Clear Sky Science · en
CHD1L promotes testicular cancer progression through epigenetic activation of the CXCR6/PI3K/AKT pathway
Why this research matters to young men
Testicular cancer is the most common solid cancer in young men, and while early cases are often curable, some tumors spread or stop responding to standard chemotherapy. This study digs into the inner workings of testicular cancer cells and the immune environment around them, uncovering a key driver molecule called CHD1L. By showing how CHD1L helps tumors grow, spread, and dodge immune attack, the work points to a new weak spot that future drugs might target when current treatments fail.
A hidden switch inside testicular tumors
The researchers began by asking whether CHD1L, a protein known to reshape DNA packaging in other cancers, also plays a role in testicular cancer. Using tumor samples from patients and data from The Cancer Genome Atlas, they found that CHD1L levels were much higher in testicular tumors than in normal testis tissue. High CHD1L was especially common in more advanced and invasive tumors, suggesting that this protein is not just present, but linked to aggressive disease. Statistical analyses showed that CHD1L expression could help distinguish cancer tissue from normal tissue with good accuracy.
A tumor that grows faster and moves farther
To test what CHD1L actually does, the team manipulated its levels in testicular cancer cell lines grown in the lab. When they boosted CHD1L, cells divided more quickly, formed many more colonies, moved faster across artificial “wounds,” and invaded through barriers that mimic tissue. When they reduced CHD1L with targeted RNA tools, the opposite happened: cell growth slowed, programmed cell death increased, and the cells became less able to migrate and invade. In mice, tumors grown from CHD1L‑boosted cells were larger, grew faster, and showed more of both CHD1L and a proliferation marker, confirming that this protein can drive tumor growth in a living organism.
How CHD1L rewires cell signals
CHD1L does not act like a simple on–off switch; it works by reshaping how DNA is packaged, which in turn controls which genes are turned on. The scientists used powerful genome‑wide methods to measure both which regions of DNA were open and accessible, and which genes were active, in cells with or without CHD1L. They found that CHD1L helps keep certain stretches of DNA open near key genes involved in chemical messaging between cells. One standout target was CXCR6, a receptor on the cell surface that can activate an internal growth pathway known as PI3K/AKT. When CHD1L was knocked down, the DNA region near the CXCR6 gene became more tightly packed, CXCR6 levels dropped, and the PI3K/AKT pathway was dampened. Further experiments showed that this packaging change involved an increase in a repressive chemical mark on histones called H3K9me3, which accumulated at the CXCR6 control region when CHD1L was missing.
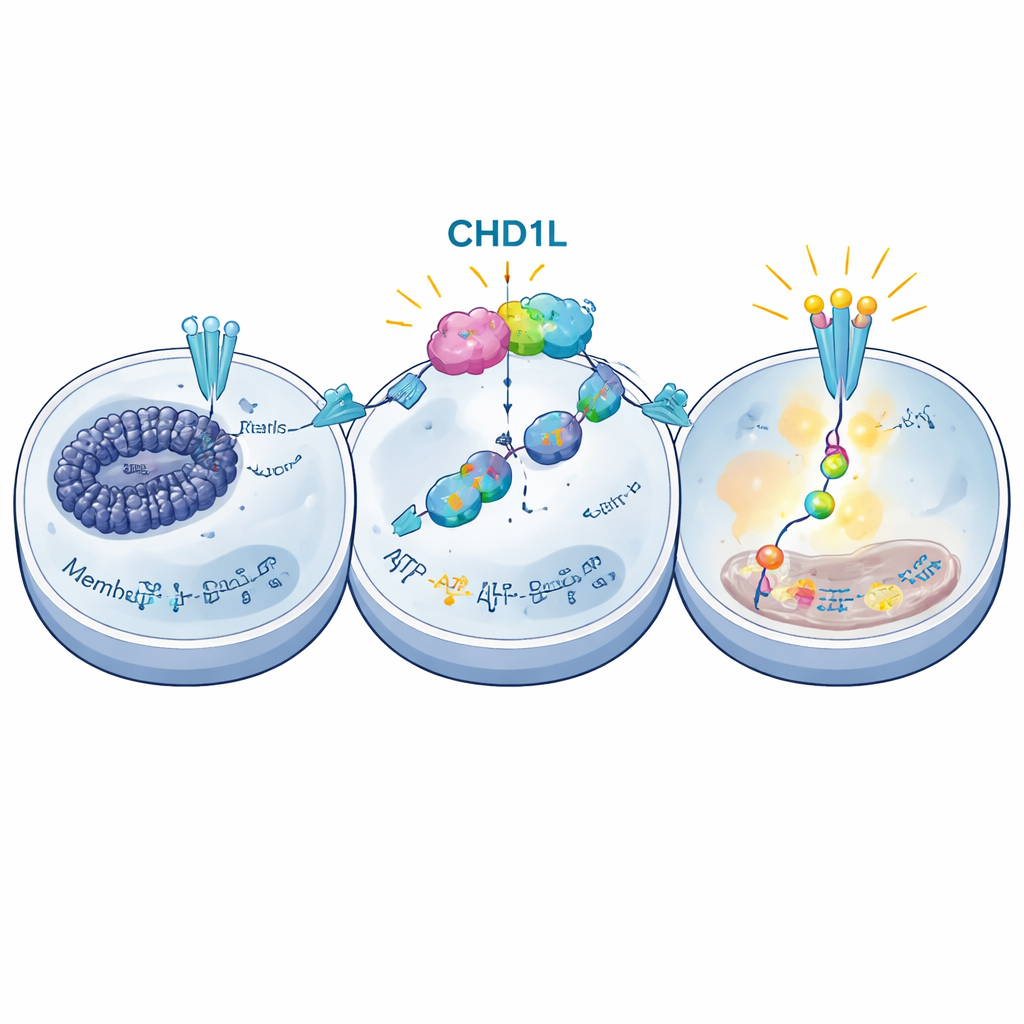
Escaping immune attack in the tumor neighborhood
Beyond the cancer cells themselves, the team also explored how CHD1L relates to the immune cells that infiltrate tumors. By analyzing large patient datasets, they observed that tumors with more CHD1L tended to show patterns of immune “exclusion,” where harmful immune cells such as killer T cells and natural killer cells were less active or kept at bay, while cell types linked to suppression, such as regulatory and certain helper T cells, were more prominent. These tumors had higher overall immune and stromal scores but still signs of impaired immune function, suggesting that CHD1L‑high cancers may surround themselves with an immune landscape that looks busy but is ineffective at attacking the tumor.
What this means for future treatment
Taken together, the findings show that CHD1L acts as a master controller that helps testicular cancer cells grow, spread, and possibly evade immune destruction by epigenetically switching on the CXCR6/PI3K/AKT signaling axis and altering the tumor microenvironment. For patients, especially those with advanced or treatment‑resistant disease, this points to CHD1L as a promising new marker to identify high‑risk tumors, and as a potential target for next‑generation therapies that work by reprogramming the cancer cell’s gene activity rather than simply killing dividing cells. While more studies are needed, including drug development and tests in more realistic models, this work opens a new avenue for tackling difficult cases of testicular cancer.
Citation: Zhao, Y., Yan, H., Chen, X. et al. CHD1L promotes testicular cancer progression through epigenetic activation of the CXCR6/PI3K/AKT pathway. Sci Rep 16, 13955 (2026). https://doi.org/10.1038/s41598-026-43901-1
Keywords: testicular cancer, CHD1L, epigenetic regulation, CXCR6 PI3K AKT pathway, tumor microenvironment