Clear Sky Science · en
Inhibitory influence of three new synthesized cationic gemini surfactants on the corrosion rate of carbon steel in 1 M HCl
Why protecting everyday metals matters
From cars and bridges to oil pipelines deep underground, carbon steel is one of the workhorses of modern life. Yet in harsh acidic environments, this metal can quietly eat away, leading to leaks, failures, and expensive repairs. The study described here explores a new family of detergent‑like molecules that can coat steel and dramatically slow this hidden damage, offering a smarter way to extend the life of critical infrastructure.
New twin‑headed helpers for steel
The researchers designed and built three closely related “gemini” surfactants—molecules that look like two connected soap heads with long oily tails. These twin‑headed molecules carry positive charges and were tailored with different tail lengths to see how structure affects performance. Using established chemical steps, they first formed a backbone containing nitrogen‑rich units and then attached hydrocarbon tails of eight, twelve, or sixteen carbon atoms. Laboratory techniques such as infrared spectroscopy and nuclear magnetic resonance confirmed that the intended structures had been successfully synthesized in high purity.
How these molecules behave in water
Like household detergents, the new surfactants migrate to interfaces and cluster together in water. The team measured how strongly they reduce surface tension and at what concentration they begin to form tiny aggregates known as micelles. They discovered that all three compounds self‑assemble at very low concentrations, but the version with twelve‑carbon tails strikes the best balance: it packs tightly at the water surface, lowers surface tension the most, and forms micelles more readily than its shorter‑tailed cousin. Surprisingly, making the tails even longer to sixteen carbons made clustering less favorable, likely because the extra length causes the molecules to coil and crowd each other. These measurements also showed that both surface adsorption and micelle formation happen spontaneously, driven by a favorable change in free energy.
Putting the shield to the test on steel
To see whether this molecular behavior translates into real protection, the scientists immersed carbon steel samples in strong hydrochloric acid, with and without the new surfactants. They tracked how much metal dissolved by weighing the samples before and after exposure, and they probed the corrosion process using sensitive electrical measurements. In every case, adding the gemini surfactants reduced the rate at which steel dissolved, and higher concentrations offered stronger protection. The twelve‑carbon version again performed best, cutting corrosion rates by more than ninety percent under many conditions. Electrical tests showed that these molecules slow both the metal‑dissolving and gas‑forming sides of the corrosion reaction, acting as “mixed‑type” inhibitors without fundamentally changing the underlying chemistry.
How the invisible barrier works
Careful analysis of the data revealed that the surfactant molecules stick to the steel surface in an orderly, single‑layer fashion that follows a simple packing rule known as the Langmuir isotherm. Thermodynamic calculations and how performance improves with temperature suggest this is mainly a chemical bonding process rather than a loose physical attachment. The positively charged heads can interact with negatively charged species at the steel surface, while the nitrogen‑rich units donate electrons into empty orbitals of iron atoms, strengthening the bond. Once anchored, the long oily tails extend away from the metal, forming a dense, water‑repelling film that blocks acidic species from reaching the steel. Microscopy images back up this picture: bare steel exposed to acid looks rough and scarred, whereas steel treated with the new surfactants appears smooth, indicating a continuous protective coating. 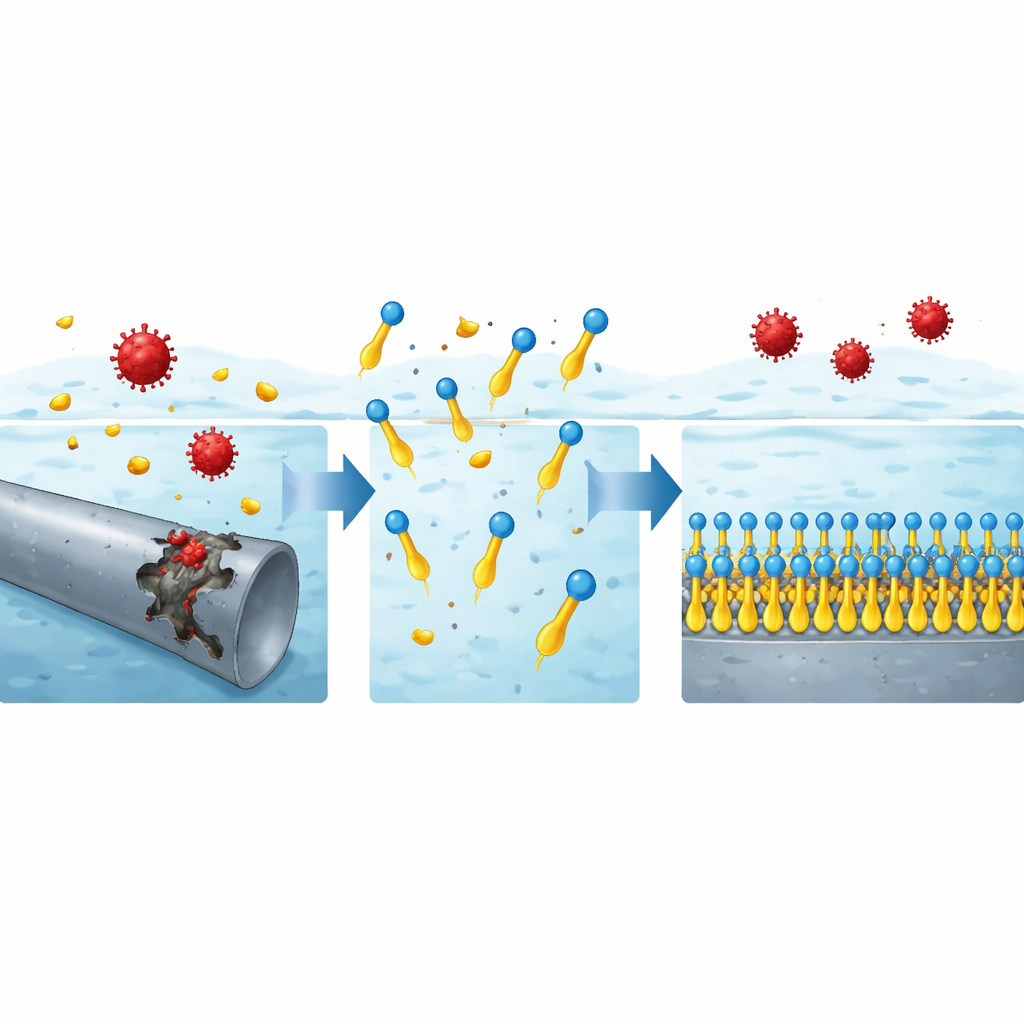
What this means for real‑world systems
In plain terms, the study shows that carefully engineered twin‑headed surfactants can behave like a cling‑on raincoat for steel in acid, greatly slowing the rate at which the metal dissolves. Among the three variants tested, the molecule with medium‑length tails offers the strongest, most efficient protection, thanks to its ability to pack tightly and bond firmly to the surface. Because such inhibitors can be added in small amounts to existing acidic cleaning and processing solutions, they offer a practical way to extend the lifetime of pipelines, reactors, and other steel equipment while potentially reducing maintenance costs and environmental impact. 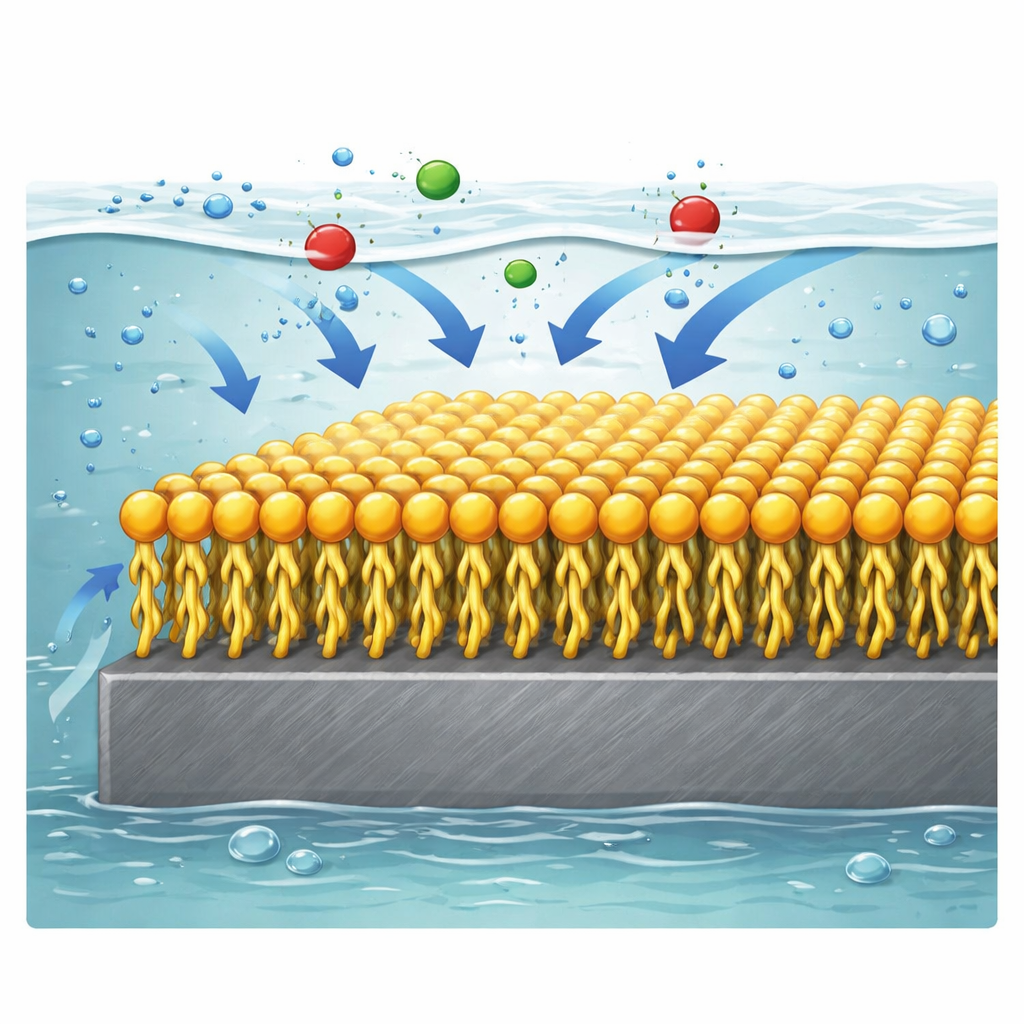
Citation: Abdelhafiz, F.M., Sami, R.M., Ghiaty, E.A. et al. Inhibitory influence of three new synthesized cationic gemini surfactants on the corrosion rate of carbon steel in 1 M HCl. Sci Rep 16, 12055 (2026). https://doi.org/10.1038/s41598-026-44281-2
Keywords: carbon steel corrosion, corrosion inhibitors, gemini surfactants, acidic environments, surface protection