Clear Sky Science · en
Experimental and computational insights into the CuFeMoO₄ modified CMC microbeads for effective removal of methylene blue from aqueous media
Cleaning Up Brightly Colored Water
Many industries leave behind brilliantly colored wastewater, and those bright blues and reds can be bad news for rivers and for human health. This study explores a new, low-cost material that can rapidly strip a common blue dye out of water while also helping release clean hydrogen gas. The researchers combine lab experiments with computer simulations to understand exactly how their material works, paving the way for smarter, greener treatment of industrial wastewater.
A New Tiny Bead for Dirty Water
The heart of the work is a new kind of microscopic bead made by tying together two components: a mixed metal oxide containing copper, iron, and molybdenum (CuFeMoO₄) and a plant-derived thickener called carboxymethyl cellulose (CMC). Using a simple salt as a crosslinker, the team forms nearly spherical microbeads where hard, rod-like metal nanoparticles are wrapped in a soft, biodegradable polymer shell. Tests with several imaging and surface-analysis tools show that these beads have a moderate surface area, nanoscale pores, and a fairly uniform internal structure, all of which help them interact efficiently with pollutants in water. 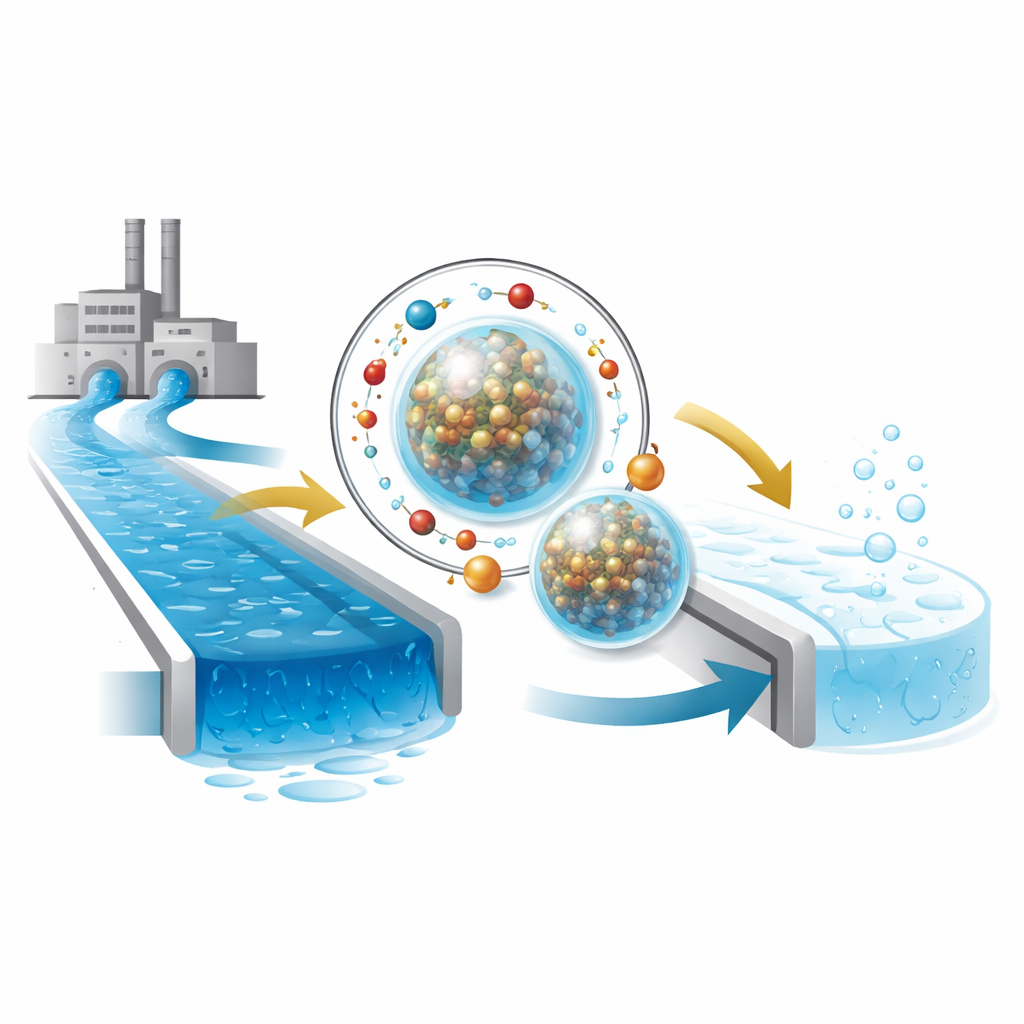
How the Beads Erase Blue Dye
To test the beads, the researchers focused on methylene blue, a bright dye widely used in textiles, inks, cosmetics, and medicine. Rather than simply sticking the dye to a surface, they use a chemical helper, sodium borohydride, which can donate electrons and hydrogen atoms. On its own, this helper barely affects the dye, and the beads alone do not remove the color either. But when both are present together with the new microbeads, the blue color fades almost completely within 45 minutes. The dye is chemically transformed into a colorless form rather than just moved from water to a solid, which makes the cleanup more permanent.
Tuning the Conditions for Best Performance
The team systematically varied how much catalyst they used, how concentrated the dye and borohydride were, the acidity (pH), salt content, and temperature. Increasing the bead dose sped up the reaction, because more active surfaces were available. Making the dye solution more concentrated slowed things down, as the limited surface became crowded and partially clogged. Higher borohydride levels boosted the speed by supplying more electrons. The reaction worked best at nearly neutral pH around 6: under strongly acidic conditions the bead surface repelled the positively charged dye, while in strongly basic conditions it repelled the negatively charged borohydride. Adding common salt hindered the process, because the salt ions competed with dye and borohydride for the same sites on the bead surface. Higher temperatures generally increased the rate but also revealed that the process needs a certain energy input and is not naturally spontaneous under all conditions.
Inside the Invisible Reaction Path
Laboratory tests with a radical-trapping molecule showed that the reaction does not rely on short-lived, aggressive radicals, but instead on a more orderly flow of electrons across the bead surface. The metals in the bead act like tiny relay stations: copper, iron, and molybdenum ions are reduced by borohydride, then pass electrons to the dye, and finally return to their original state, ready to repeat the cycle. To understand why the dye reacts this way, the authors used modern quantum-chemistry calculations. These simulations map out how electrons are distributed in the dye molecule and identify its most vulnerable spot. They found that the sulfur atom at the center of the dye ring, carrying a positive charge, is the prime target for electron attack, matching the observed pathway where the colored form is reduced to a colorless one. 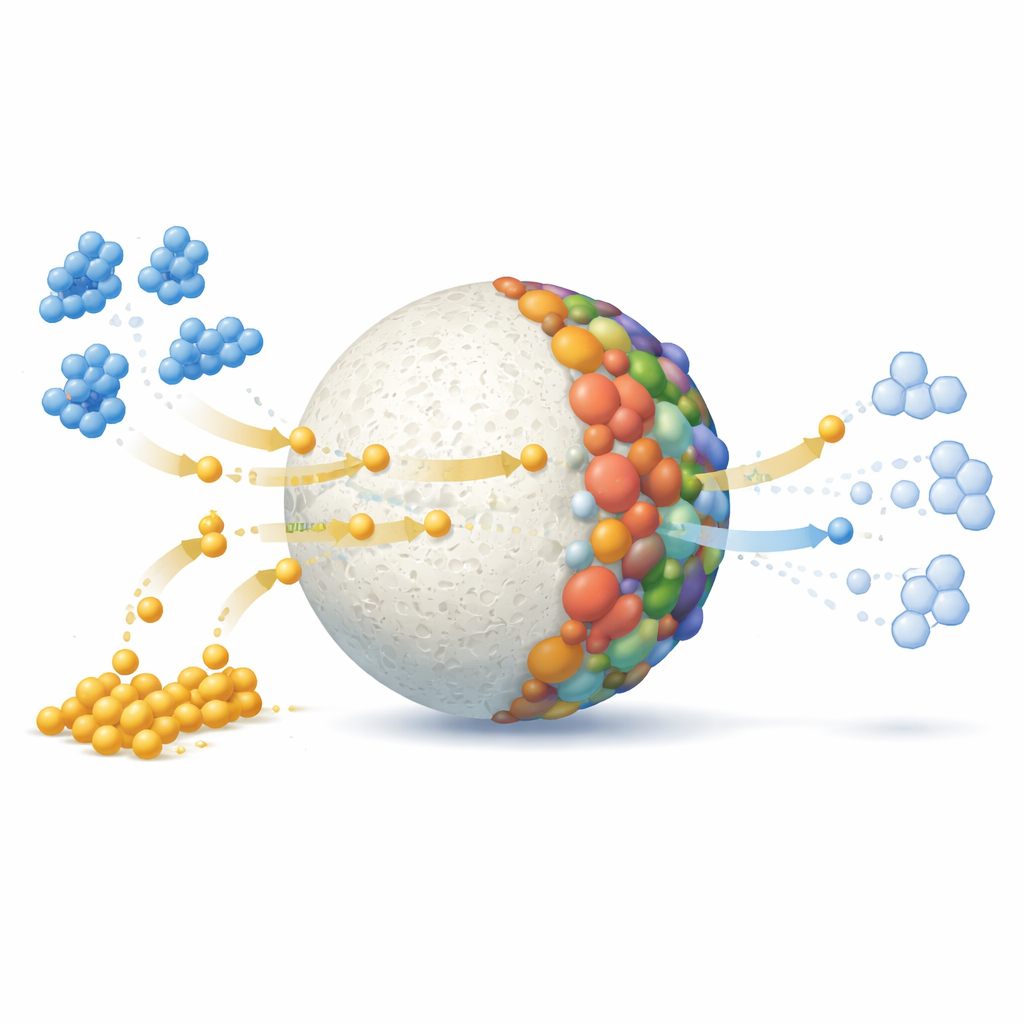
Staying Strong Through Reuse
For any real-world treatment plant, a catalyst is only useful if it can be reused many times. The researchers ran multiple cycles of dye removal, each time filtering, washing, and drying the beads before using them again. The beads kept almost their full power through five runs, only dropping to about half efficiency by the eighth. Microscopy after use showed some roughening and clumping of the nanoparticles inside, but the key elements and overall structure remained intact, explaining the good durability. When they compared their material to many other reported catalysts for methylene blue removal, the new beads stood out with both very high efficiency and a relatively fast reaction.
What This Means for Cleaner Water
Put simply, this work shows that a simple, inexpensive blend of common metals and a biodegradable polymer can very effectively and repeatedly scrub a stubborn dye from water when paired with a mild chemical reducing agent. The experiments and simulations reinforce each other, revealing how electrons move through the system and which part of the dye is attacked. Because the beads are easy to recover and reuse, and because their building blocks are abundant and low-cost, they offer a promising route toward scalable treatment of dyed wastewater streams, helping industries reduce their ecological footprint without relying on rare or precious metals.
Citation: Salem, M.A., Awad, M.K., Sleet, R.K. et al. Experimental and computational insights into the CuFeMoO₄ modified CMC microbeads for effective removal of methylene blue from aqueous media. Sci Rep 16, 12040 (2026). https://doi.org/10.1038/s41598-026-43208-1
Keywords: wastewater treatment, methylene blue, nanocatalyst, carboxymethyl cellulose, density functional theory