Clear Sky Science · en
Albumin-on-a-chip: binding profiling of circulating human albumin via selective immunocapture and real-time SPR analysis
A Tiny Chip With Big Clues About Our Blood
When doctors prescribe a drug, they rely on how it usually behaves in the body. But in reality, each person’s blood chemistry is slightly different, especially when illnesses like diabetes or liver disease are present. This study introduces a small laboratory-on-a-chip that can test, in real time, how an individual’s main blood protein, albumin, actually binds drugs in their own blood, potentially opening the door to more tailored and safer treatments.
The Workhorse Protein in Our Blood
Human serum albumin is the most abundant protein in our bloodstream. It helps keep fluid in our vessels, mops up damaging oxidants, and shuttles a wide array of natural substances and medicines. Albumin is not a single, uniform molecule: in every person it exists as a mixture of slightly different versions, shaped by everyday metabolism, aging, inflammation, and disease. Chemical changes such as oxidation and sugar attachment can subtly warp its structure. These changes may alter how tightly albumin holds on to drugs, which in turn can influence how much active drug is available to act—and how long it stays in the body.
Why Traditional Tests Fall Short
Most previous studies of albumin’s binding behavior have relied on purified, commercially produced protein that is altered in simple, artificial ways. Such models do not capture the true complexity of albumin found in real patients, where many modifications coexist and shift as disease progresses or treatment changes. Other approaches that use whole plasma are confounded by many different proteins that also bind drugs, while more precise methods require slow and expensive purification steps. As a result, scientists have lacked a practical way to measure, for each patient, how their own circulating albumin actually interacts with important medicines.
Building Albumin-on-a-Chip
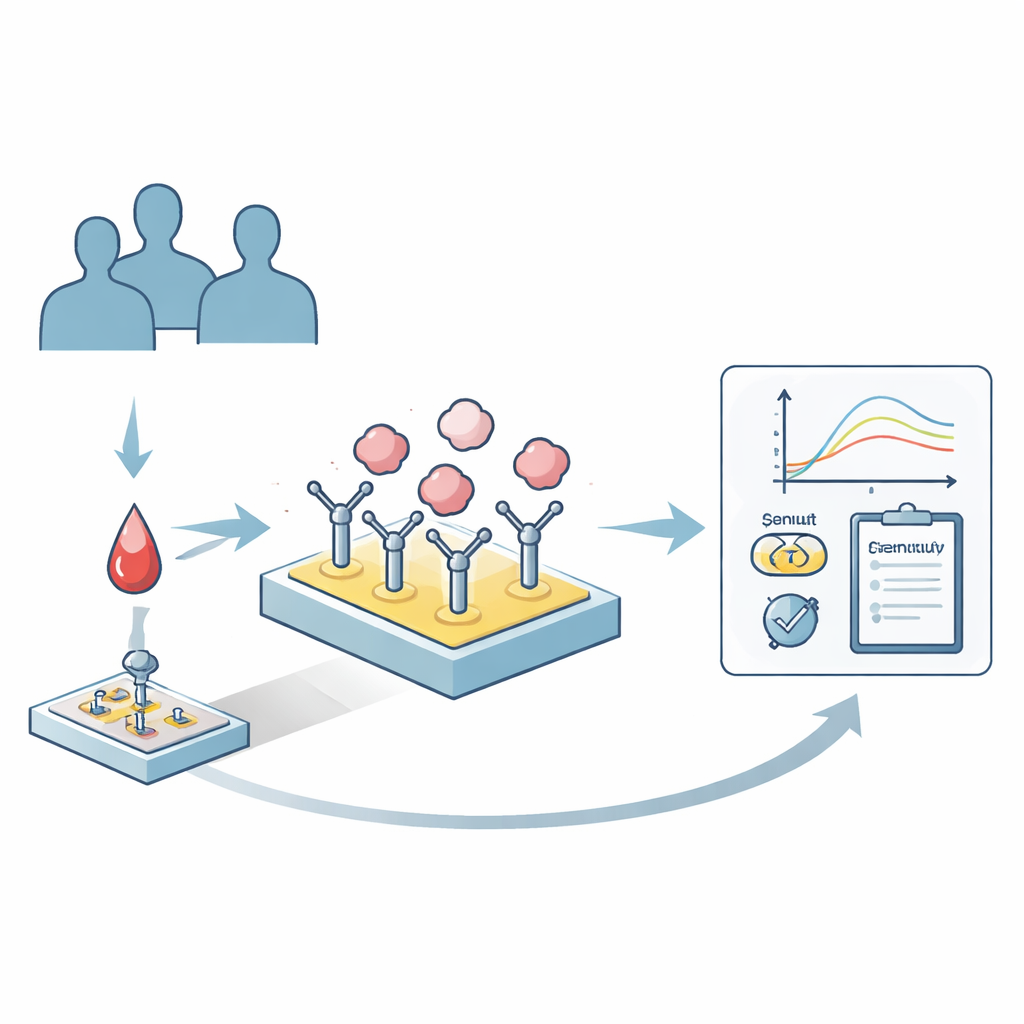
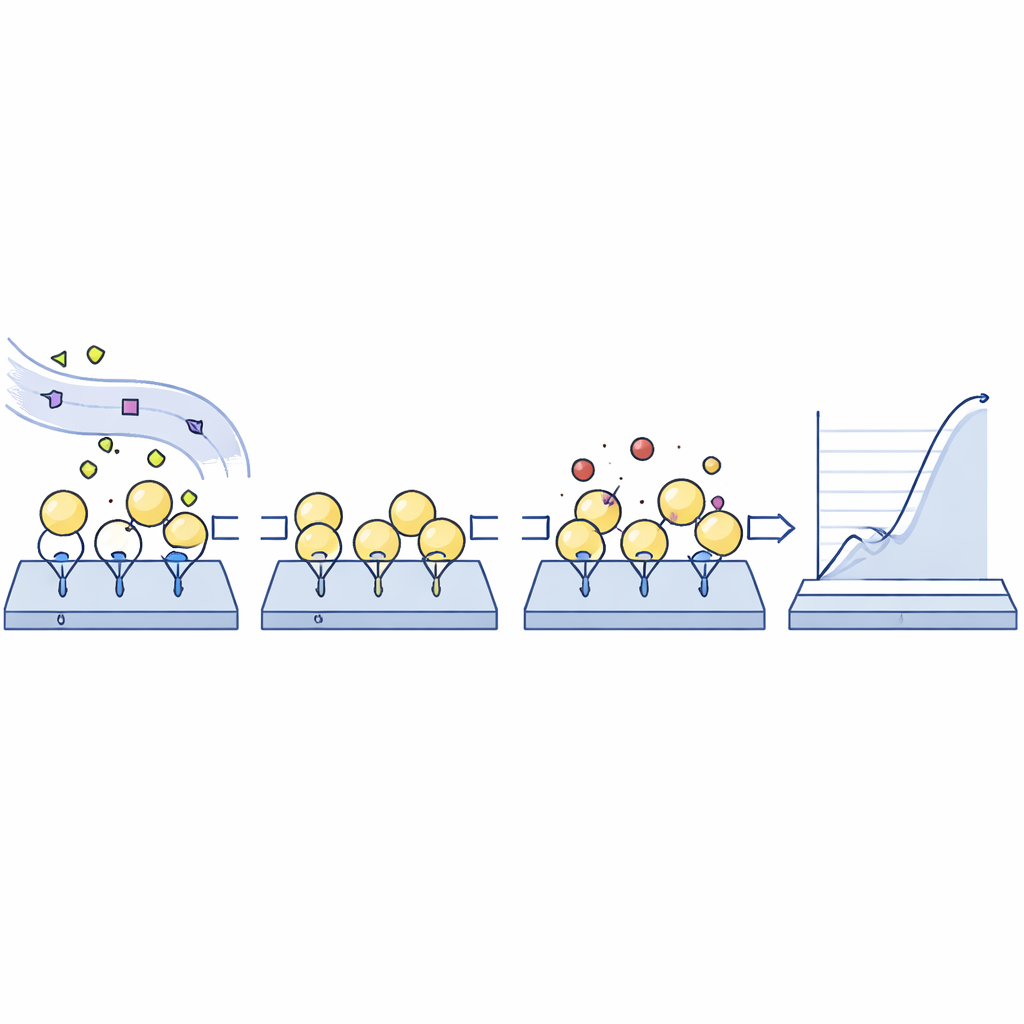
The researchers developed a sensing platform based on surface plasmon resonance, a technique that detects binding events on a chip surface in real time. First, they coated a standard sensor chip with antibodies that specifically recognize human albumin. When a tiny amount of patient plasma flows over the chip, only albumin is selectively “caught” on the surface, with all its natural variants preserved and other plasma proteins largely excluded. After this capture step, the chip is exposed to candidate drugs or probe molecules, and the system records how strongly and how fast these molecules bind to the immobilized albumin mixture. A key feature is that the albumin can then be washed away without damaging the antibody layer, so the same chip can be reused hundreds of times, dramatically reducing cost.
Putting the Device to the Test in Real Patients
To confirm that this immunocapture really reflects what is happening in the bloodstream, the team used mass spectrometry to compare albumin variants before and after capture. They found that the chip retrieved native and modified albumin forms in the same proportions as in the original plasma, while leaving other proteins behind. They then applied the system to two small patient groups where albumin damage is known or suspected: people with advanced liver cirrhosis complicated by acute-on-chronic liver failure, and people with type 2 diabetes, some of whom had serious kidney impairment. In the liver group, the chip revealed that despite extensive structural damage to albumin, the major drug-binding regions remained mostly functional, though one key site showed slightly altered behavior. Notably, binding of the antibiotic teicoplanin was significantly weakened in these patients, which could affect how well the drug works. In contrast, in the diabetic kidney group, where albumin was only moderately altered, teicoplanin binding appeared largely unchanged.
A Step Toward More Personalized Drug Dosing
Overall, the study shows that a reusable, chip-based sensor can profile how a particular patient’s albumin binds drugs under realistic conditions, using only small plasma samples and without elaborate preparation. For lay readers, the key message is that the same drug dose may not behave identically in everyone, especially in serious liver or kidney disease, and that technologies like albumin-on-a-chip could help doctors measure these differences directly. In the long run, this kind of personalized binding profile might support safer dosing of critical medicines and provide a new window into how chronic illnesses reshape one of the bloodstream’s most important proteins.
Citation: Nugnes, M., Baldassarre, M., Caraceni, P. et al. Albumin-on-a-chip: binding profiling of circulating human albumin via selective immunocapture and real-time SPR analysis. Sci Rep 16, 10920 (2026). https://doi.org/10.1038/s41598-026-44934-2
Keywords: human serum albumin, drug binding, liver disease, diabetes complications, biosensor chip