Clear Sky Science · en
Naringenin-loaded nanoparticles ameliorate scopolamine-induced neurotoxicity
Why a Citrus Molecule and Tiny Particles Matter for the Brain
As populations age, more families are touched by memory loss and dementia, yet today’s drugs mostly offer temporary symptom relief. This study explores an inventive idea: taking naringenin, a natural compound found in citrus fruits, and packaging it into tiny particles so it can better reach and protect the brain. The researchers then test whether this nano‑form of naringenin can soften the impact of a drug that temporarily scrambles memory in mice, and whether pairing it with the standard Alzheimer’s drug donepezil might boost benefits while limiting side effects.
Turning a Fruit Molecule into a Brain‑Ready Medicine
Naringenin has long intrigued scientists because it can calm inflammation, neutralize harmful oxygen molecules, and influence key brain signaling systems. The catch is that when swallowed in its usual form, very little of it actually makes it into the bloodstream, let alone the brain. To solve this, the team created naringenin‑loaded nanoparticles—spheres about 95 nanometers across, far smaller than a red blood cell—using common surfactants to keep them stable and evenly dispersed. Detailed imaging and physical measurements showed that these particles were smooth, uniform, and carried a negative surface charge that helps them stay separated in liquids and move through the body. Lab tests suggested they release naringenin slowly over many hours, which could help maintain steady protection rather than sharp bursts.
Stress‑Testing the New Particles in a Memory Model
To see whether these particles actually help a living brain, the researchers turned to a well‑known mouse model of temporary memory trouble. They gave animals scopolamine, a drug that briefly blocks a key communication system based on the messenger acetylcholine and also stirs up oxidative stress and inflammation—features that echo some early changes seen in neurodegenerative disease. Mice then received either the naringenin nanoparticles alone, donepezil alone, both together, or no treatment. The team measured learning in a water maze, blood fats linked to vessel health, chemical signs of oxidative damage and inflammation in brain tissue, activity of protective enzymes, expression of certain brain‑related genes, and microscopic changes in the hippocampus, a region crucial for memory.
What Happened Inside the Brain
Scopolamine alone impaired maze learning, damaged brain cells, increased harmful oxidants and inflammatory molecules, and disturbed the brain’s lipid profile. Naringenin nanoparticles clearly blunted these problems. Treated mice learned the maze more quickly, showed higher levels of the brain’s own antioxidant defenses, and had lower levels of damaging by‑products. Inflammatory signals and markers of tissue injury dropped, while brain fats shifted toward a healthier pattern that may support blood vessel and cell‑membrane function. At the microscopic level, brain sections from treated animals showed more orderly cell layers and far fewer signs of degeneration. When nanoparticles were combined with a modest dose of donepezil, improvements were even more striking, suggesting the two approaches complement one another—one boosting chemical signaling, the other protecting cells from stress and inflammation.
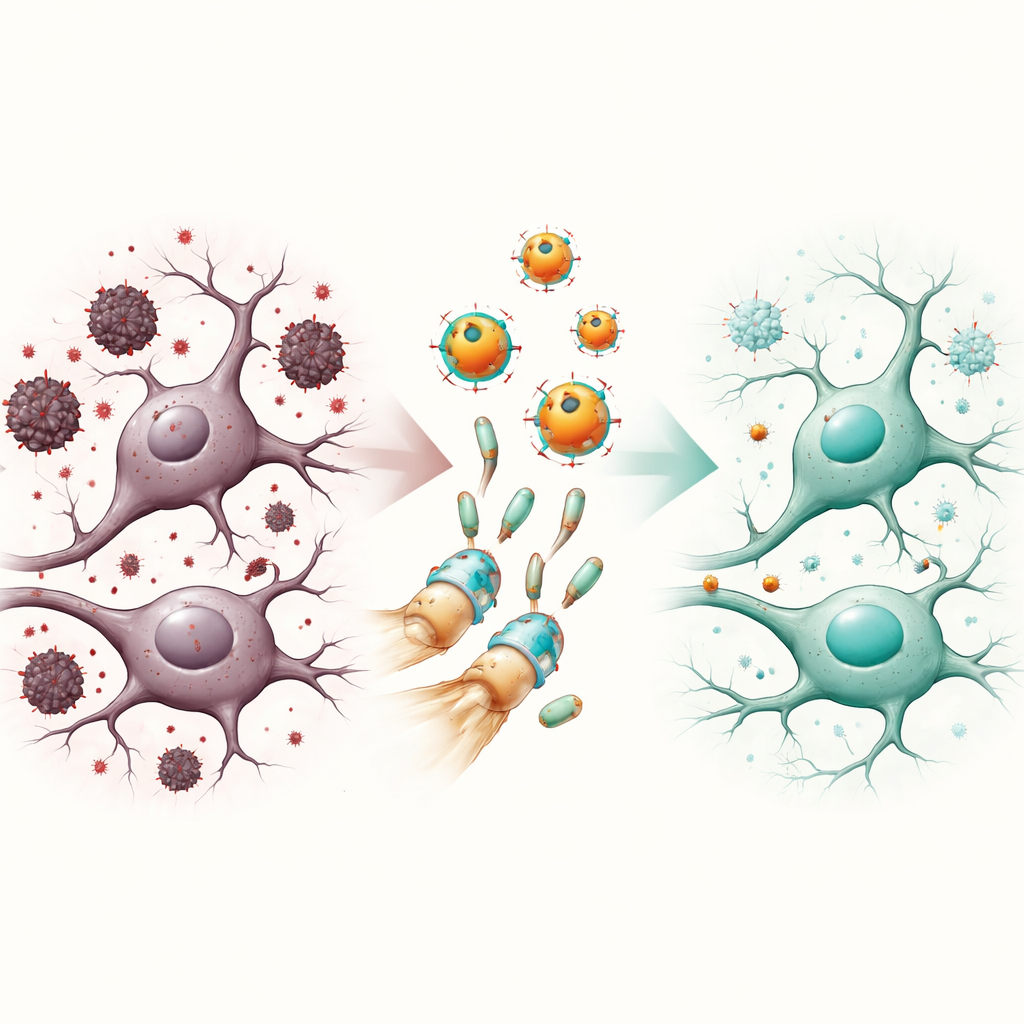
Clues to How the Protection Works
Beyond these broad patterns, the team probed several molecular “hubs.” They found that scopolamine reduced levels of a receptor subunit linked to calming brain activity and raised levels of a kinase enzyme often tied to harmful protein changes and inflammation. Naringenin nanoparticles reversed these shifts, and computer‑based docking studies suggested that naringenin can physically fit into and influence both targets. The treatment also restored activity in a signaling pathway associated with cell survival and plasticity. Together, these findings paint the nanoparticles as multi‑talented protectors that guide brain circuits back toward balance, not just by mopping up chemical damage, but by nudging key switches that control how neurons respond to stress.
What This Means—and What It Doesn’t
For a lay reader, the bottom line is that packaging a citrus‑derived compound into tiny carriers made it safer, more stable, and far more effective at shielding mouse brains from a short‑term chemical insult. The nano‑naringenin eased memory problems, calmed oxidative and inflammatory storms, improved blood fat patterns, and preserved brain structure, especially when paired with a lower dose of a standard dementia drug. However, the authors are careful to stress that this model mimics an acute, reversible disruption, not the slow, relentless build‑up of disease‑related proteins seen in Alzheimer’s. That means these results highlight symptom‑relevant protection rather than proof of a true disease‑modifying therapy. To know whether such nanoparticles can actually slow or prevent degenerative brain diseases in people, they must next be tested in long‑term animal models with progressive pathology and in studies that directly confirm how and where naringenin acts inside the brain.
Citation: Alqarni, A., Abd-Elghany, A.A., Bedewi, M.A. et al. Naringenin-loaded nanoparticles ameliorate scopolamine-induced neurotoxicity. Sci Rep 16, 13468 (2026). https://doi.org/10.1038/s41598-026-44225-w
Keywords: naringenin nanoparticles, neuroprotection, oxidative stress, Alzheimer’s models, donepezil combination