Clear Sky Science · en
Green synthesis of 4,4′-diaminotriarylmethane derivatives using a recyclable magnetic acidic nano catalyst
Colorful Molecules with a Hidden Role
Many everyday products—from cancer drugs to textile dyes—rely on a family of colorful molecules called triarylmethanes. Making these compounds usually involves harsh chemicals, high energy use, and difficult waste disposal. This paper describes a cleaner, reusable "magnetic" catalyst that helps chemists build important triarylmethane building blocks more gently, using a simple alcohol solvent and moderate heat. The work sits at the crossroads of medicine, materials science, and green chemistry, showing how smarter tools can reduce pollution without sacrificing performance.
Why These Bright Molecules Matter
Triarylmethanes are more than just pretty colors. Their chemical backbone appears in a range of medicines, including anticancer, antifungal, antibiotic, and anticonvulsant drugs, as well as in many industrial dyes used to color fibers, inks, and optical components. Because the same basic skeleton can be tuned to interact with biological targets or absorb light in precise ways, chemists need reliable routes to make many different versions. Traditional methods can deliver good amounts of product, but they often rely on corrosive acids, toxic solvents, or intense heating. This tension—between our appetite for advanced molecules and the environmental cost of making them—motivates the search for greener methods.
Building a Tiny Magnetic Helper
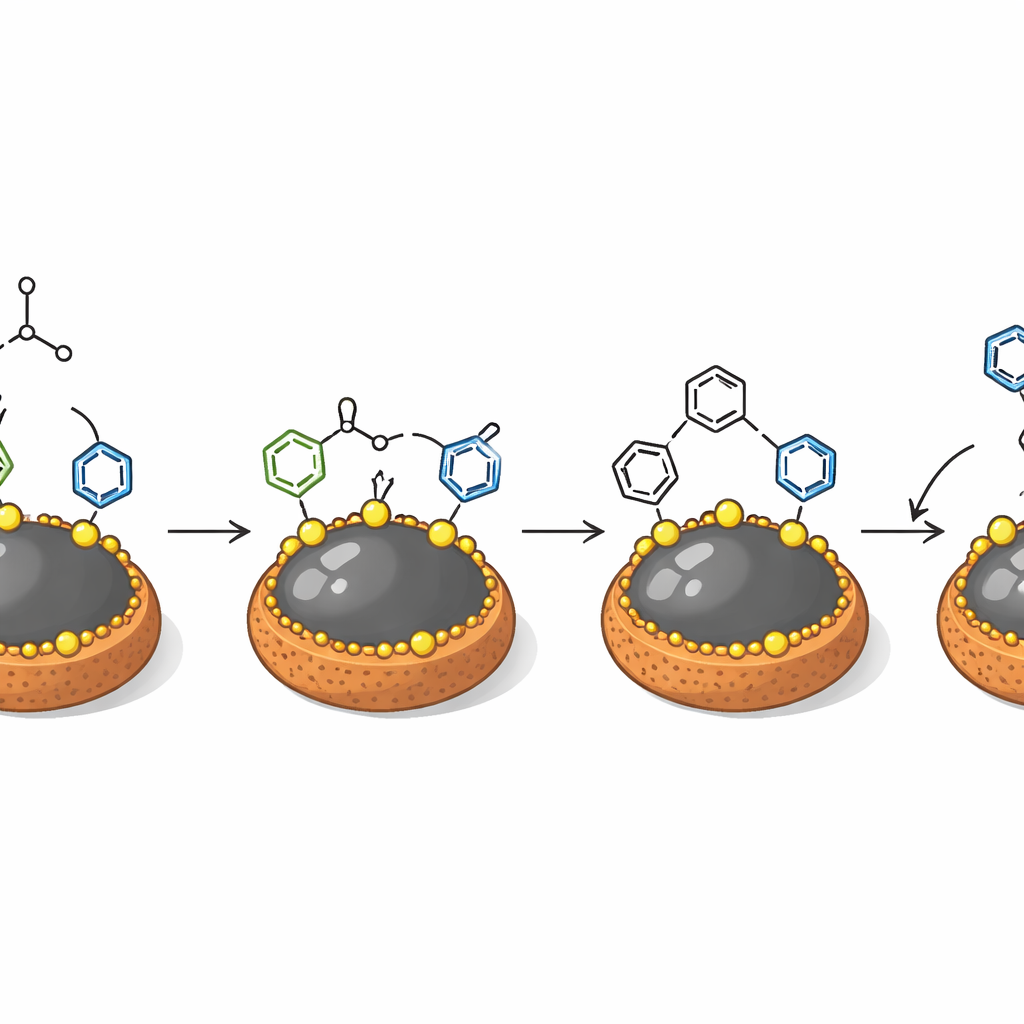
The researchers designed a new solid catalyst built around iron oxide nanoparticles, which are naturally magnetic. They coated this iron core with a small natural amino acid, L-proline, and then added strongly acidic sulfonic groups to create a material denoted Fe3O4@L-proline@SO3H. Under an electron microscope, these particles appear as nearly spherical grains with a thin organic shell surrounding a dense core. Tests using X-ray diffraction, surface area measurements, and magnetic studies showed that the iron oxide core keeps its crystal structure and strong magnetic response even after functionalization. At the same time, the outer shell provides many acidic sites and pores where reactions can take place, marrying the reactivity of a liquid acid with the easy handling of a solid powder.
One-Pot Chemistry in a Greener Solvent
With the catalyst in hand, the team explored a simple recipe: mix an aromatic aldehyde, a dimethylaniline derivative, ethanol, and the magnetic catalyst, then heat to about 80 °C. Under these mild conditions, the starting materials combine in a single step to form 4,4′-diaminotriarylmethane derivatives in respectable to excellent yields, typically between 73% and 92%. Systematic tests varied the amount of catalyst, solvent, temperature, and reaction time. Ethanol—a relatively benign, biodegradable solvent—emerged as the best medium, and only a modest amount of catalyst (0.2 g in their standard test) was needed. Control experiments showed that bare iron oxide particles were far less effective, highlighting the importance of the carefully engineered outer layer in speeding up bond formation.
How the Catalyst Does Its Job
At the molecular level, the acidic groups on the nanoparticle surface activate the aldehyde, making its central carbon more eager to bond. Dimethylaniline molecules then attack this activated site stepwise, ultimately installing two such units around the central carbon and forming the triarylmethane core. Because the reaction happens at the catalyst surface, the material behaves like a true solid helper: when the mixture is hot-filtered to remove the particles halfway through, product formation nearly stops. The particles can then be pulled out of the reaction with a simple magnet, washed, and reused. Over at least six cycles, the catalyst retains most of its performance, and a brief acid wash can restore surface area lost to pore blockage.
Reusability and Real-World Impact
Compared with many earlier methods, this magnetic catalyst offers a compelling balance of efficiency and sustainability. It avoids highly corrosive liquid acids and problematic solvents, uses moderate temperatures, and sharply reduces waste by allowing the catalyst to be recycled multiple times. The team also quantified how many reactions each catalytic site can carry out and how quickly, finding consistent activity across a broad range of products. In head-to-head comparisons with other catalyst systems from the literature, the new material stacks up well in yield while adding the crucial benefits of easy magnetic separation and reuse.
Cleaner Routes to Color and Medicine
In everyday terms, this work shows that we can still make the bright molecules needed for medicines and dyes while treading more lightly on the environment. By shrinking the active chemistry onto magnetic nanoparticles, the authors created a solid helper that can be stirred into a simple ethanol mixture, drive the formation of valuable triarylmethane building blocks, and then be plucked out with a magnet and used again. This kind of smart, recyclable catalyst points the way toward chemical manufacturing that keeps its useful colors and therapeutic power, but with a much smaller environmental footprint.
Citation: Karamifar, S., Behbahani, F.K. & Keshmirizadeh, E. Green synthesis of 4,4′-diaminotriarylmethane derivatives using a recyclable magnetic acidic nano catalyst. Sci Rep 16, 12800 (2026). https://doi.org/10.1038/s41598-026-42570-4
Keywords: green chemistry, magnetic nanoparticle catalyst, triarylmethane dyes, recyclable catalysis, eco-friendly synthesis