% of their initial activity after nine weeks in the refrigerator and still showed about 95% activity after 23 repeated use cycles, outperforming several previously reported supports. When applied to real whey permeate, a byproduct of cheesemaking, the immobilized β-galactosidase broke down nearly 80% of the lactose in a single day, clearly surpassing the free enzyme under the same conditions and remaining effective over six consecutive daily runs. To a non-specialist, the key message is that a simple, safe plant gum can be upgraded into a tough, reusable enzyme carrier that turns waste dairy streams into more digestible and potentially more valuable products, offering a promising step toward more sustainable food processing.
Citation: Wahba, M.I. Preparation of novel karaya gum derived covalent immobilizers via polyethylene-imine and glutaraldehyde processing. Sci Rep 16, 12069 (2026). https://doi.org/10.1038/s41598-026-45030-1
Keywords: karaya gum, enzyme immobilization, lactose-free dairy, whey valorization, biopolymer hydrogels