Clear Sky Science · en
Causal relevance of the JAK/STAT pathway for cardioprotection via cold acclimation
Why Cold May Be Kinder to the Heart Than We Think
Most of us think of cold weather as a threat to heart health, and with good reason: sudden chills can trigger heart attacks in vulnerable people. But this study explores the other side of the story. When cold exposure is gentle and prolonged, the body can adapt in ways that actually make the heart more resistant to damage from a heart attack. The researchers set out to discover how this happens inside heart cells, focusing on a signaling route called JAK/STAT3 that links immune signals, energy production, and cell survival.
Training the Body with Gentle Cold
To probe this question, scientists gradually acclimated rats to a cool but non-freezing environment of about 9 °C (48 °F) over five weeks—cold enough to activate defenses, but not so harsh as to cause shivering or stress. Other rats stayed at room temperature. All animals were then subjected to a controlled heart attack in the lab, where blood flow to part of the heart was blocked and then restored, mimicking what happens in people with a sudden coronary artery blockage followed by treatment. This allowed the team to measure how much heart tissue died and how well the heart pumped afterward. They also gave some animals a drug that blocks JAK2, the enzyme that switches on STAT3, to test whether this pathway is truly required for the benefits of cold acclimation. 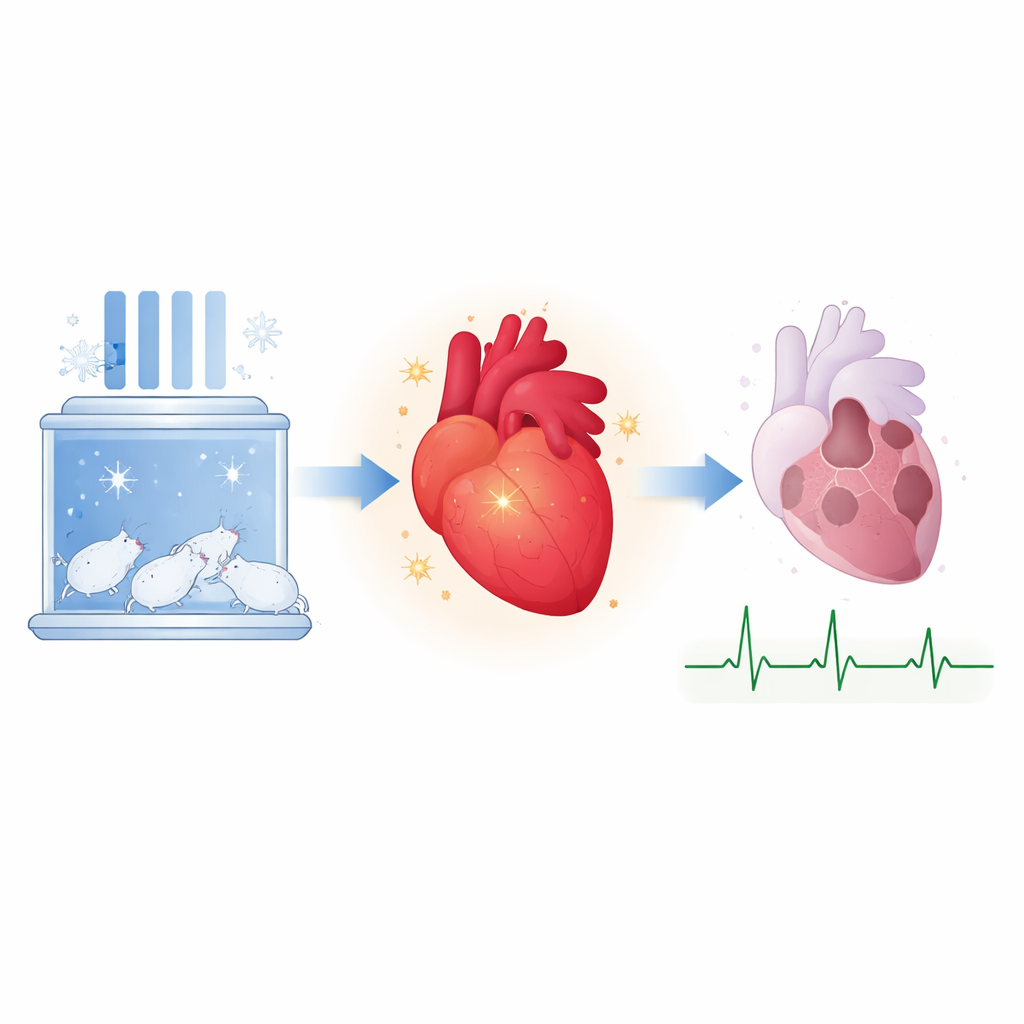
A Protective Switch Inside Heart Powerhouses
The cold-acclimated rats fared strikingly better. Compared with room-temperature controls, their hearts lost less than half as much tissue during the simulated heart attack. A key feature of this protection was healthier mitochondria—the tiny powerhouses inside cells that generate energy. When mitochondria fail under stress, they open a structure called the permeability transition pore, which can trigger cell death. In the cold-acclimated hearts, these pores opened more slowly when challenged with calcium, a stress signal, indicating sturdier mitochondria. But when the JAK2-blocking drug AG490 was given just before the end of the cold exposure, this mitochondrial resilience vanished, and the size of the heart attack injury jumped back toward control levels. This firmly tied the cold-induced protection to an active JAK/STAT3 signaling route.
Signals that Calm Damage and Cell Death
Diving deeper, the researchers looked at how STAT3 and related proteins moved around within heart cells. In normal hearts, much of STAT3 sits in mitochondria and in cell membranes. After cold acclimation, there was a reshuffling: total STAT3 shifted toward the cell surface and away from mitochondria, while a specific activated form associated with the nucleus decreased in certain compartments. Despite these subtle changes, mitochondrial STAT3 activity appeared preserved, supporting the idea that its non-genetic role—directly stabilizing energy production and keeping death-triggering pores closed—matters most for protection. At the same time, cold acclimation lowered levels of p38 MAPK, a protein linked to stress-driven cell death, without causing heart enlargement or raising blood pressure. When JAK2 was blocked, p38 levels rose again and markers of apoptosis, or programmed cell death, increased, suggesting that the cold regimen normally reins in harmful death signals through this pathway.
Immune Messages that Help, Not Harm
The study also examined chemical messengers of the immune system within the heart. Cold acclimation boosted levels of interleukin-6 (IL-6), a cytokine often associated with inflammation but also known to activate STAT3 in a protective way. Interestingly, classic pro-inflammatory signals such as tumor necrosis factor alpha (TNF-α) and interleukin-1β did not rise with cold alone and even tended to be dampened in cold-acclimated animals when the JAK2 pathway was blocked. This pattern suggests that moderate, well-controlled cold does not inflame the heart; instead, it appears to fine-tune immune signals so that IL-6 supports survival pathways without unleashing damaging inflammation. Overall, cold acclimation produced a balanced immune profile consistent with adaptation rather than injury. 
What This Could Mean for Future Heart Care
In plain terms, this work shows that living in a steady, moderately cool environment can "train" the heart to better withstand a future heart attack, and that the JAK2/STAT3 signaling axis is a crucial part of this training program. By strengthening mitochondria and dialing down harmful cell-death and inflammatory signals, cold acclimation shrinks the area of the heart that dies when blood flow is interrupted and restored. While long-term cold exposure is not a practical treatment for patients, understanding how it recruits JAK/STAT3 and related pathways could inspire new drugs or gentle cooling strategies that mimic its protective effects—offering a way to make the heart more resilient before trouble strikes.
Citation: Kasik, P., Galatik, F., Matous, P. et al. Causal relevance of the JAK/STAT pathway for cardioprotection via cold acclimation. Sci Rep 16, 12124 (2026). https://doi.org/10.1038/s41598-026-40532-4
Keywords: cold acclimation, cardioprotection, JAK2 STAT3, mitochondria, ischemia reperfusion