Clear Sky Science · en
Validation of ferroptosis in zebrafish as a reliable model for phenotypic studies
Why tiny fish matter for understanding cell death
Scientists have discovered a relatively new way that cells can die, called ferroptosis, which is driven by iron and runaway chemical reactions that damage fats in cell membranes. This process has been linked to cancer, brain disease, heart problems, and blood vessel disorders, but most research so far has relied on mice, which are slow and costly to study. This paper explores whether zebrafish—small, fast‑growing aquarium fish whose genes closely resemble ours—can provide a quicker, clearer window into ferroptosis and its effects on growing tissues like blood vessels and bone.
A new kind of cell death under the microscope
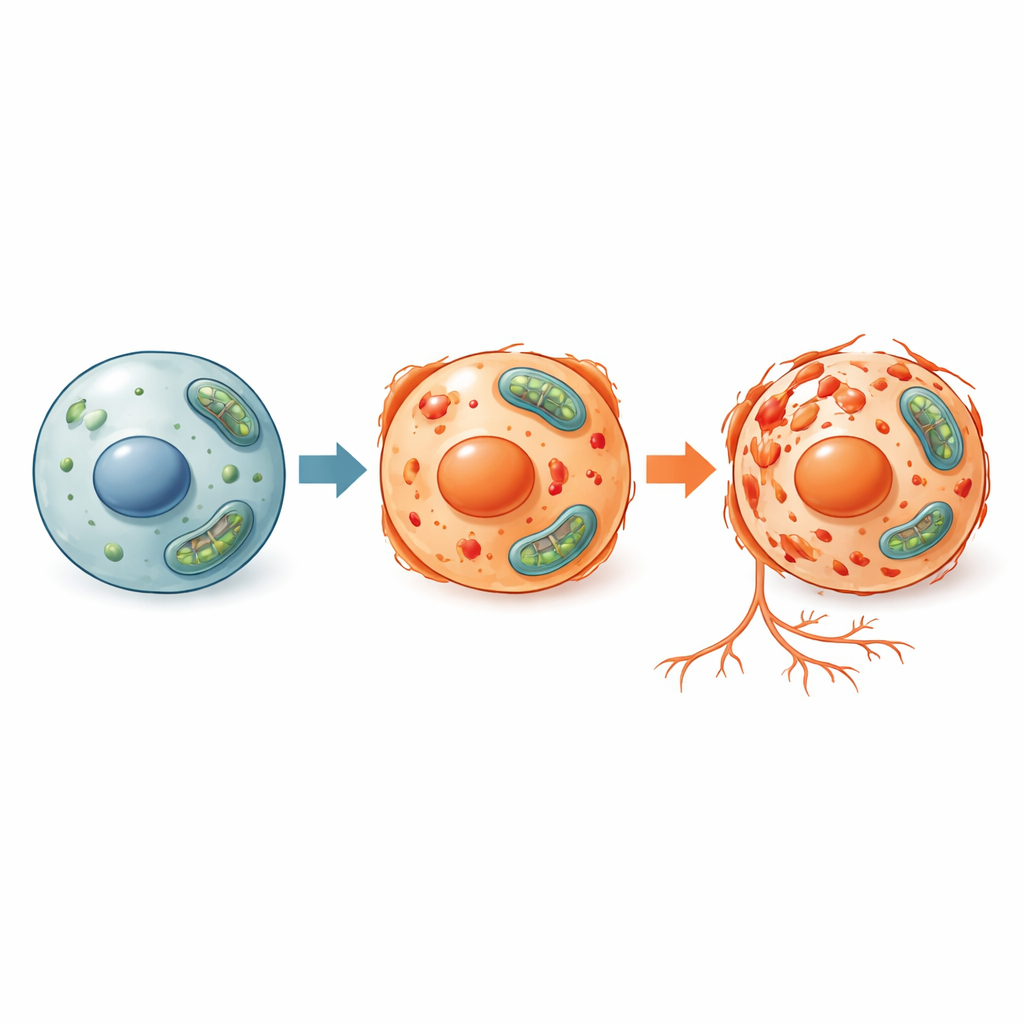
Ferroptosis differs from better‑known forms of cell death such as apoptosis (a tidy, programmed self‑destruction) and necrosis (a more chaotic collapse). In ferroptosis, iron and reactive oxygen molecules team up to attack the fats that make up cell membranes, eventually causing the cell to fail. Because this pathway is tightly tied to iron handling and antioxidant defenses, it may play a key role in conditions where iron builds up or where tissues are under oxidative stress, including some tumors, strokes, and degenerative diseases. To study this process and test possible treatments, researchers need animal models that are both biologically relevant and experimentally flexible.
Why zebrafish are a powerful stand‑in for humans
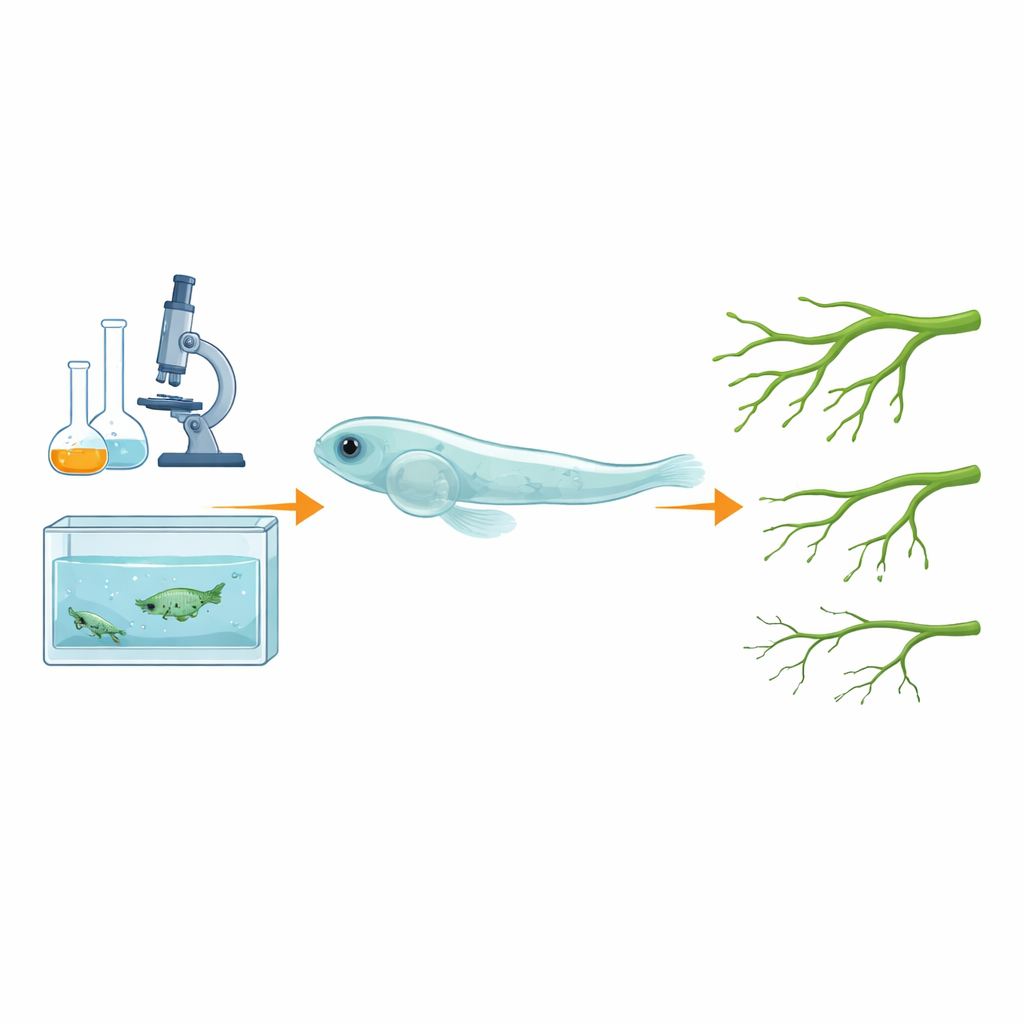
Zebrafish offer several advantages as a living laboratory. They are vertebrates with about 85 percent of their genes matching human genes, and their signaling pathways for growth and disease are strikingly similar. Their embryos develop outside the mother and are transparent, so scientists can directly watch organs, blood vessels, and bones form in real time. Zebrafish are inexpensive to breed in large numbers, making them ideal for testing many genetic changes or drug candidates at once. The key question this study asked was whether the hallmarks of ferroptosis seen in mammalian cells—iron‑dependent oxidative damage, changes in protective molecules, and shifts in certain genes and proteins—also appear in zebrafish, and whether this can be cleanly separated from other types of cell death.
Triggering ferroptosis in living zebrafish
The researchers exposed young zebrafish to three different triggers that are known to cause ferroptosis: two chemical inducers (Erastin and FIN56) and a form of iron called ammonium ferric citrate. They measured several signs of stress inside the fish, including the buildup of reactive oxygen species, the balance between a key antioxidant (glutathione) and its oxidized form, and the presence of damaged fats. They also examined which genes were turned on or off and measured levels of a central protective protein called GPX4, which normally shields cell membranes from oxidative attack. In all three treatment conditions, zebrafish cells showed the expected ferroptosis signature: more oxidative damage, a disturbed antioxidant balance, reduced GPX4 protein, and activation of ferroptosis‑linked genes. At the same time, markers of apoptosis and autophagy changed little, indicating that the cell death being observed was specifically ferroptotic.
How ferroptosis disrupts growing blood vessels
To see how this process affects a whole organ system, the team used a transgenic zebrafish line whose blood vessels glow under the microscope. When these fish were treated with the ferroptosis inducer Erastin, the normally bright, well‑branched vessels along the back appeared thinner and dimmer, suggesting that vessel growth had been stunted. When the scientists added drugs that block ferroptosis or bind excess iron, many of these defects were eased, implying that iron‑driven ferroptosis in the cells lining blood vessels was directly impairing vascular development. Because blood vessel growth is tightly connected to bone formation—a relationship known as osteogenesis‑angiogenesis coupling—these findings position zebrafish as a promising model not just for vascular diseases, but also for bone disorders linked to iron overload and oxidative damage.
What this means for future disease research
By showing that ferroptosis in zebrafish closely mirrors what has been seen in mammalian systems, and that it can be induced, measured, and even reversed with specific drugs, this study validates zebrafish as a practical, reliable model for studying iron‑driven cell death. For a lay reader, the takeaway is that tiny transparent fish can help scientists quickly test how ferroptosis contributes to conditions such as cancer, stroke, vessel disease, and osteoporosis, and screen for treatments that might protect vulnerable tissues. In short, zebrafish offer a fast, visual, and genetically tractable way to probe a destructive form of cell death that is increasingly recognized as a common thread in many human illnesses.
Citation: Cao, Z., Liu, G., Zhang, R. et al. Validation of ferroptosis in zebrafish as a reliable model for phenotypic studies. Sci Rep 16, 14357 (2026). https://doi.org/10.1038/s41598-026-43493-w
Keywords: ferroptosis, zebrafish, oxidative stress, angiogenesis, iron overload