Clear Sky Science · en
Wearable sensors for monitoring drug pharmacokinetics in patients with Parkinson’s disease
Why this new wearable matters
People with Parkinson’s disease often rely on a decades‑old drug called L‑DOPA to keep their movements steady. Yet the drug’s effects can come and go unpredictably over the course of a day, making it hard for patients and doctors to know when the dose is just right. This study explores a small wearable patch that reads drug levels from sweat, offering a painless way to track medication in real time and relate it directly to symptoms like hand tremor.
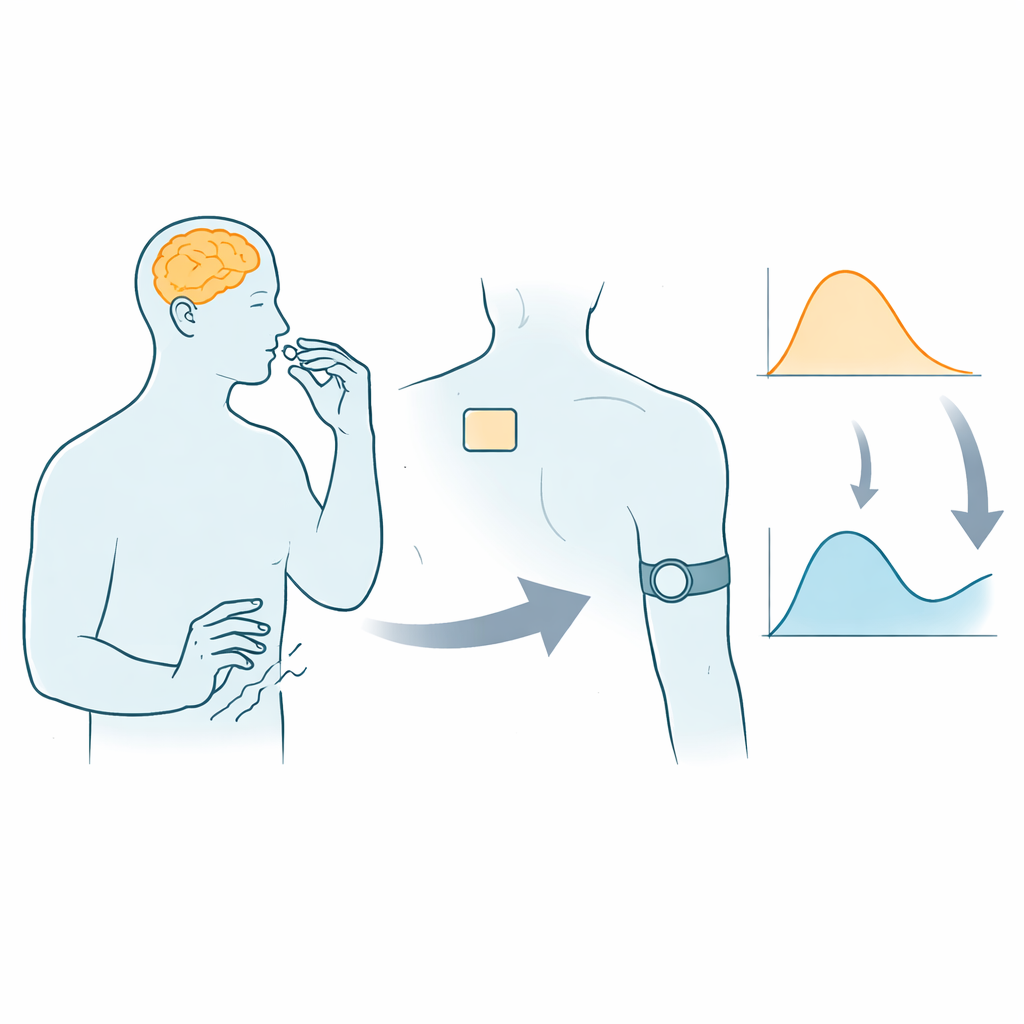
The challenge of timing Parkinson’s medicine
Parkinson’s disease gradually robs people of smooth movement and can also affect sleep, mood, and thinking. L‑DOPA remains the most effective medicine to ease many of these problems, but its benefits depend strongly on how much of the drug is in the body at any given moment. That level can swing up and down because of differences in digestion, diet, and disease stage. Today, doctors mostly adjust doses using rating scales and patient diaries, which provide only brief snapshots and are influenced by memory and judgment. As a result, many people spend part of the day “off,” when symptoms surge back, or “over‑dosed,” when excessive movements appear.
Turning sweat into a drug readout
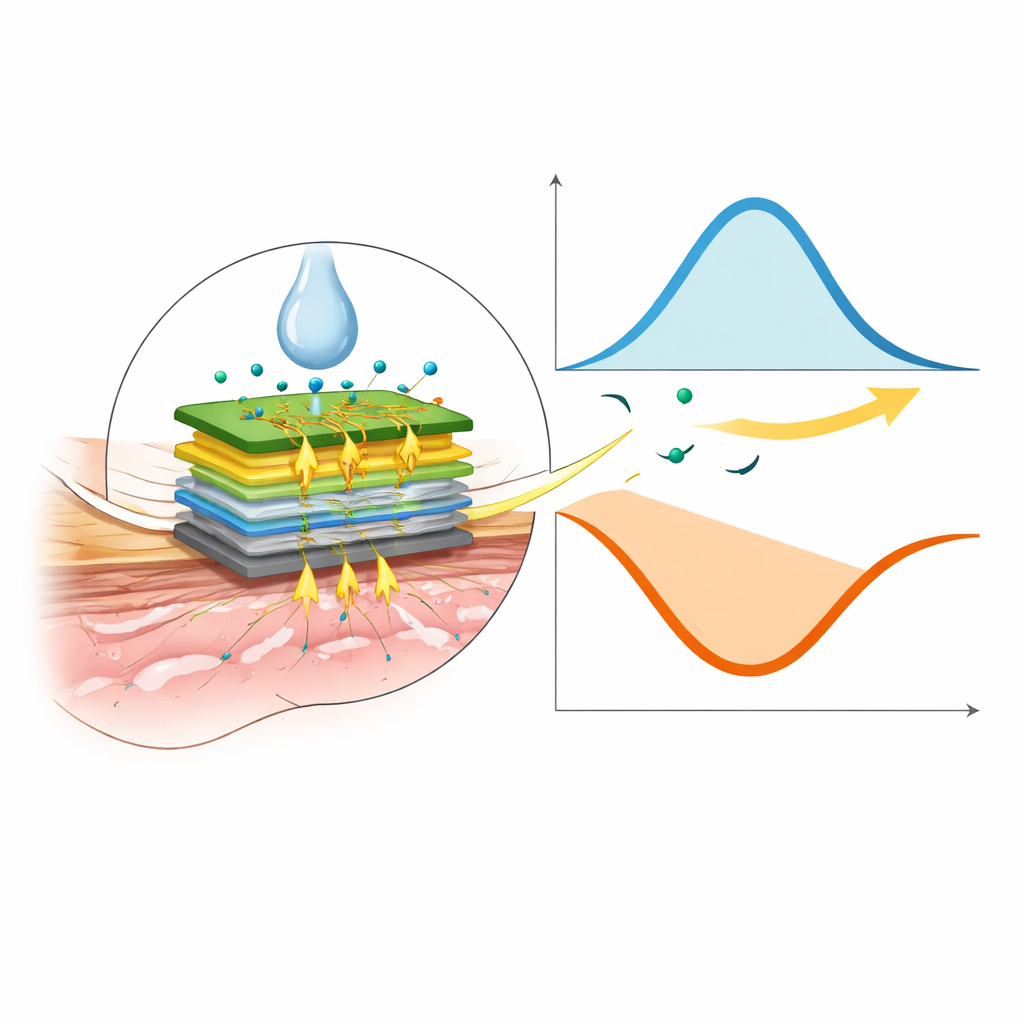
The researchers set out to build a wearable sensor that could track L‑DOPA in an easy‑to‑reach body fluid: sweat. They used a small commercial gold electrode and coated it with several microscopic layers, including branching gold structures and an enzyme that reacts with L‑DOPA. When sweat touches this surface, the reaction generates an electrical signal whose strength reflects how much drug is present. In the lab, the team showed that the sensor responds linearly to L‑DOPA across the medically relevant range, with only small changes caused by normal shifts in skin temperature or sweat acidity. Tests in real human sweat confirmed that the device can reliably distinguish L‑DOPA from common interfering substances such as uric acid, vitamin C, and glucose.
Testing the patch in real patients
To see how the technology performs in daily‑life conditions, the team recruited 39 people with Parkinson’s disease at a hospital in Taiwan; 32 completed the full study. After skipping their usual Parkinson’s drugs overnight, participants went through four rounds of testing spaced 30 minutes apart. In each round, they wore an inertial sensor on the arm to record tremor, had the sweat patch attached to the upper back, and performed a series of movement tasks such as hand‑cycling, walking in a hallway with a smartwatch, and drawing circles. After the first round, they took their usual oral L‑DOPA tablets, and the later rounds captured how both drug levels and movement changed as the medication took effect and then began to wear off.
Linking drug levels to body movement
For 24 participants who produced enough sweat to analyze, the scientists compared two time‑courses: L‑DOPA levels from the patch and tremor intensity from the motion sensor. In about four out of five of these patients, higher sweat L‑DOPA readings were associated with weaker tremor, showing a clear inverse relationship. The study also grouped tremor patterns into several types, such as a steady improvement right after dosing or an initial improvement followed by a rebound as the drug level fell. Beyond tremor, other movement measures—hand‑cycling speed, walking pace, and the time needed to complete a drawing task—often rose and fell in step with the sweat drug profile, hinting that a single biochemical readout can summarize complex physical responses.
What this could mean for everyday care
This pilot study suggests that a thin sweat patch, paired with simple motion sensors, can provide a window into how Parkinson’s medication behaves inside the body without drawing blood. By showing that sweat L‑DOPA levels track closely with changes in tremor for most patients, the work points toward a future in which doctors fine‑tune doses based on objective, round‑the‑clock data rather than occasional clinic visits and patient recall. With further validation against blood measurements and longer‑term studies, such wearables could support more personalized treatment schedules, reduce time spent in disabling “off” periods, and eventually feed into smart, closed‑loop systems that automatically adjust therapy as a person’s symptoms fluctuate.
Citation: Guo, YJ., Li, CC., Huang, JA. et al. Wearable sensors for monitoring drug pharmacokinetics in patients with Parkinson’s disease. Sci Rep 16, 13332 (2026). https://doi.org/10.1038/s41598-026-43825-w
Keywords: Parkinson’s disease, wearable sensors, sweat monitoring, L-DOPA, tremor tracking