Clear Sky Science · en
Progressive cognitive impairment and ventricular tachycardia in a boy with biallelic POLG variants and a de novo RYR2 variation
When Two Genetic Glitches Collide
Most of us think of a disease as having a single cause, but doctors are discovering that some patients are ill because of more than one hidden flaw in their DNA. This article tells the story of a boy whose worsening learning problems and sudden, fatal heart rhythm were ultimately traced to two separate genetic defects—one affecting the tiny power stations inside cells, and the other disturbing the electrical rhythm of the heart. His case shows how modern genetic tools and clever lab models, including yeast cells, can untangle such medical mysteries and improve diagnosis for future patients.
A Puzzling Story of Brain and Heart
The boy in this report developed normally at first, aside from delayed speech. By age five his thinking skills were still in the average range, but over the next few years his school performance declined sharply, and by age nine he met criteria for intellectual disability. Brain scans and early heart tests were unremarkable, and standard genetic studies did not reveal a cause. Then, at twelve, he began to have fainting spells linked to abnormal fast rhythms in the upper chambers of his heart. Despite medication, his heart function worsened, and he died suddenly of cardiac arrest at age thirteen. The combination of progressive cognitive problems and dangerous heart rhythm disturbances prompted a deeper search in his DNA.
Finding Two Separate Genetic Culprits
Using next-generation sequencing, which can scan hundreds of genes at once, the team first found three changes in a gene called POLG. This gene encodes a key part of the enzyme that copies mitochondrial DNA—the small ring of genetic material inside the cell’s energy factories. Two of the changes formed a known harmful pair on the father’s chromosome, previously linked to mitochondrial disease. The third change, called W113R, came from the mother and had never been seen before, making its medical importance unclear. Later, a more focused re-analysis of the boy’s exome data uncovered an additional change in a different gene, RYR2, which helps control calcium flow in heart muscle cells. This RYR2 variant, Y4725C, had been described in other patients with life-threatening fast heart rhythms and was not present in either parent, meaning it arose anew in the boy.
Using Yeast to Test a New Mutation
To decide whether the unknown POLG variant truly damaged the enzyme, the researchers turned to an unexpected ally: baker’s yeast. Yeast cells have their own version of the same mitochondrial DNA polymerase, called Mip1. Because the affected amino acid is conserved between species, the team copied the human W113R change into the yeast gene at the matching position. They then compared yeast carrying normal Mip1 with yeast carrying the altered version. At normal temperature, the mutant yeast could grow by breathing, but it showed more cells that had lost or damaged their mitochondrial DNA. Under heat stress, these problems became dramatic: almost all cells with the mutant enzyme were unable to respire, and their mitochondrial DNA content dropped sharply. The mutant enzyme also made slightly more copying errors, even though the protein level itself was not reduced. Together, these results showed that the W113R change impairs mitochondrial DNA maintenance, especially under stress.
Putting the Pieces Together
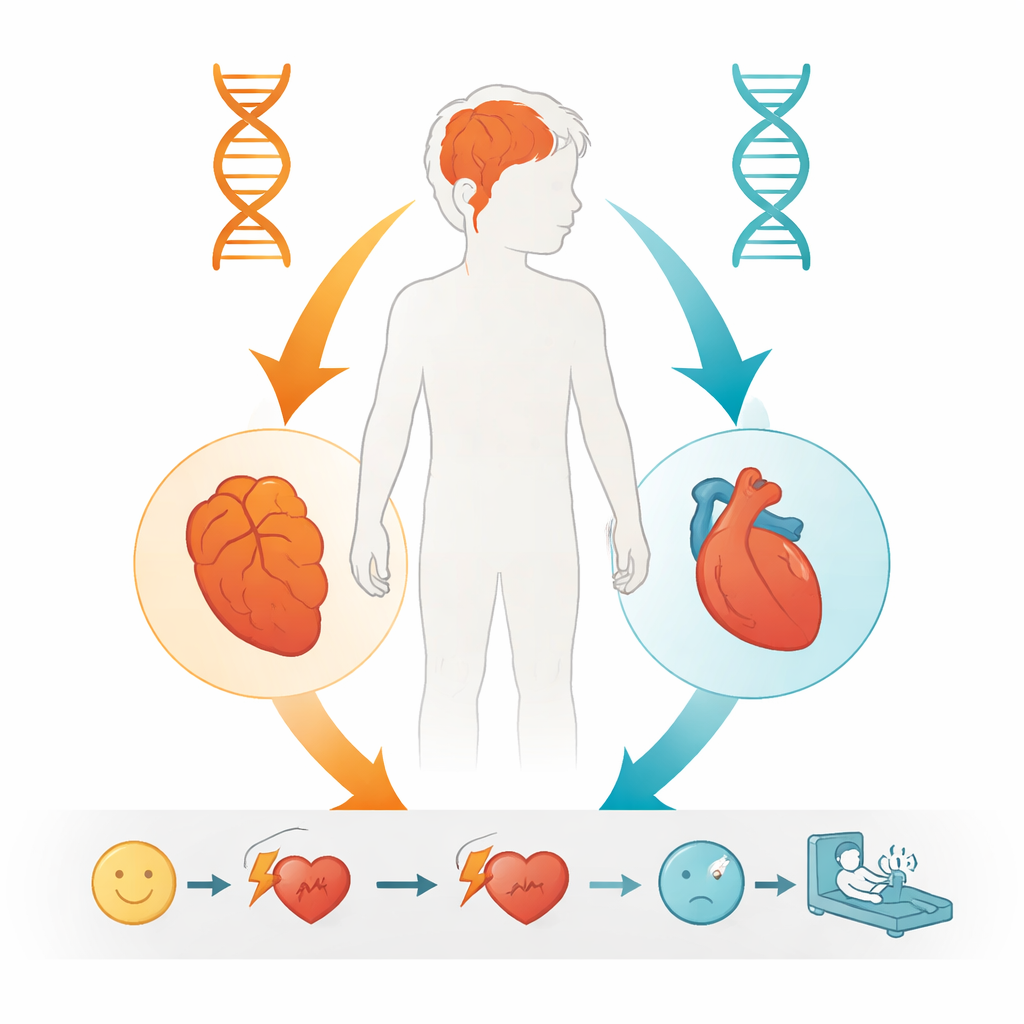
Given these findings, the authors concluded that the boy’s worsening cognitive problems were best explained by his “double-hit” in POLG: one known harmful allele from his father and the newly proven harmful W113R allele from his mother. This fits with other patients who have two faulty POLG copies and develop progressive neurological symptoms starting in childhood or adolescence. The heart rhythm disorder, on the other hand, closely matched what has been seen in people with the Y4725C variant in RYR2, a gene well known to cause dangerous ventricular tachycardia. While the authors cannot fully exclude some interaction between the mitochondrial and heart-channel defects, the clearest picture is that two largely independent genetic problems affected the boy’s brain and heart in parallel.
Why This Case Matters
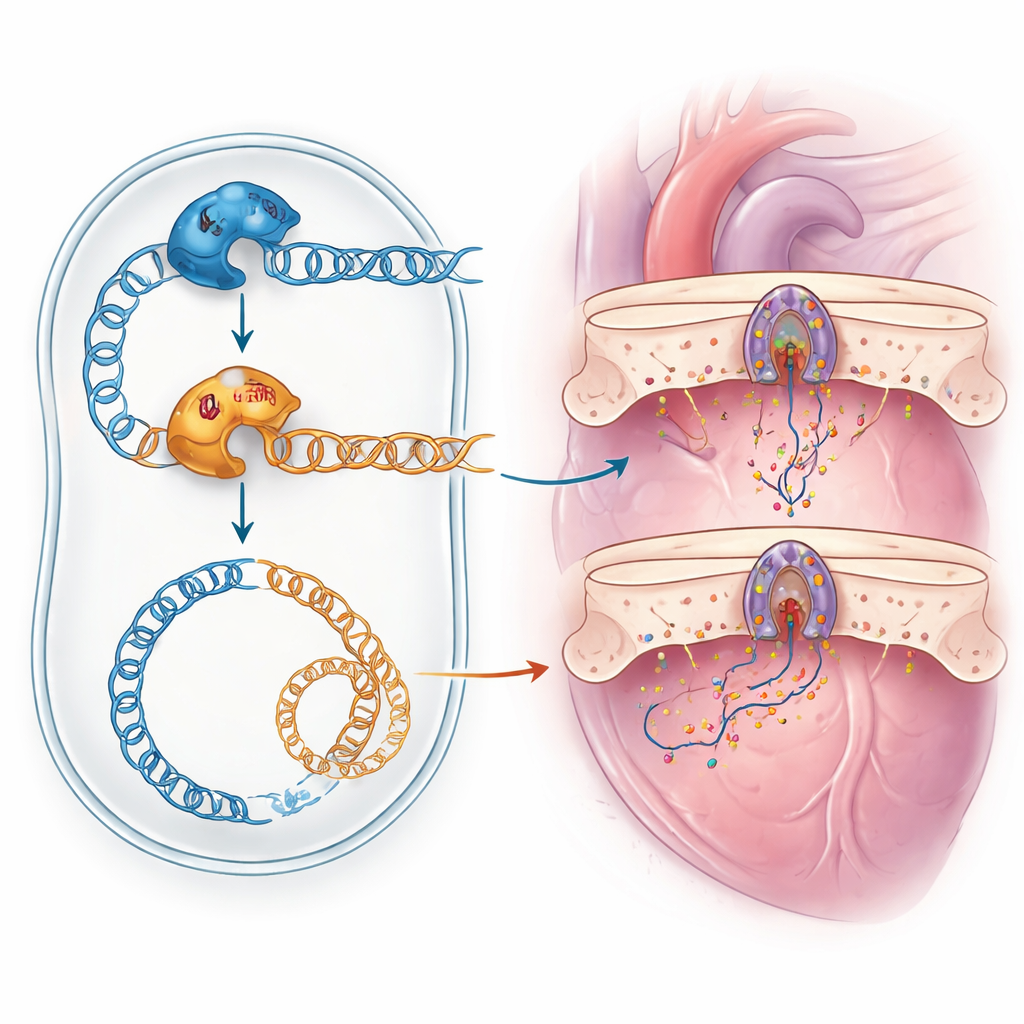
This case highlights several lessons for modern medicine. First, some patients’ complex symptoms are not due to a single rare disease but to “double trouble” from more than one genetic defect, which can easily be missed without careful re-analysis of sequencing data. Second, computer programs alone cannot reliably judge whether a newly discovered DNA change is harmful; functional tests, such as modeling the mutation in yeast, can provide decisive evidence. Finally, clarifying which gene causes which symptom is crucial for accurate counseling of families and for matching patients to emerging treatments, such as experimental drugs aimed at rescuing POLG-related mitochondrial defects. For families and clinicians facing unexplained, multi-system illnesses, this study shows how combining broad genetic testing with smart laboratory models can finally provide answers.
Citation: Fumini, V., Gilea, A.I., Tacchetto, E. et al. Progressive cognitive impairment and ventricular tachycardia in a boy with biallelic POLG variants and a de novo RYR2 variation. Sci Rep 16, 14289 (2026). https://doi.org/10.1038/s41598-026-44913-7
Keywords: mitochondrial disease, genetic arrhythmia, POLG mutation, RYR2 channelopathy, yeast functional assay