Clear Sky Science · en
Selective extraction of lithium from acidic chloride leachates of spent batteries
Why old batteries still matter
Lithium ion batteries power our phones, laptops, and electric cars, but the lithium inside them is hard to recover once the batteries reach the end of their life. Today, much of that lithium is lost or recovered only after many costly steps. This study explores a simpler way to pull lithium out early from the acidic liquid produced when shredded batteries are chemically dissolved, with the goal of saving energy, chemicals, and valuable metals.
Turning shredded cells into useful liquid
Modern battery recycling often starts with “black mass,” a dark powder made by shredding spent cells. This powder is rich in lithium but also in nickel, cobalt, manganese, and other metals. One common approach is to dissolve black mass in hydrochloric acid, making a salty liquid that holds almost all the metals at once. The challenge is that existing recovery methods usually focus on the more expensive metals first, leaving lithium to the end, when it is diluted, partly lost, and mixed with many impurities. Recovering lithium earlier from the same acidic liquid would be more efficient, but it requires a material that can selectively grab lithium without bringing along other metals.
A tailored liquid that prefers lithium
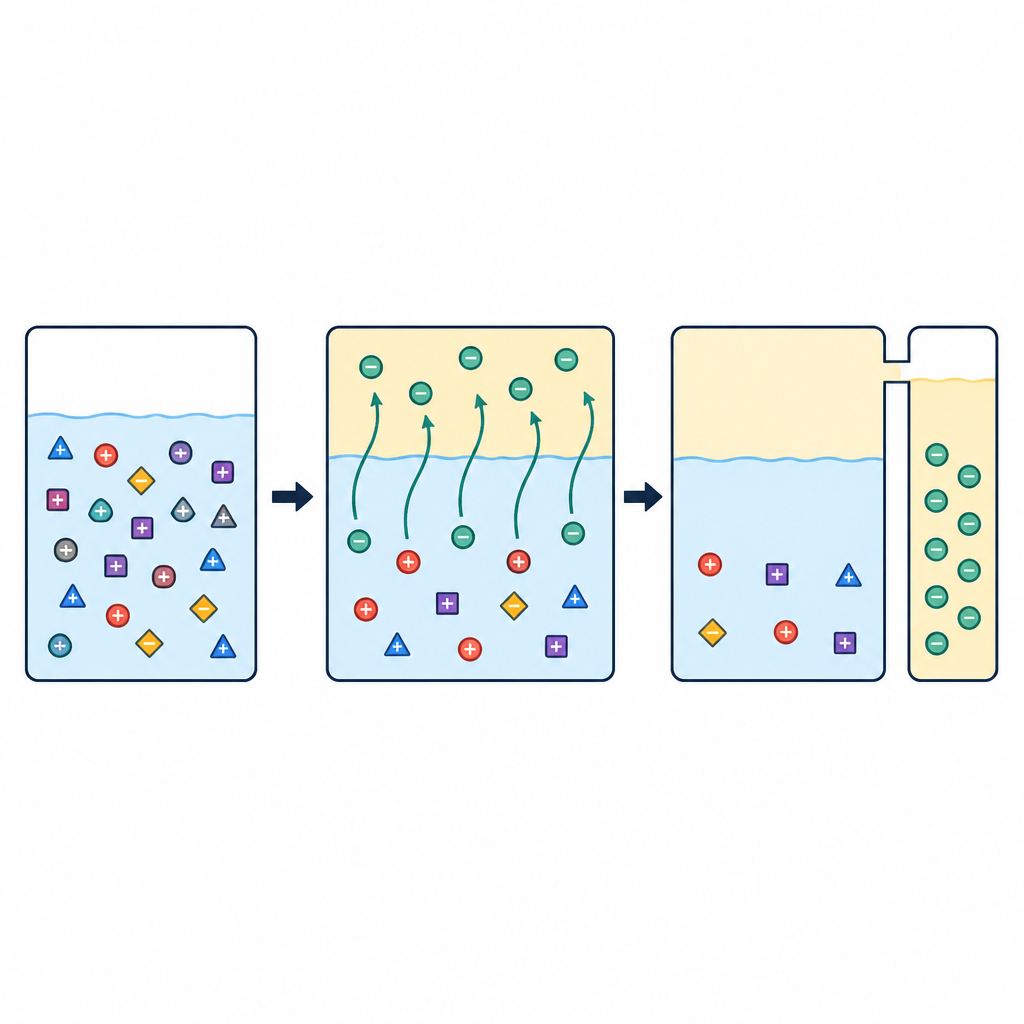
The researchers designed a special organic liquid that acts like a filter at the molecular level. It combines three ingredients: iron ions, a common industrial solvent called tributyl phosphate, and an acidic helper molecule known as P507. When this organic phase is mixed with the acidic battery leachate, lithium ions move into the organic liquid much more readily than nickel, cobalt, or manganese. The team carefully tuned the ratio between P507 and iron so that the liquid could both pick up lithium from the leachate and later let it go again into clean water, keeping the iron locked inside the organic phase for reuse.
Step by step: from mixed soup to clean lithium
Two slightly different process layouts were tested. In the first, the organic liquid was loaded with lithium directly from the battery leachate, and P507 was added afterward to stabilize the system. In the second, iron was first preloaded into the organic phase using a simple stock solution, and only then was this liquid brought into contact with the real leachate. Both routes were followed by three key stages: scrubbing to wash away trace impurities, stripping to move lithium from the organic liquid into water, and regeneration to prepare the organic phase for another cycle. With an appropriate mixing ratio between organic and aqueous phases and a P507 to iron ratio of about 1.5 to 1.7, the process extracted more than 90 percent of lithium over several contact stages while leaving nickel and cobalt almost completely behind in the original solution.
Keeping the system stable and reusable
The team used infrared and ultraviolet visible light measurements to confirm how iron and the organic molecules interact inside the liquid filter. These tests showed that iron stays bound in a form that remains in the organic phase even when the chloride level drops during water stripping. As a result, the same solvent can be reused for multiple cycles without significant loss of iron or extraction performance. Over six repeated runs, single stage lithium extraction stayed near 65 percent, and the final lithium rich water stream contained 11 to 14 grams of lithium per liter while levels of nickel and cobalt were below detection and manganese was nearly absent.
What this means for cleaner battery recycling
For a non specialist, the key outcome is that lithium can be pulled out early from a harsh, salty battery leach solution using a recyclable liquid filter and only water for the final recovery step. This avoids heating the black mass to high temperatures, cuts down on added acids and bases, and prevents the introduction of extra impurity metals. In an industrial recycling plant, the cleaned leachate could then be treated with existing methods to recover nickel, cobalt, and manganese, while the lithium rich water stream could be turned into battery grade lithium salts. Together, these steps offer a more efficient and potentially more sustainable way to close the loop on lithium in rechargeable batteries.
Citation: Saleem, U., Buvik, V., Bandyopadhyay, S. et al. Selective extraction of lithium from acidic chloride leachates of spent batteries. Sci Rep 16, 14984 (2026). https://doi.org/10.1038/s41598-026-43332-y
Keywords: lithium recycling, battery leachate, solvent extraction, black mass, hydrometallurgy