Clear Sky Science · en
Inhibition efficacy of expired citalopram drug towards the damage of C-steel in hydrochloric acid solution
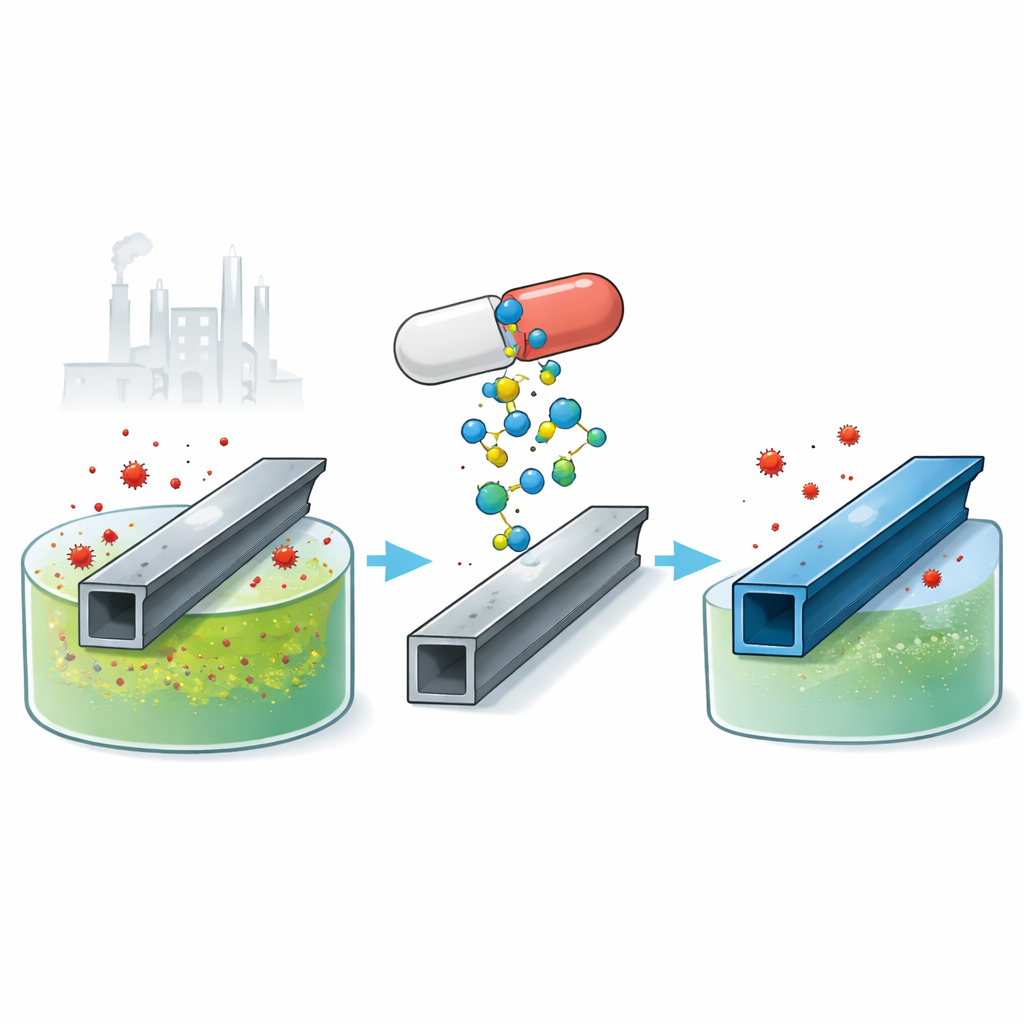
Why an Old Pill Matters for New Metal Problems
Bridges, pipelines, and factory equipment around the world quietly corrode away every day, costing industry billions of dollars and raising safety concerns. To slow this damage, engineers often add special chemicals called inhibitors to harsh cleaning acids used on steel. This study explores a surprising and eco-friendly twist: using an expired antidepressant drug, citalopram, to shield carbon steel from attack in hydrochloric acid. Turning waste medicine into a protective coat for metal could cut costs, reduce chemical waste, and give a second life to drugs that would otherwise be thrown away.
How Acid Eats Steel
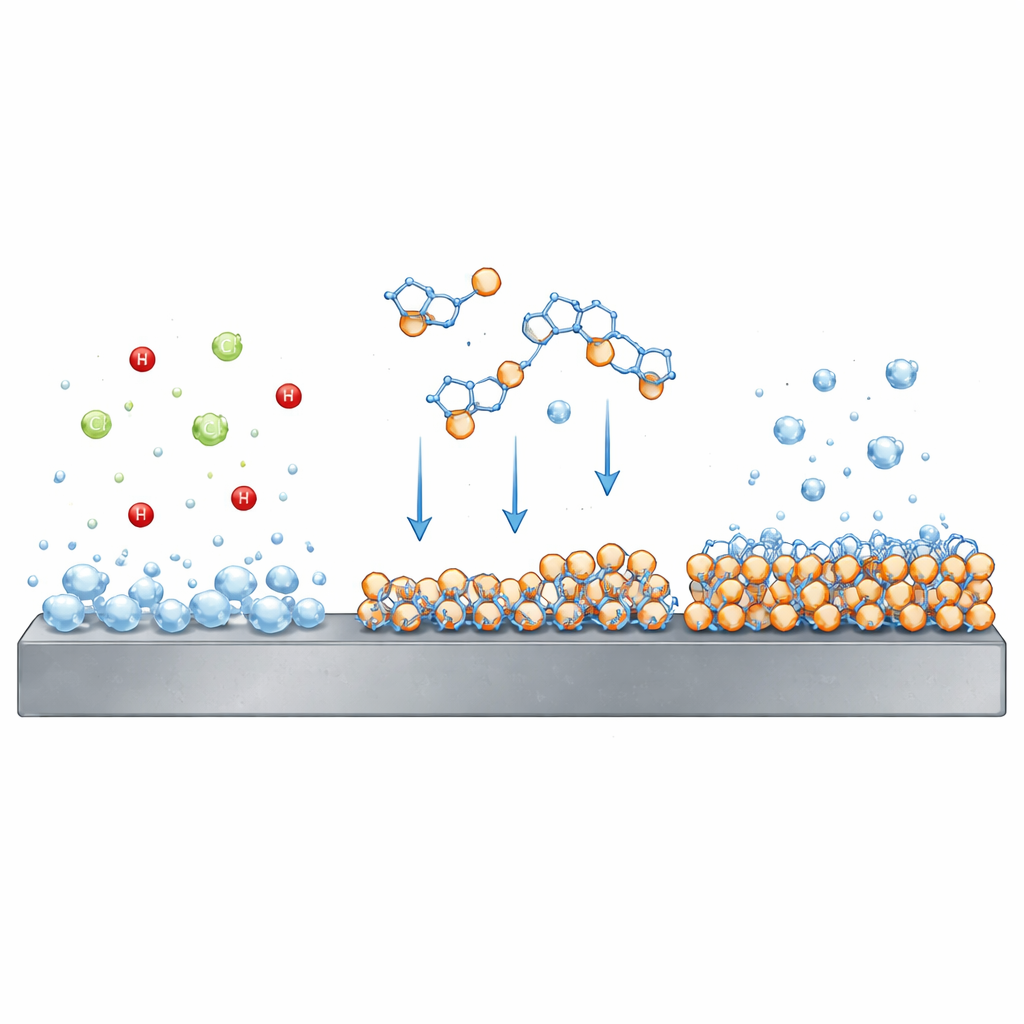
Carbon steel is a workhorse material in oil and gas fields, construction, and many industrial systems because it is strong and affordable. But when it meets strong mineral acids like hydrochloric acid—used to remove rust and scale or to clean and activate metal surfaces—it corrodes rapidly. In simple terms, acid strips metal atoms from the surface and turns them into dissolved ions, while hydrogen gas bubbles off. Over time, this process thins and weakens the steel. To keep equipment safe and long-lasting, industries rely on additives that form a temporary barrier between the steel and the aggressive acid.
Giving Expired Medicine a Second Job
The researchers tested expired citalopram, a common antidepressant, as a protective additive for carbon steel in a hydrochloric acid solution. Rather than being discarded as pharmaceutical waste, the drug was dissolved in the acid at low concentrations and its effect on metal damage was measured. They used several complementary approaches: weighing steel samples before and after long immersion, tracking how much hydrogen gas was released as the steel reacted, and using electrical methods to see how easily corrosion currents could flow. Across these tests, adding small amounts of citalopram cut the corrosion rate dramatically—by over 90% at the highest tested level and room temperature.
How the Invisible Shield Forms
Citalopram molecules have several sites that can interact with metal surfaces and with charged particles in solution. In the acidic liquid, parts of the molecule become positively charged and are drawn toward the negatively charged steel surface, which is enriched with chloride ions from the acid. At the same time, other parts of the molecule can share electrons directly with iron atoms. Together, these effects encourage the drug molecules to settle onto the metal and spread out to form a thin, nearly uniform film. The team’s measurements showed classic signs of such a film: lower corrosion currents, higher resistance to charge moving across the metal–solution boundary, and a reduction in the effective “capacitor” behavior of the surface as water was pushed aside by the organic layer.
What Temperature and Microscopes Reveal
To understand how robust this shield is, the researchers repeated their tests at higher temperatures. As the solution got hotter, the protective effect weakened somewhat, suggesting that some of the adsorbed citalopram molecules detach from the steel, exposing fresh metal to the acid. Calculations of energy and free-energy changes confirmed that the binding is strong but still partly physical in nature—more like sticky attraction than permanent chemical bonding. Electron microscope images backed this up visually: bare steel soaked in acid showed a rough, heavily scarred surface, while steel exposed to acid containing citalopram remained much smoother, with fewer pits and more intact metal. Elemental analysis even detected nitrogen from the drug on the protected steel, direct evidence of the barrier layer.
From Lab Insight to Real-World Impact
Overall, the study shows that expired citalopram can serve as an efficient, low-cost, and relatively green corrosion inhibitor for carbon steel in hydrochloric acid. By forming a closely packed molecular film on the metal surface, the drug slows both the loss of iron and the release of hydrogen gas, even at very small doses. While the protection fades somewhat at higher temperatures, the performance compares well with many other recycled drugs already proposed for the same purpose. For lay readers, the key takeaway is that medicines past their shelf life do not have to go straight to the trash—they may be repurposed as smart coatings that help keep vital steel infrastructure safer for longer, while reducing waste and the need for new synthetic chemicals.
Citation: Saleh, M.G.A., Felaly, R.N., Hawsawi, H. et al. Inhibition efficacy of expired citalopram drug towards the damage of C-steel in hydrochloric acid solution. Sci Rep 16, 11619 (2026). https://doi.org/10.1038/s41598-026-40754-6
Keywords: corrosion inhibition, carbon steel, expired drugs, hydrochloric acid, citalopram