Clear Sky Science · en
Development and validation of a machine learning model for individualised meropenem dosing in CRRT patients
Why this matters for patients in intensive care
When people develop life-threatening infections in intensive care, doctors often rely on powerful antibiotics and artificial kidney machines to keep them alive. Yet figuring out the right antibiotic dose for each patient on continuous dialysis-like treatment is surprisingly hard. This study shows how a machine learning–based tool can help doctors choose safer, more effective doses of the broad‑spectrum antibiotic meropenem for some of the sickest patients in the hospital.
The dosing dilemma in fragile kidneys
Sepsis, a severe whole‑body infection, frequently damages the kidneys so badly that patients need continuous renal replacement therapy (CRRT)—a form of slow, around‑the‑clock dialysis. Meropenem is commonly used to treat these infections, but its behavior in the body changes dramatically in critical illness. Fluid shifts, failing organs, and the CRRT machine itself all alter how quickly the drug is cleared. Too little meropenem risks treatment failure and antibiotic resistance; too much raises the chance of side effects such as brain and kidney toxicity. Traditional dosing formulas based on average patients often miss the mark for individuals whose physiology is far from normal.
Building a virtual intensive care population
Because large, high‑quality drug studies in such unstable patients are rare, the researchers first created a virtual population of 48,000 simulated CRRT patients using an existing pharmacokinetic model of meropenem. For each simulated patient, they varied age, body weight, kidney function, CRRT settings, meropenem dose, dosing interval, infusion time, and the toughness of the infecting bacteria (summarized as the minimum inhibitory concentration, or MIC). They then calculated whether each dosing plan kept meropenem levels high enough in the blood for the entire dosing period—both at a standard threshold (100% of the time above MIC) and at a very strict one (100% above four times MIC), which is thought to better protect against treatment failure and resistance.
Training machines to predict success
Using these virtual patients, the team trained several kinds of machine learning models to answer a simple yes‑or‑no question: will a particular patient, on a specific CRRT setup and dosing plan, hit the desired drug exposure target? Algorithms such as random forests, gradient boosting, and decision trees learned patterns linking patient features, CRRT clearance, dose size, dosing interval, infusion duration, and MIC to the likelihood of success. Across the simulated test data, most models were highly accurate, with many correctly classifying outcomes in more than 95% of cases. To open the “black box” of machine learning, the researchers used SHAP (Shapley Additive Explanations) to rank which inputs mattered most. Bacterial MIC was the single strongest driver, followed by how often and how much meropenem was given and how efficiently the patient and CRRT machine cleared the drug.
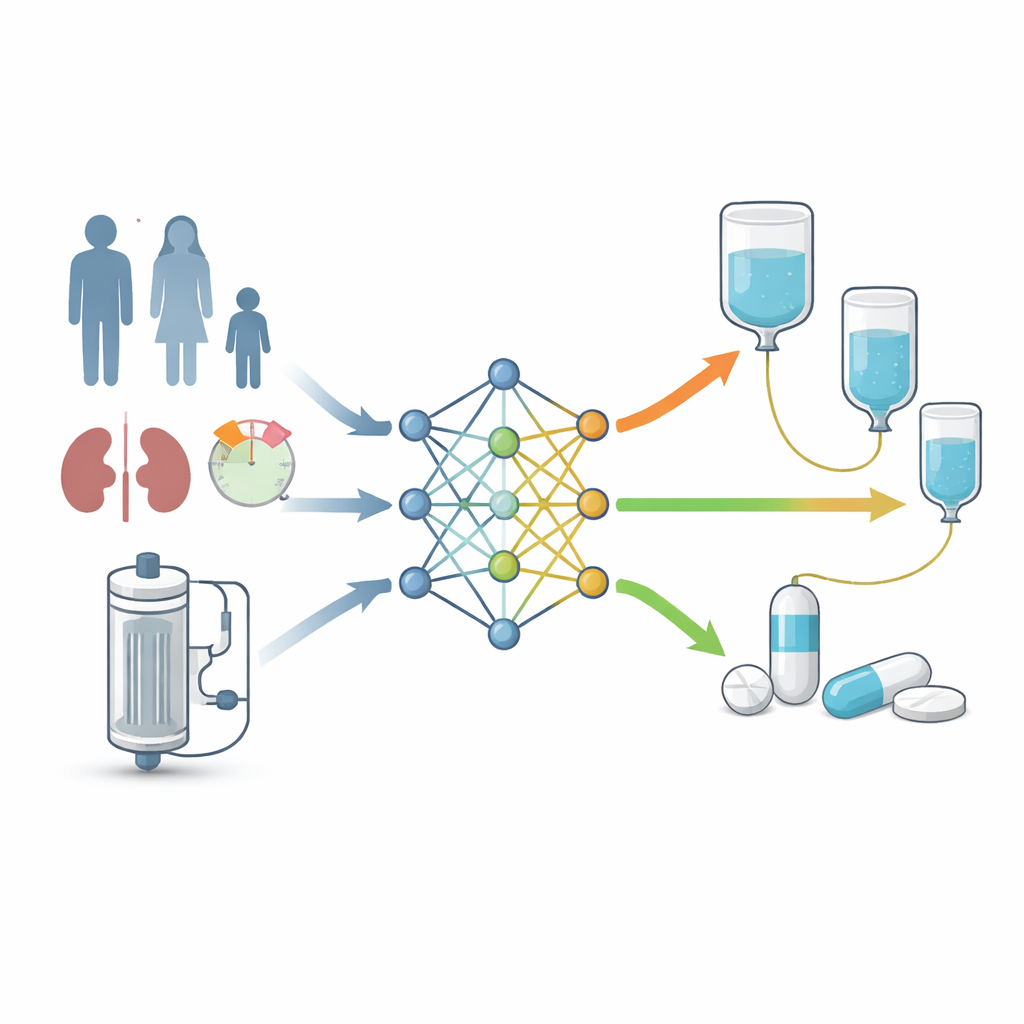
Real‑world testing and a bedside tool
The next step was to see how these models performed in actual patients. The authors drew on data from 78 critically ill individuals on different CRRT systems, compiled from nine published studies. For each patient, a traditional population model reconstructed detailed drug concentration curves, which served as the reference standard for whether the dosing achieved the strict exposure targets across a range of plausible MIC values. When the machine learning models were asked to predict the same outcomes using only patient characteristics, CRRT settings, and dosing information, they generally outperformed the conventional pharmacokinetic approach. In particular, a gradient boosting model achieved higher discrimination for both exposure targets than the established model. This best‑performing model was then embedded in a simple web application, called “MeroDose,” built with the Streamlit framework.
From complex data to clearer choices
At the bedside, the MeroDose tool allows clinicians to enter a patient’s basic information, kidney function, CRRT parameters, proposed meropenem dose and schedule, and an assumed MIC. The system instantly estimates the chance that this regimen will keep drug levels high enough throughout the dosing interval, at both standard and very strict targets. While it does not replace blood‑level monitoring or clinical judgment, it gives a patient‑specific starting point instead of a one‑size‑fits‑all dose. The study concludes that this machine learning–driven support system can meaningfully improve how initial meropenem doses are chosen for CRRT patients, and it calls for prospective trials to confirm that such tools translate into better outcomes and fewer resistant infections.
Citation: Li, D., Qiao, Z., Xiong, X. et al. Development and validation of a machine learning model for individualised meropenem dosing in CRRT patients. Sci Rep 16, 12398 (2026). https://doi.org/10.1038/s41598-026-43012-x
Keywords: sepsis, continuous renal replacement therapy, meropenem dosing, machine learning in medicine, clinical decision support