Clear Sky Science · en
Physicochemical optimization of zinc oxide nanoparticles enhances their antimicrobial and anticancer activities via RmpA, fnbA, cna, and LuxS gene expression suppression
Small Particles, Big Potential
Antibiotic-resistant infections and cancer are two of the most worrying health threats of our time. This study explores whether one material—tiny grains of zinc oxide—could help tackle both problems at once. By shrinking zinc oxide down to the nanoscale and carefully tuning its physical and chemical properties, the researchers show that these particles can not only kill or slow harmful microbes and cancer cells, but also quietly switch off key bacterial “attack” programs even at very low doses.
Why Superbugs Are So Hard to Treat
Many dangerous bacteria have learned to withstand multiple antibiotics, turning routine infections into life-threatening events. These microbes do damage not just because they grow, but because they deploy special tools: surface hooks that help them cling to tissues, protective slime layers called biofilms, and chemical “chatter” systems that coordinate their attacks. The team focused on four such genetic control switches—rmpA, fnbA, cna, and luxS—that help bacteria stick, form biofilms, and communicate. If those switches can be turned down without necessarily killing the bacteria outright, it may be possible to make infections milder and easier to control while putting less evolutionary pressure on microbes to develop resistance.
Building and Testing the Tiny Grains
The researchers created zinc oxide nanoparticles using a straightforward wet chemical recipe in water. A zinc-containing salt and a carbonate salt were gently combined and heated to form an intermediate compound, then baked to yield zinc oxide in the form of minute, nearly spherical particles about 30 billionths of a meter across. They confirmed the size, shape, crystal structure, and surface charge of these particles using standard laboratory tools such as electron microscopy, light absorption measurements, and electrical charge analysis in liquid. The particles were stable in suspension, and brief treatment with ultrasound made them even more evenly dispersed and slightly smaller, an important factor because size and surface charge strongly influence how nanoparticles interact with cells.
Stopping Germs and Tumors
When the team exposed a panel of hospital-related bacteria to these zinc oxide nanoparticles, they observed clear zones where growth was halted, especially for Escherichia coli. The minimum amounts needed to stop growth varied by species, with some bacteria succumbing at relatively low concentrations and others, such as Staphylococcus aureus, requiring higher doses. This reflects differences in cell wall structure, protective layers, and internal defense systems. The particles were also tested against two human cancer cell lines, one from breast tissue (MCF-7) and one from liver tissue (HepG2). In both cases, cancer cell survival dropped sharply as nanoparticle concentration increased, with half the cells dying at around 79 micrograms per milliliter for breast cancer cells and about 151 micrograms per milliliter for liver cancer cells. Microscopy showed treated cells becoming rounded, shrinking, and losing their intact membranes—visual signs of stress and programmed cell death, likely driven by oxidative damage within the cells.
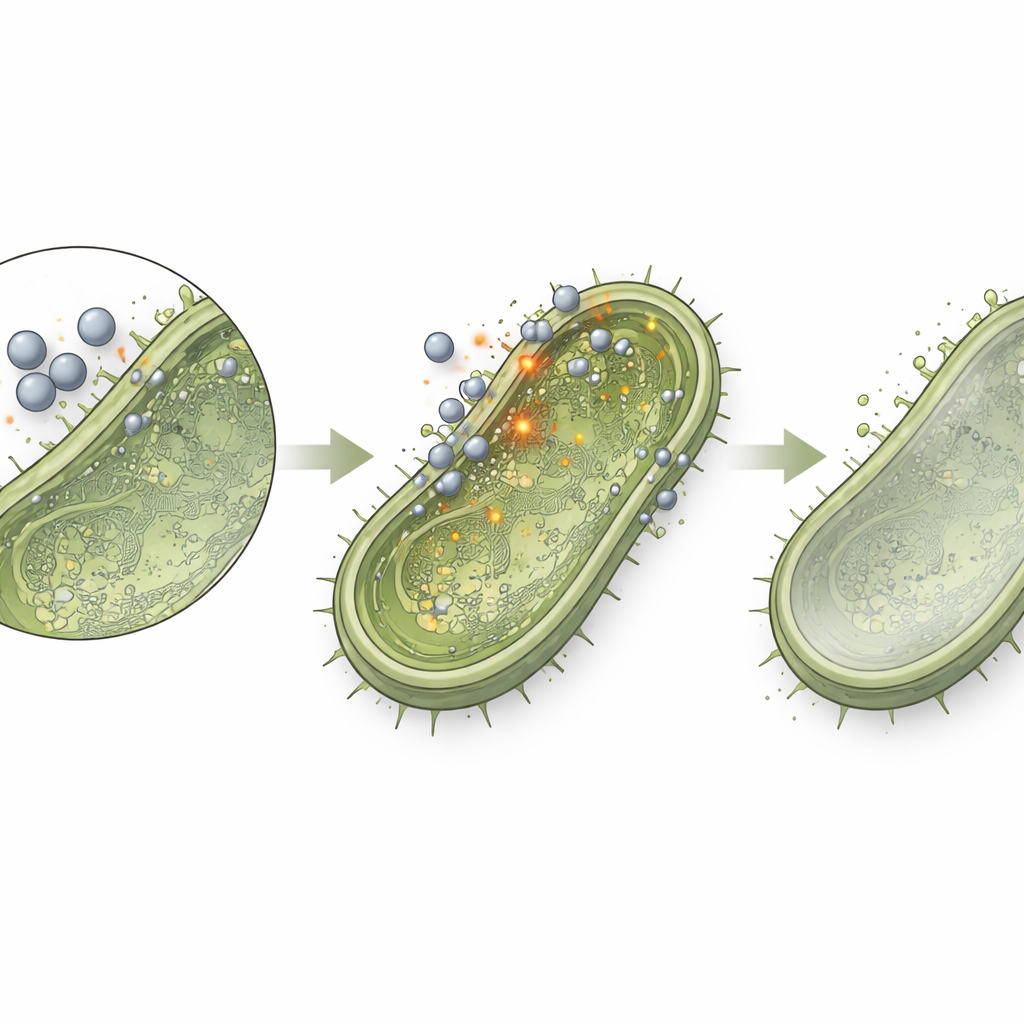
Quietly Disarming Bacteria
Perhaps the most intriguing part of the study came when the researchers deliberately used zinc oxide nanoparticles at levels too low to fully stop bacterial growth. At these “sub-MIC” doses, bacteria remained alive but were exposed to a steady, gentle nanoparticle presence. By measuring the activity of specific genes, the team found that key virulence and communication programs were dialed down. Three genes tied to stickiness and tissue invasion—rmpA in Klebsiella pneumoniae, fnbA in certain staphylococci, and cna in Staphylococcus aureus—dropped to about 60% of their normal expression. The luxS gene, a central player in the chemical signaling system bacteria use to coordinate group behavior and biofilms, fell to about 80% of normal. This means that even when the nanoparticles are not killing the microbes outright, they are making them less organized, less invasive, and potentially easier for the immune system and existing drugs to handle.
What This Could Mean for Future Treatments
Taken together, the findings suggest that carefully engineered zinc oxide nanoparticles could serve as a flexible new tool in medicine. At higher doses, they can directly damage and kill a range of dangerous bacteria and show strong, dose-dependent toxicity toward cancer cells. At lower, non-lethal doses, they act more like molecular saboteurs, weakening bacterial virulence genes and disrupting the communication systems that make infections so hard to treat. For non-specialists, the key message is that tiny, well-designed particles can be tuned not just to attack cells, but to subtly reshape how harmful microbes behave. This dual action—killing when needed and disarming when full killing is not required—may help extend the life of existing antibiotics and open new avenues for gentler, more targeted cancer and infection therapies.
Citation: khedr, M., Emam, A.N., Dora, M.S. et al. Physicochemical optimization of zinc oxide nanoparticles enhances their antimicrobial and anticancer activities via RmpA, fnbA, cna, and LuxS gene expression suppression. Sci Rep 16, 11367 (2026). https://doi.org/10.1038/s41598-026-42733-3
Keywords: zinc oxide nanoparticles, antibiotic resistance, nanomedicine, anticancer therapy, bacterial virulence