Clear Sky Science · en
Multifunctional PVC-based metal oxide/graphene composites for high-performance DSSC counter electrodes
Turning Common Plastic into a Solar Power Helper
Solar cells that mimic photosynthesis using colorful dyes are a promising route to low-cost, flexible clean energy. But one of their key parts, the counter electrode, is usually made from pricey platinum. This study shows how an everyday plastic, PVC—the same material found in pipes and cables—can be upgraded with tiny particles of zinc oxide and sheets of graphene to almost match platinum’s performance, potentially cutting costs and giving old plastics a valuable second life in solar technology.
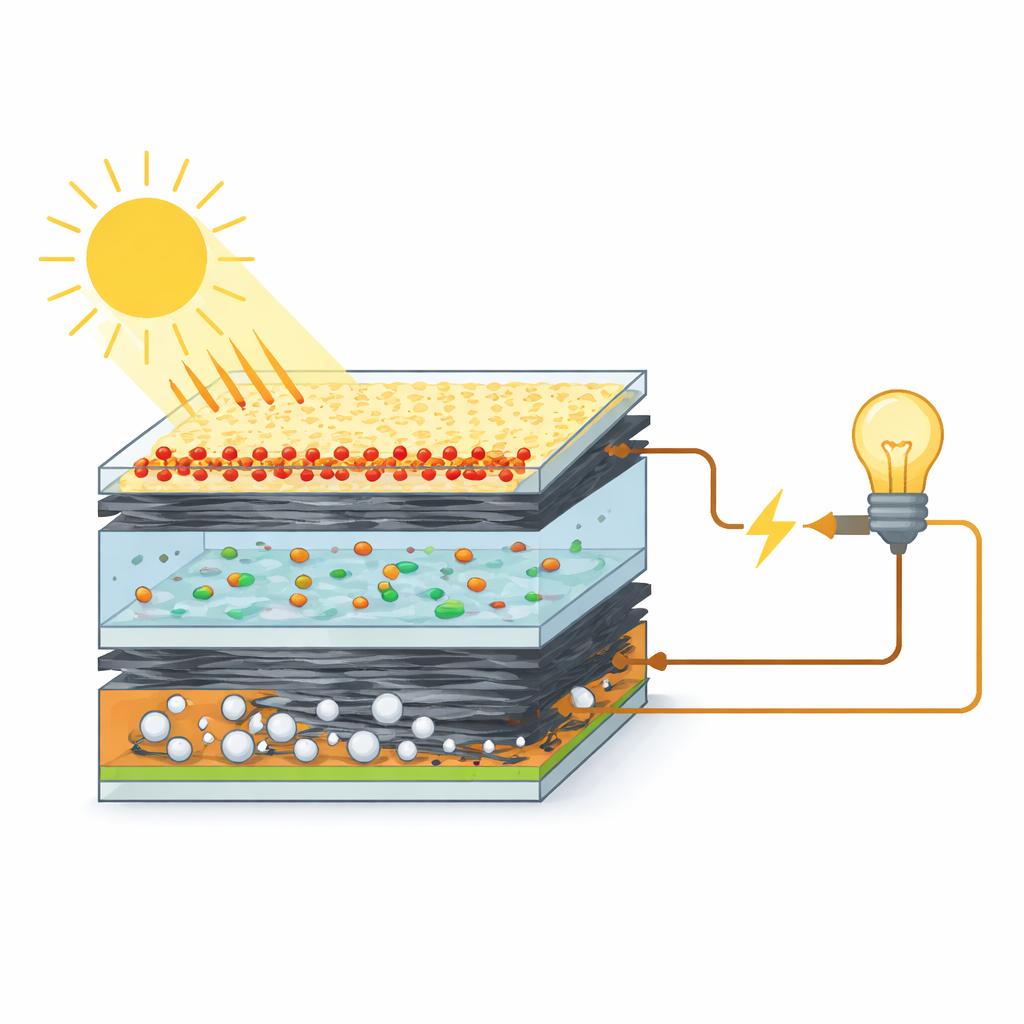
Why the Back Contact of a Solar Cell Matters
Dye-sensitized solar cells work a bit like an artificial leaf. Light excites dye molecules on a semiconductor layer, creating electrons that flow through an external circuit. On the opposite side of the cell sits the counter electrode, which completes the loop by helping a liquid electrolyte shuffle charges back to the dye. If this back contact is slow or resistive, electrons pile up, energy is wasted as heat, and the overall efficiency drops. Platinum does this job extremely well, but it is expensive and scarce, so researchers worldwide are hunting for cheaper, abundant materials that can move charges just as quickly.
Designing a Better Electrode on the Computer First
The team began not in the lab, but on the computer, using quantum-level calculations to predict how different metal oxides would behave when mixed into PVC. They screened several candidates—such as titanium oxide, nickel oxide, and zinc oxide—by probing how easily electrons could move through the resulting mixtures, how stable they would be, and how strongly they might interact with their surroundings. Zinc oxide stood out: it narrowed PVC’s electronic "gap," making it easier for electrons to move, and increased the material’s tendency to respond to electric fields. These changes suggested that PVC loaded with zinc oxide could become far more conductive and reactive than the plain plastic, a promising sign for its role in a solar cell.
Adding Graphene for a Fast Lane of Electrons
Building on the zinc oxide results, the researchers then explored what would happen if they wove graphene—single-atom-thick carbon sheets—into the same PVC blend. Their calculations predicted that adding graphene would dramatically shrink the energy barrier for electron flow, turning the composite into a highly responsive and conductive network. In this design, PVC acts as a flexible host, zinc oxide nanoparticles provide catalytic "hot spots" where chemical reactions in the electrolyte can proceed quickly, and graphene forms long-range expressways for electrons. Together, these ingredients create a material architecture in which charges can move with much less resistance than in PVC alone.
From Simulation to Real Solar Cells
To test these ideas, the team synthesized zinc oxide nanoparticles, mixed them with PVC and graphene, and deposited thin films onto conducting glass to serve as counter electrodes. Microscopy and surface measurements revealed that adding zinc oxide and graphene carved out larger, more connected pores and made the surface rougher, increasing the contact area with the liquid electrolyte. Electrical tests showed that the best blend, containing both zinc oxide and graphene, reached a conductivity of 66 S/m—over three times that of pure PVC—and the largest average pore size. When assembled into full dye-sensitized solar cells, this composite delivered a power conversion efficiency of about 7.5%, compared with 4.7% for plain PVC and only slightly below a platinum-based reference cell.
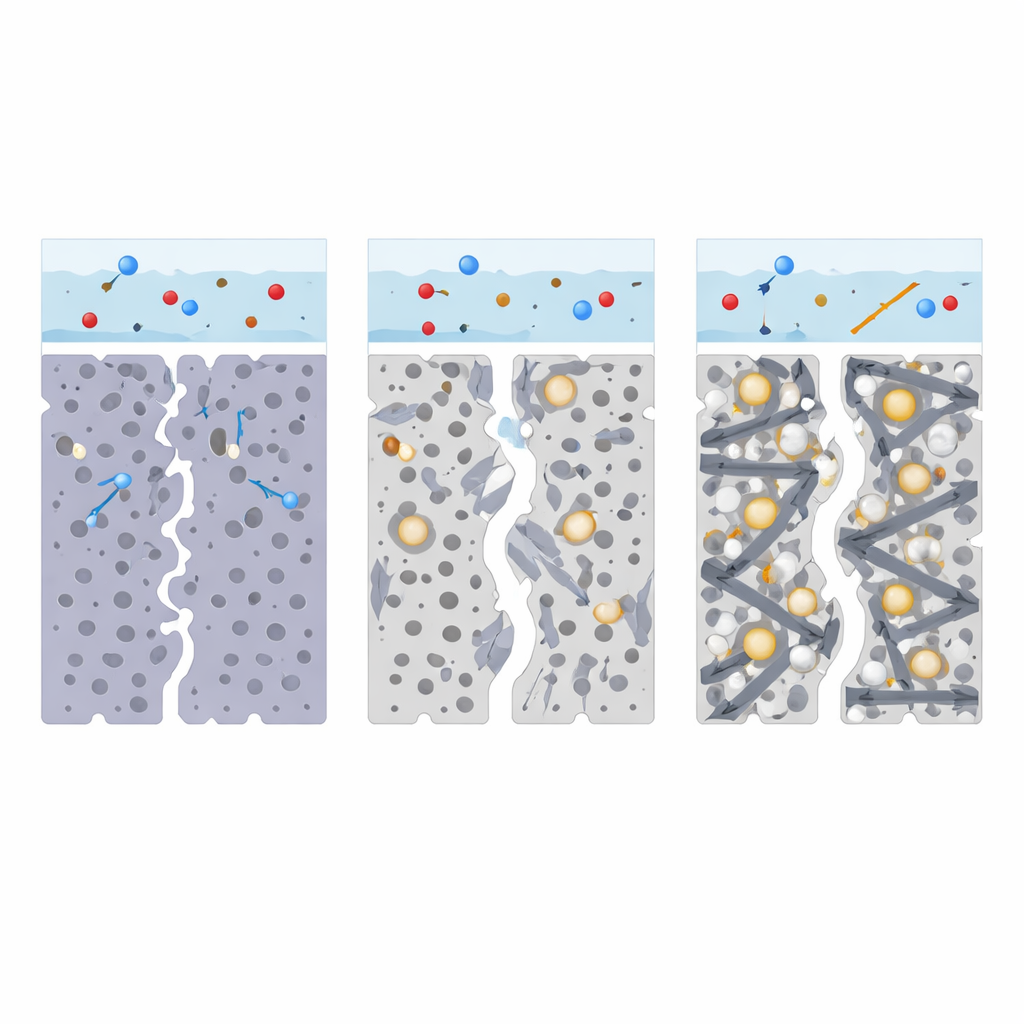
How the New Material Speeds Up Charge Flow
Electrochemical measurements provided a closer look at why the new composite works so well. In cells with plain PVC electrodes, the resistance to charge transfer at the interface with the electrolyte was high, indicating sluggish reactions and frequent charge bottlenecks. Introducing zinc oxide or graphene individually improved some aspects—zinc oxide offered more active reaction sites, while graphene lowered electrical resistance—but neither solved all the problems on its own. The combined PVC/zinc oxide/graphene electrode, however, showed a sharply reduced interfacial resistance, approaching that of platinum. This means electrons can move swiftly through the graphene network, reach zinc oxide catalytic spots with ease, and drive the key redox reactions in the electrolyte efficiently, boosting current and stabilizing the device’s output.
What This Means for Future Solar Technologies
For non-specialists, the main takeaway is that a widely used, low-cost plastic can be transformed—by sprinkling in carefully chosen nanoparticles and carbon sheets—into a smart, highly active component for solar cells. The resulting counter electrode nearly matches platinum’s performance while relying on abundant materials and a potentially scalable fabrication process. Beyond promising cheaper dye-sensitized solar cells, this work offers a design roadmap: use computer modeling to pick the best additives, then build porous, conductive hybrids in which each ingredient plays a clear role. Such strategies could help turn common plastics from waste into workhorses in the clean-energy transition.
Citation: Ezzat, H.A., Sebak, M.A., Aladim, A.K. et al. Multifunctional PVC-based metal oxide/graphene composites for high-performance DSSC counter electrodes. Sci Rep 16, 9817 (2026). https://doi.org/10.1038/s41598-026-41857-w
Keywords: dye-sensitized solar cells, graphene composites, zinc oxide nanoparticles, polymer nanocomposites, PVC recycling