Clear Sky Science · en
Glutamine promotes acute wound healing by mediating glutamine metabolism and M2 macrophage polarization via the MEK/ERK/SLC1A5 signaling pathway
Why a common nutrient matters for wound healing
Anyone who has dealt with a stubborn cut, surgical incision, or bedsore knows that slow healing can be painful and dangerous. This study explores how glutamine, a widely available dietary amino acid, helps the body repair skin wounds more quickly. By tracking how glutamine is used inside cells and how it shapes the behavior of immune cells, the researchers point to a signaling route that could be targeted to speed up recovery from acute injuries.
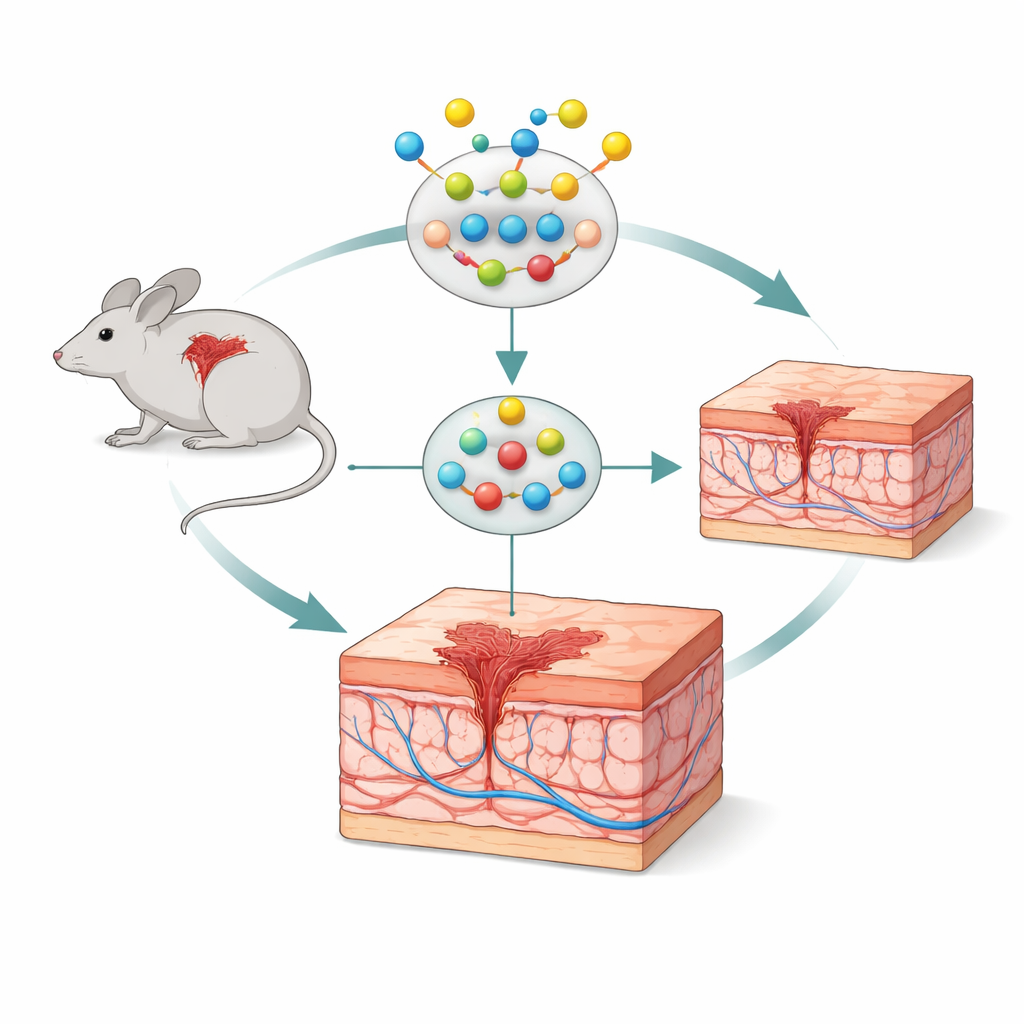
How skin normally mends itself
Skin repair is not a simple “close and done” event. After injury, blood clots form to seal the wound, then immune cells swarm in to clear debris and microbes. Over the next days, new skin cells migrate in, blood vessels sprout to feed the area, and supporting tissue is rebuilt. Finally, the new tissue is remodeled to restore strength, sometimes leaving a scar. A key player throughout this sequence is the macrophage, an immune cell that can switch between a damage-fighting “M1” mode and a repair-focused “M2” mode. A healthy transition from M1 to M2 is essential: too much early inflammation slows healing, while timely M2 activity calms inflammation and supports regrowth of vessels and connective tissue.
Glutamine as fuel and switch for repair
Glutamine is the most abundant amino acid in the body and normally produced by muscles and other tissues. Under stress, such as trauma or infection, internal supplies can fall short, and extra glutamine from the diet becomes important. Once taken up into cells through a transporter protein called SLC1A5, glutamine is broken down into metabolites that feed cellular energy pathways and help counter oxidative stress. It also shapes immune responses: specific breakdown products nudge macrophages away from an inflammatory profile toward the M2 repair state. Earlier clinical work showed that patients with severe infections often have low glutamine levels and can benefit from supplementation, but the exact mechanisms in wound healing were not fully mapped.
Testing glutamine’s role in mouse skin wounds
To dissect this, the authors created standardized full-thickness skin wounds on the backs of mice and divided them into groups. Some received daily oral glutamine, while others received glutamine combined with drugs that blocked key parts of a signaling route known as the MEK/ERK/SLC1A5 axis, or an enzyme that processes glutamine. Over two weeks, the team photographed wounds, examined tissue structure under the microscope, measured blood vessel growth and fibroblast activity, and tracked inflammatory and repair-related molecules in the blood. They also used protein and fluorescence assays to see how strongly the signaling proteins MEK and ERK and the glutamine transporter SLC1A5 were switched on in the healing skin.
What the experiments revealed inside the wound
Mice receiving glutamine alone healed faster, with smaller wound areas and more organized layers of skin, connective tissue, and muscle than untreated wounded mice. Their skin showed stronger signals of activated MEK and ERK, higher levels of the SLC1A5 transporter, and more markers of new blood vessels and contractile fibroblasts that help pull the wound closed. Blood tests showed that glutamine boosted key metabolic byproducts, including α-ketoglutarate and a sugar-linked molecule (UDP-GlcNAc), indicating that glutamine-fueled energy and building-block pathways were running at high gear. At the immune level, glutamine shifted macrophages toward the M2 repair type, reduced pro-inflammatory messengers such as IL-1β and TNF-α, and increased anti-inflammatory and pro-healing factors like IL-10, TGF-β, and VEGF. When any step of the MEK/ERK/SLC1A5 or glutamine-processing machinery was chemically blocked, these benefits largely disappeared: wounds closed more slowly, tissue remained disorganized, vessel growth weakened, metabolic markers dropped, and macrophages moved back toward a more inflammatory profile.
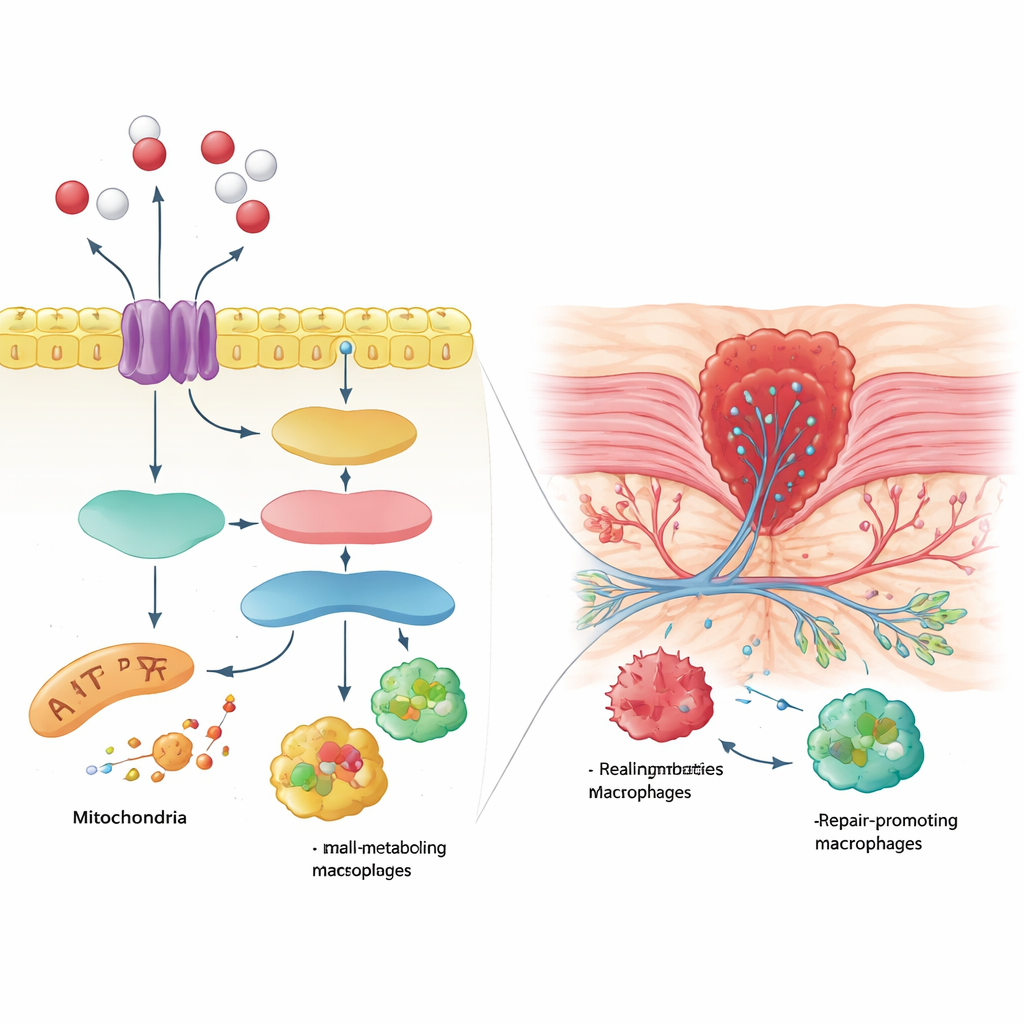
Why this pathway could guide new treatments
Taken together, the results show that glutamine does much more than simply feed cells during healing. By activating MEK/ERK signaling and boosting the SLC1A5 transporter, it rewires metabolism inside immune and tissue cells, tilting macrophages toward a calming, rebuilding mode and supplying energy and raw materials for new vessels and connective tissue. While the work was done in mice and the exact order of molecular events still needs clarification, it highlights a specific signaling and metabolic route that might be harnessed in future therapies. In practical terms, the study suggests that carefully targeted use of glutamine, or drugs that mimic its effects along this pathway, could one day help clinicians better manage acute skin wounds and perhaps reduce complications in high-risk patients.
Citation: Shi, Y., Pan, M., Chen, X. et al. Glutamine promotes acute wound healing by mediating glutamine metabolism and M2 macrophage polarization via the MEK/ERK/SLC1A5 signaling pathway. Sci Rep 16, 14241 (2026). https://doi.org/10.1038/s41598-026-41545-9
Keywords: wound healing, glutamine, macrophages, skin repair, immune metabolism