Clear Sky Science · en
Clonidine protects rat hippocampal and cortical neurons from oxygen-glucose deprivation and reoxygenation-induced injury through HCN Channels
Why this matters for stroke and brain health
When a stroke cuts off blood and sugar to the brain, nerve cells can die within minutes, leaving lasting problems with movement, memory, or mood. Doctors have only a short time to restore blood flow, and current drugs cannot fully protect vulnerable brain cells. This study looks at how an existing blood pressure and sedation drug, clonidine, might help shield brain cells from damage after a stroke-like event in the lab, and uncovers how it acts deep inside these cells.
Brain cells under stress
The researchers used rat brain cells taken from two key memory and thinking areas, the cortex and the hippocampus. They exposed these cells to a stroke-like challenge called oxygen-glucose deprivation, which mimics what happens when blood flow stops, followed by reoxygenation, which mimics medical rescue. This combination can injure cells further, much like how restoring blood flow in patients can trigger extra damage. The team measured how many cells survived and how much of a damage-linked enzyme leaked out, giving them a window into cell health under different treatments.
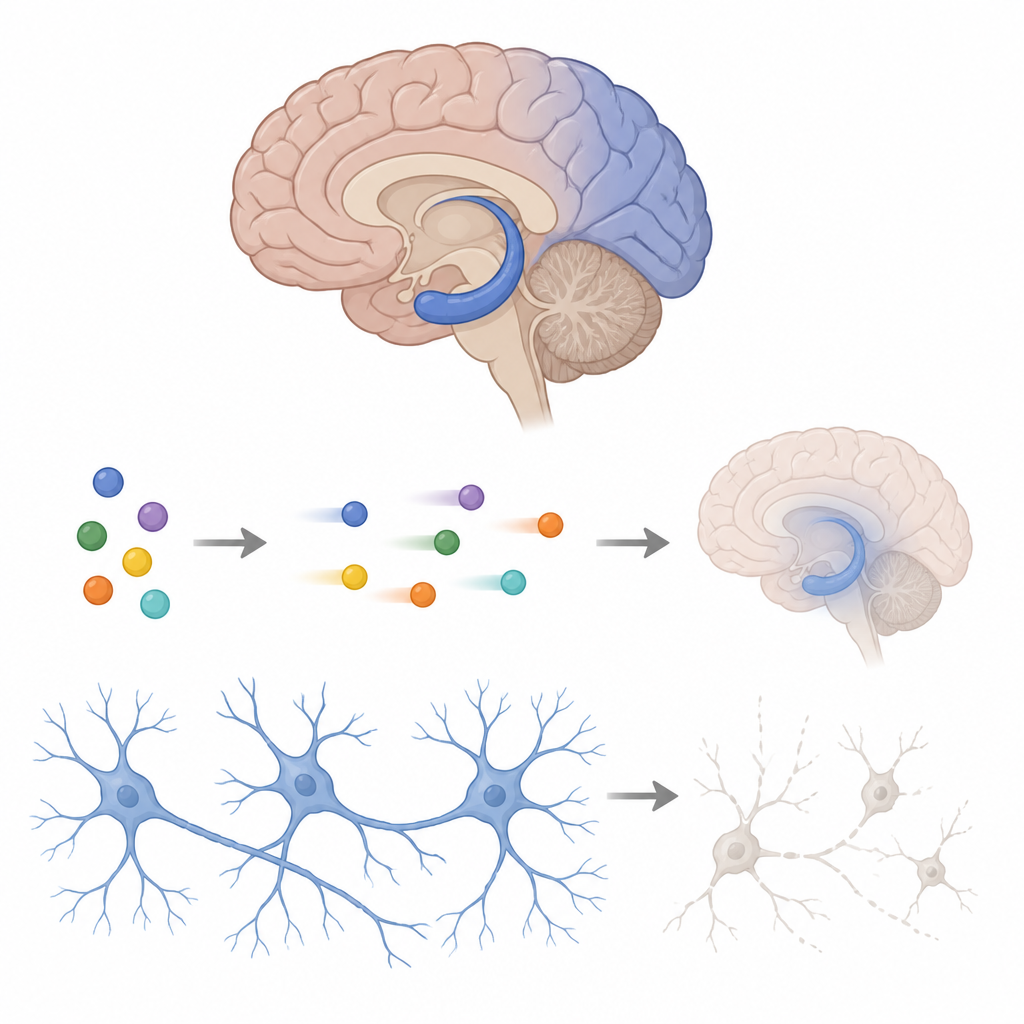
A blood pressure drug as a cell shield
Clonidine, best known for lowering blood pressure and easing withdrawal symptoms, activates certain docking sites on nerve cells called alpha2 adrenergic and imidazoline receptors. In this study, pretreating cells with clonidine helped more neurons survive the stroke-like insult and reduced leakage of the damage enzyme. Another compound that directly blocks a set of ion channels, called HCN channels, also protected the cells, and combining the two worked even better. When the researchers added drugs that block clonidine’s main receptors, its protection weakened, especially when its alpha2 receptor was blocked, showing that this receptor is the main entry point for its protective action.
Calming overactive ion “gates”
HCN channels sit in nerve cell membranes and act like tiny gates that let charged particles in and out, helping set how easily a cell fires signals. After the stroke-like injury, brain cells ramped up production of two common HCN channel types, HCN1 and HCN2. This rise is linked to harmful overactivity and increased stress inside the cell. Clonidine, the HCN-blocking drug, and a separate blocker of a key messenger enzyme all pushed HCN1 and HCN2 levels back down, at both the genetic and protein levels. Together, these treatments reduced the number and activity of these gates, helping to keep the cells’ electrical behavior and internal chemistry from spiraling out of control.
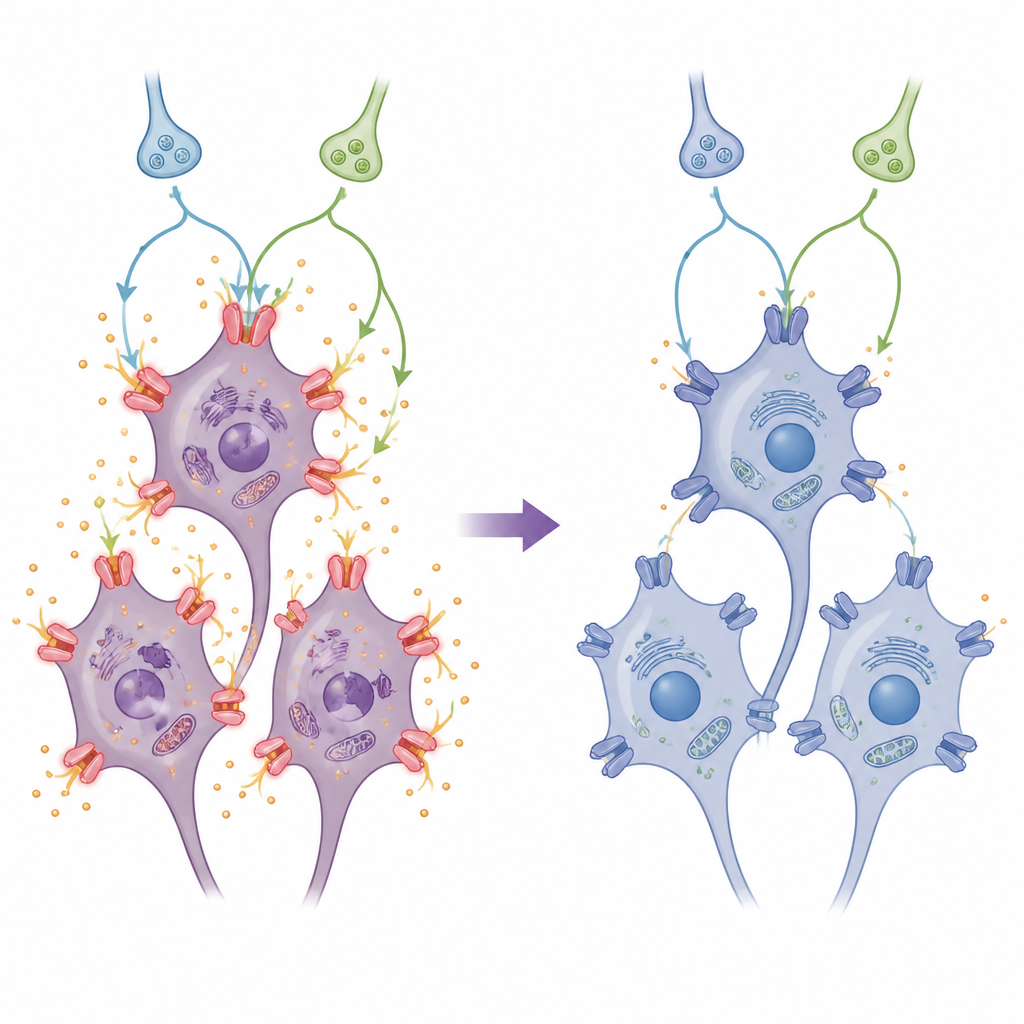
Inside the cell’s warning and survival circuits
The team also tracked two important signaling routes inside neurons. One route, often tied to stress, runs through molecules known as AC, cAMP, and PKA. The other, often linked to cell survival, passes through PI3K and Akt. Stroke-like injury pushed HCN channels up while shifting these routes in harmful ways. Clonidine reversed these shifts: it dampened the AC–cAMP–PKA pathway, which otherwise boosts HCN channel activity, and it restored activation of the PI3K/Akt pathway, which favors cell survival. When the researchers blocked PKA, clonidine’s ability to shut down HCN channels improved; when they blocked PI3K/Akt, clonidine’s protection weakened and HCN channel levels climbed again. This pattern suggests clonidine works by both turning down a damaging alarm circuit and turning up a protective one.
What this could mean for future stroke care
This laboratory work cannot yet tell us whether clonidine will save brain tissue in real patients, and it used pretreatment rather than giving the drug after injury, as would happen clinically. Still, the findings point to a clear story in simple terms: clonidine helps keep stressed brain cells alive by quieting overactive ion gates and rebalancing key survival pathways inside the cell. By tying a familiar drug to these specific molecular switches, the study highlights HCN channels and their linked signaling routes as promising targets for new treatments that might one day limit brain damage after stroke.
Citation: Wang, K., Yan, WJ., Li, G. et al. Clonidine protects rat hippocampal and cortical neurons from oxygen-glucose deprivation and reoxygenation-induced injury through HCN Channels. Sci Rep 16, 15128 (2026). https://doi.org/10.1038/s41598-026-44378-8
Keywords: ischemic stroke, clonidine, HCN channels, neuronal protection, cell signaling