Clear Sky Science · en
A hybrid pipeline for carotid artery segmentation using YOLOv11n and contour models
Why this matters for stroke prevention
Strokes often strike without warning, yet many begin silently in the neck, where the carotid arteries carry blood to the brain. Doctors can spot early damage in these arteries using ultrasound, a quick and painless scan, but carefully tracing the blood channel by hand on every image is slow and subjective. This study introduces a fully automatic computer method that can rapidly outline the carotid artery in common ultrasound views, even on modest hardware, aiming to make early stroke risk assessment faster, more reliable, and easier to deploy in everyday clinics. 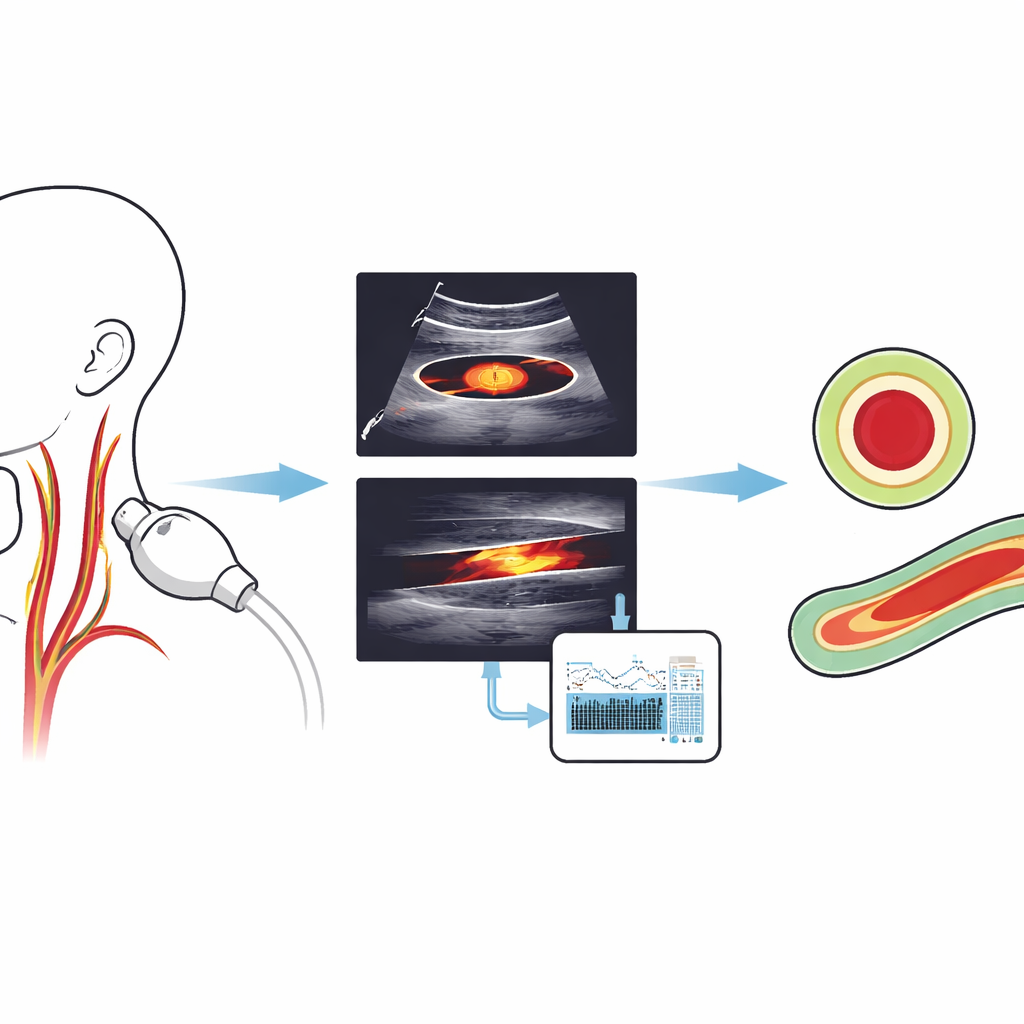
Hidden dangers in neck blood vessels
Carotid arteries can narrow as fatty deposits build up in their walls, restricting blood flow to the brain and raising the risk of stroke. Because symptoms often appear only after severe damage, regular screening of at-risk people—such as older adults, smokers, and patients with diabetes or heart disease—is vital. Ultrasound imaging is ideal for such screening: it is inexpensive, safe, and widely available. However, the images are noisy and low in contrast, and nearby structures like the jugular vein can look deceptively similar to the artery. As a result, drawing the exact inner boundary of the vessel—the “lumen”—remains a difficult and time‑consuming task for both humans and machines.
A three‑stage digital assistant for doctors
The authors designed a hybrid pipeline that mimics how a careful technician would analyze ultrasound images but does so automatically. First, a compact deep‑learning detector, a modern version of the YOLO (“You Only Look Once”) object‑detection family, searches each image and draws a box that tightly encloses the carotid artery lumen, in both cross‑section (a circular view) and lengthwise (a long tube‑like view). This step focuses attention on the right region of the neck and proved extremely reliable, with the model almost never missing the vessel in the test data.
Next, for cross‑section images, the system improves the quality of the ultrasound within that box through a carefully tuned sequence of filters. It smooths away the grainy speckle pattern while protecting edges, boosts local contrast so the artery wall stands out, adjusts brightness, denoises again on a broader scale, and finally sharpens the boundary. These operations reshape the “energy landscape” seen by the following contour‑tracing algorithm, making the true lumen edge the most attractive path for the curve to follow rather than random noise. 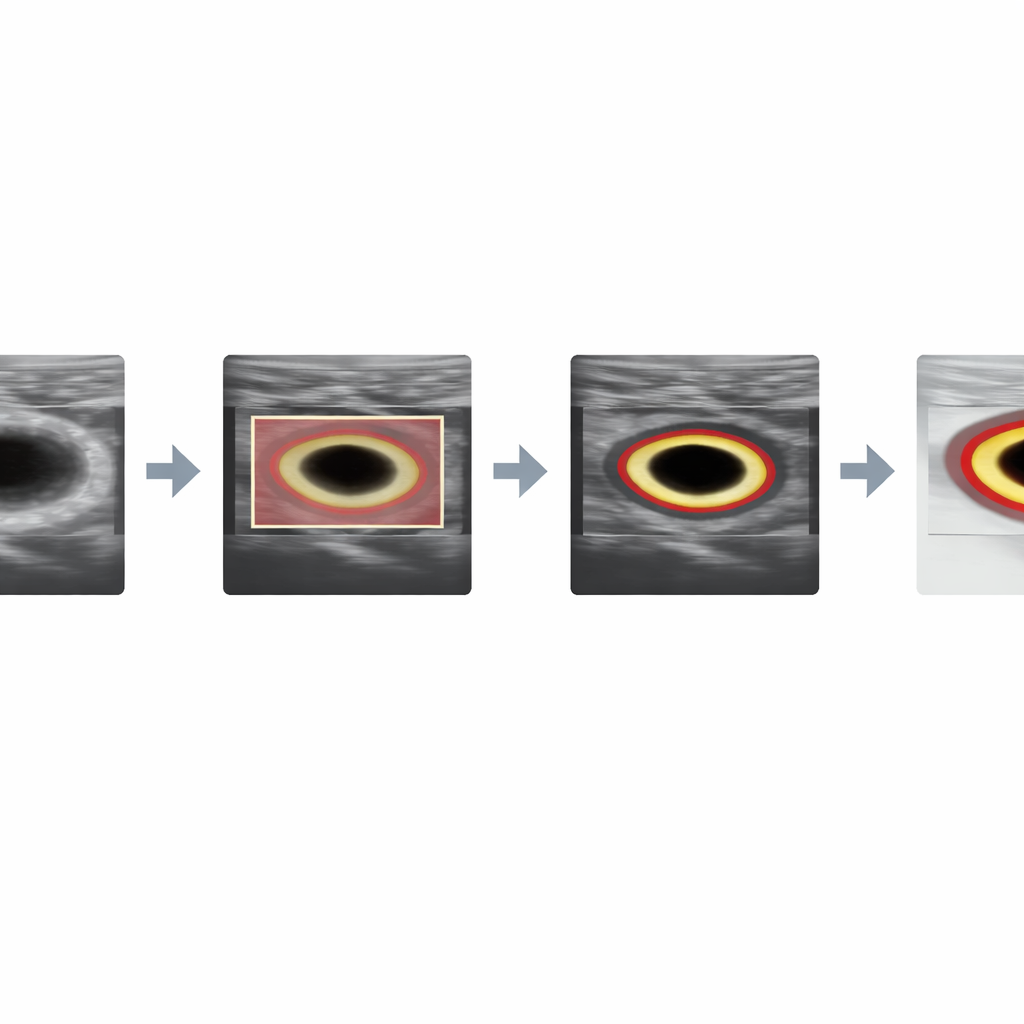
Smart contours that lock onto the right vessel
With the cross‑section images cleaned up, a classic active contour model—sometimes called a “snake”—starts from the detector’s box and gradually slides toward the nearest strong boundary. Because the preprocessing has amplified the true vessel edge and reduced distracting artifacts, the snake settles neatly around the lumen. For the lengthwise images, the challenge is different: the detector’s original box can include both the carotid artery and the jugular vein, which look similarly dark because they are filled with blood. To avoid this confusion, the authors automatically carve out a second, more precise box by scanning within the first one for the darkest, most uniform patch typical of the artery’s interior and then expanding it in a controlled way. This refined region seeds a Chan–Vese contour model, a method that grows a boundary based on how pixel intensities differ inside and outside the curve. Initialized in the right place, it can trace the long, gently curving lumen while ignoring neighboring veins.
How well does it actually work?
The team tested their system on two public datasets: over a thousand cross‑section images and dozens of lengthwise images, each with expert‑drawn lumen outlines for comparison. They used five‑fold cross‑validation and held‑out test sets to avoid overfitting and to mimic performance on unseen patients. For cross‑sections, the method reached a Dice score—a measure of overlap between automatic and expert outlines—of about 91%, with overall accuracy near 99.6%. For lengthwise images, the Dice score rose to roughly 95% and accuracy to about 97.7%. An ablation study, in which parts of the pipeline were deliberately removed, showed that both the initial localization and the tailored preprocessing or refined mask generation were essential; without them, performance dropped sharply or the algorithms even became numerically unstable.
What this means for future care
To a non‑specialist, these numbers mean that a low‑cost computer system can now trace the blood‑carrying channel in carotid ultrasound scans almost as faithfully as a trained specialist, and can do so in under a second for cross‑section views even on a basic laptop. By combining a modern detection network with well‑understood curve‑tracing methods, the approach offers both speed and interpretability. If adopted widely and expanded to analyze plaque as well as lumen, such tools could help turn routine neck ultrasound into a more powerful early‑warning system for stroke, supporting clinicians with consistent, objective measurements wherever ultrasound machines are used.
Citation: Salama, G.M., Safy, M., Hassanin, D.A. et al. A hybrid pipeline for carotid artery segmentation using YOLOv11n and contour models. Sci Rep 16, 9808 (2026). https://doi.org/10.1038/s41598-026-41007-2
Keywords: carotid ultrasound, stroke screening, medical image segmentation, deep learning in medicine, vascular imaging