Clear Sky Science · en
Thioredoxin protects against diabetic hearing loss by regulating TOMM22 mediated mitochondrial autophagy in hair cells and inhibiting microglial M1 polarization
Why diabetes can quietly steal your hearing
Many people know that diabetes can harm the eyes, kidneys, and nerves, but few realize it can also slowly erode hearing. This study explores why that happens and highlights a natural protective protein in our bodies, thioredoxin, that may both signal early damage and help shield the delicate sound-sensing cells inside the inner ear. Understanding this hidden connection could open the door to earlier diagnosis and new ways to preserve hearing in people living with diabetes.
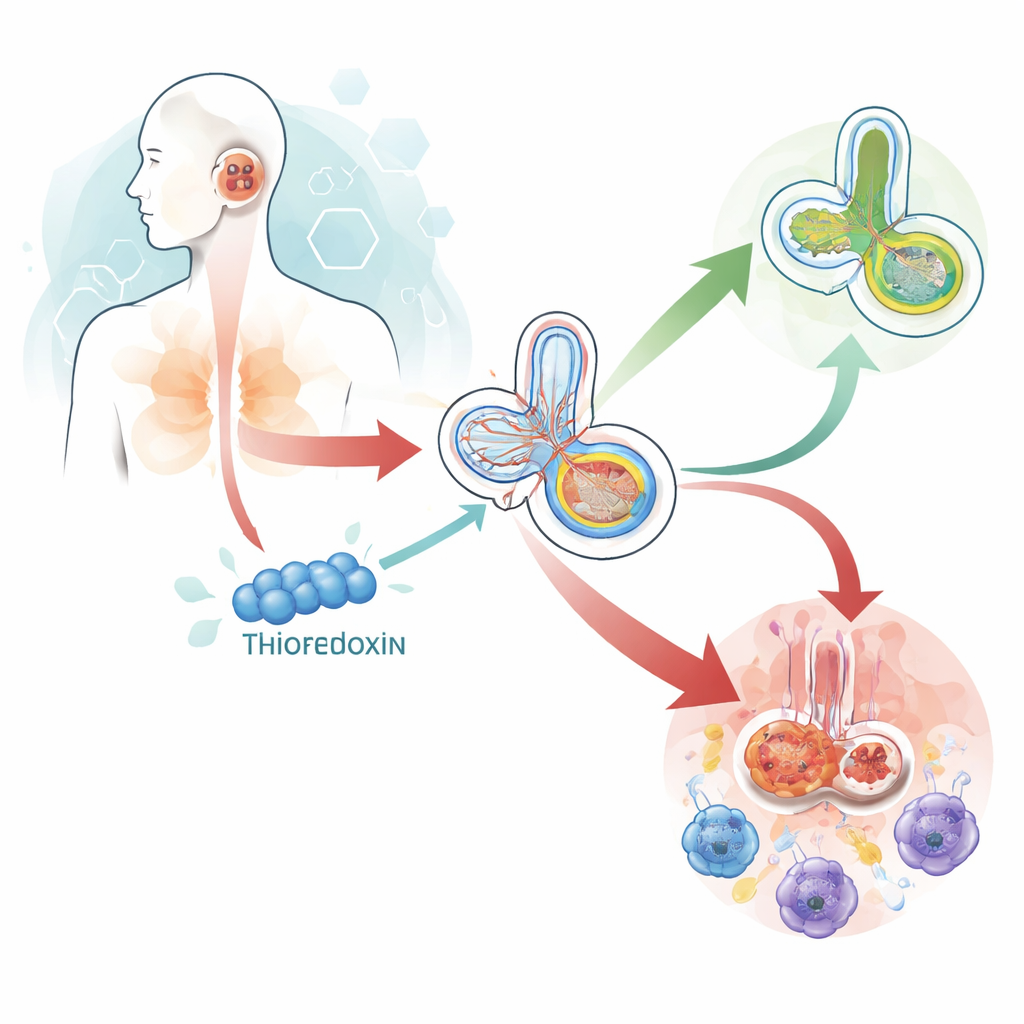
How high blood sugar stresses the inner ear
Diabetes keeps blood sugar elevated for long periods, which churns out harmful by-products and overwhelms the body’s defenses. The inner ear is especially vulnerable because its hair cells and nerve cells are energy-hungry and packed with mitochondria, the cell’s power plants. In a high-sugar environment, these mitochondria generate excess reactive molecules that damage proteins, fats, and DNA. When the damage is too great, cells either destroy faulty mitochondria through a cleanup process or, if overwhelmed, trigger self-destruct programs. Over time, this can thin out the outer hair cells that amplify sound, leading to the characteristic high‑frequency hearing loss seen in many people with diabetes.
A natural defender and early warning signal
Thioredoxin is a small protein that helps keep the cell’s internal chemistry in balance and dampens harmful oxidation. The researchers measured thioredoxin levels in the blood of people with type 2 diabetes and compared them with detailed hearing tests that pick up faint echoes from the cochlea. They found that individuals with diabetic hearing loss had poorer echo signals and higher blood thioredoxin levels. Across the diabetic group, more thioredoxin in the blood went hand in hand with worse cochlear performance. Rather than being purely good or bad, thioredoxin appears to rise as the body struggles to counter stress, making it a promising blood marker that something is going wrong in the inner ear—potentially before everyday conversation becomes difficult.
Protecting the ear’s power plants from overload
To move from correlation to cause, the team used diabetic mice and cultured inner-ear cells to follow what happens inside the cochlea. In diabetic mice, the cochlea showed increased activity of death‑related proteins and an overactive mitochondrial cleanup pathway centered on the PINK1/Parkin system, a well-known quality-control mechanism. Using computational analyses, the authors identified a key gateway protein on the mitochondrial surface, TOMM22, as a hub linking thioredoxin to this pathway. In the diabetic animals, TOMM22 levels dropped while PINK1 and related signals rose, suggesting a shift from orderly maintenance toward destructive overdrive. When mice were engineered to make more thioredoxin, this pattern reversed: cell death signals eased, TOMM22 levels were better maintained, and the mitochondrial cleanup machinery calmed down.
A two-pronged shield: hair cells and immune cells
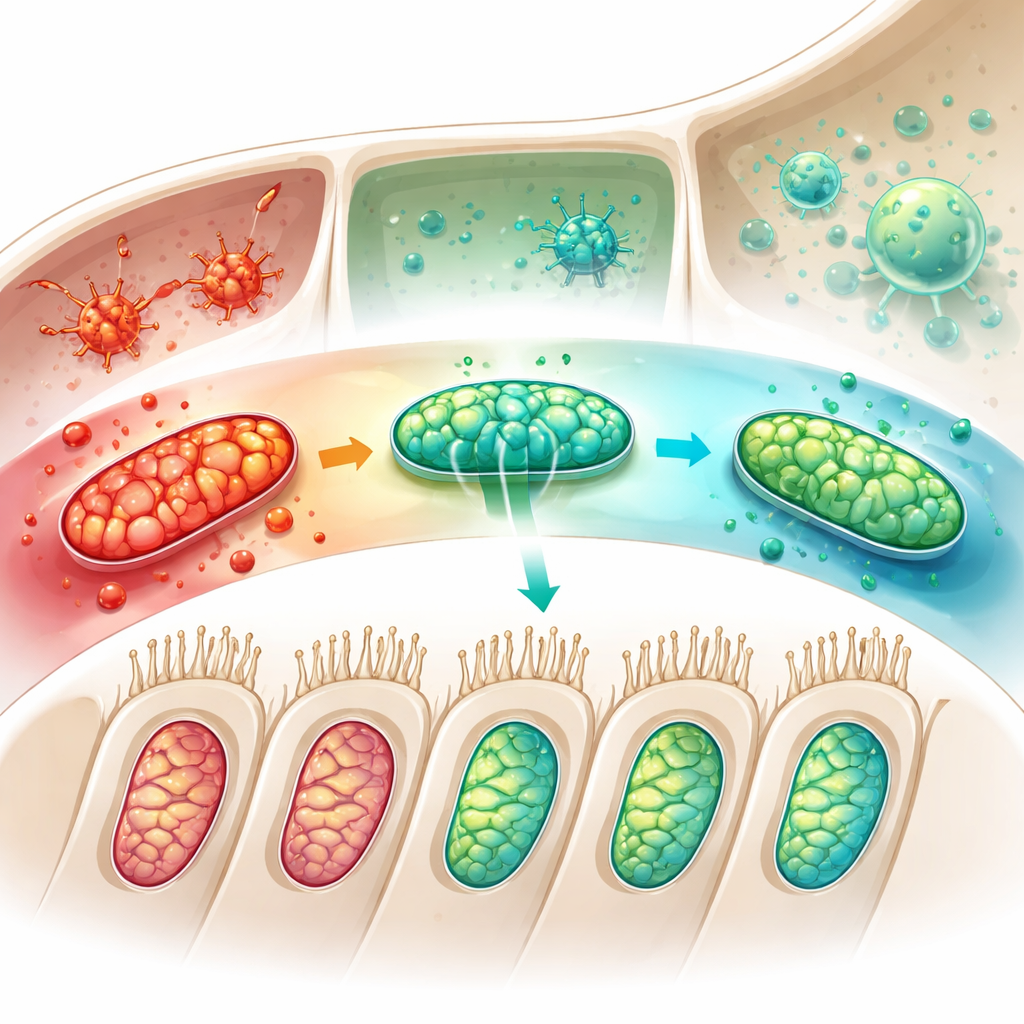
In cultured auditory cells exposed to diabetes‑like stress, boosting thioredoxin restored mitochondrial membrane health, reduced cell death, and dialed back excessive cleanup activity, again in step with preserving TOMM22. When the researchers artificially disrupted mitochondrial function and suppressed TOMM22, these protective effects largely disappeared, underscoring TOMM22’s central role. The study also turned to microglia—immune cells that patrol the inner ear. Under sugar‑related stress, these cells shifted into an aggressive, inflammatory state that can worsen tissue damage. Elevating thioredoxin nudged them instead toward a calmer, repair‑oriented profile. When the secretions from thioredoxin‑rich microglia were applied to auditory cells, mitochondrial function improved and damaging cleanup signals fell, showing that thioredoxin can protect hearing both directly within hair cells and indirectly by reshaping their immune neighborhood.
What this means for people with diabetes
Put simply, this work suggests that thioredoxin acts like both a smoke alarm and a firefighter for the diabetic inner ear. Higher levels in the blood may warn that the cochlea is under oxidative siege, while within the ear thioredoxin helps stabilize a key mitochondrial gatekeeper, reins in overzealous cleanup of damaged power plants, and calms inflammatory immune cells. Together, these actions help preserve the fragile hair cells that convert sound into nerve signals. Although more long-term and human-focused studies are needed, thioredoxin and its partners now stand out as promising targets for blood-based screening and future therapies aimed at preventing or slowing hearing loss in people living with diabetes.
Citation: Zhong, S., Xu, M., Wang, Q. et al. Thioredoxin protects against diabetic hearing loss by regulating TOMM22 mediated mitochondrial autophagy in hair cells and inhibiting microglial M1 polarization. Sci Rep 16, 14332 (2026). https://doi.org/10.1038/s41598-026-44909-3
Keywords: diabetic hearing loss, thioredoxin, mitochondria, inner ear, neuroinflammation