Clear Sky Science · en
Design, characterization, DFT studies, and molecular docking of new benzofuran–pyrazol-acrylamide hybrids as insecticidal agents against Spodoptera littoralis and Tribolium castaneum
New weapons for protecting food crops
Farmers around the world fight a constant battle against insects that devour leaves in the field and grains in storage. Two especially damaging culprits are the Egyptian cotton leafworm and the red flour beetle, which together can destroy large portions of harvests and spoiled stored flour. This paper describes the design and testing of a new family of lab‑made molecules that aim to kill these pests more precisely by targeting their nervous systems, while offering the potential for lower risks to people and the environment.
Why these pests are such a big problem
The Egyptian cotton leafworm feeds on dozens of crops, from cotton and tomatoes to wheat and strawberries, and can slash yields by up to half. The red flour beetle, meanwhile, thrives in warm grain stores and flour mills, turning valuable food into contaminated dust that also smells bad and may carry health risks. Existing insecticides are often broad‑acting, can harm non‑target organisms, and are losing effectiveness as insects evolve resistance. There is a strong push to find new chemicals that act in more specific ways, so smaller doses can work and unwanted side effects are reduced.
Building a new kind of insect‑killing molecule
The research team created eight related compounds that combine three proven building blocks often found in medicines: a benzofuran ring, a pyrazole ring, and an acrylamide group. By joining these fragments in one framework and changing just one attached ring in different ways, they generated a small "family" of candidates. They then used standard tools of chemistry—infrared and nuclear magnetic resonance spectroscopy and mass spectrometry—to confirm that each product had the intended structure. Alongside this lab work, they used advanced calculations to explore how each compound’s three‑dimensional shape and electron distribution change when a key part of the molecule flips between two mirror‑like arrangements, known as E and Z forms.
Putting the new compounds to the test
Next, the scientists measured how toxic the compounds were to fourth‑stage larvae of the cotton leafworm and to adult red flour beetles. The insects were exposed to different doses on treated surfaces, and deaths were tracked over three days. Three molecules—labeled 3a, 3b, and 3c—stood out, causing high mortality in both species, while the others were weak or inactive. Among them, 3a was most potent against the leafworm, and 3b was strongest against the beetle, achieving full kill at the highest tested dose within 72 hours. By comparing the small chemical tweaks between the active and inactive members of the family, the team found that adding electron‑donating groups, such as methyl or methoxy, in a specific position boosted insecticidal power, whereas strongly withdrawing groups, like nitro or carboxyl, largely shut it down.
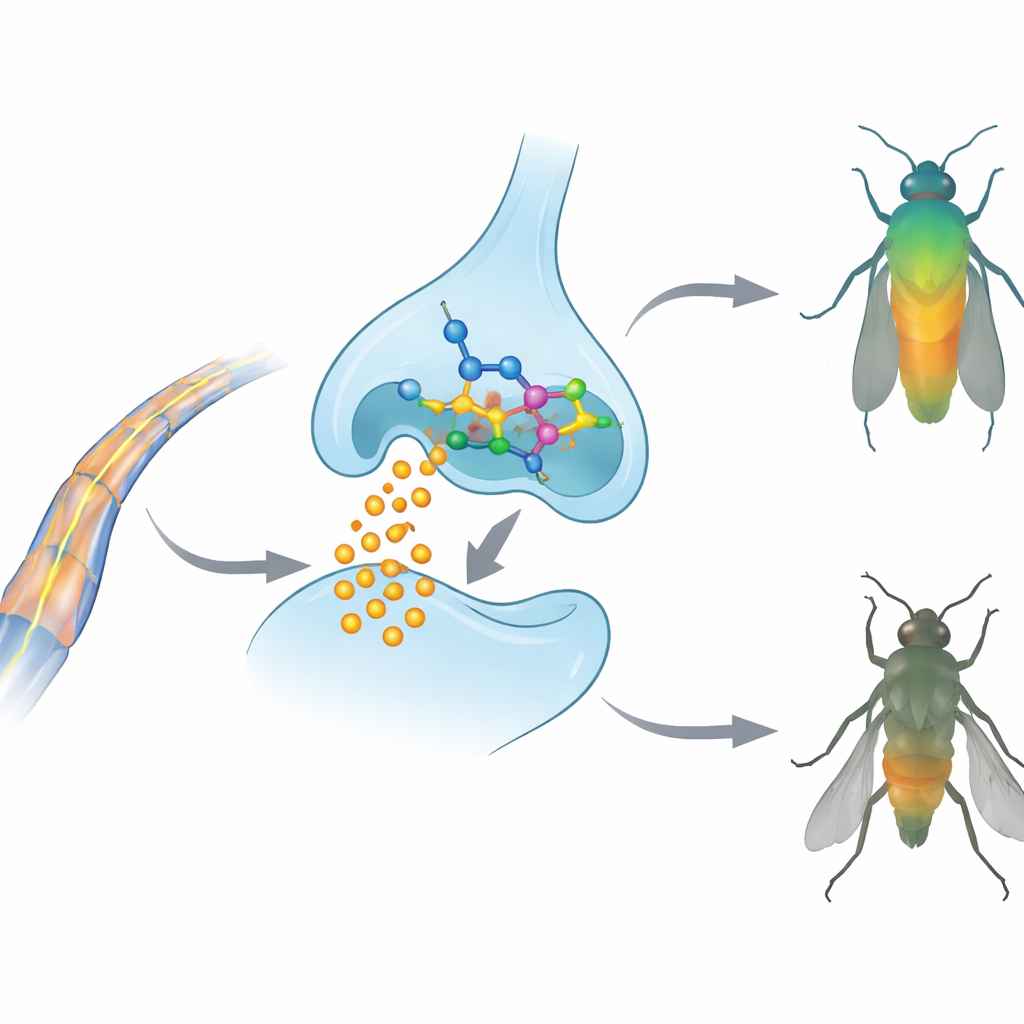
Looking inside the insect nervous system
To understand how these compounds work, the authors turned to computer models of acetylcholinesterase, a key enzyme that shuts off nerve signals by breaking down a chemical messenger called acetylcholine. Many classic insecticides disable this enzyme. Using molecular docking, the team simulated how compounds 3a, 3b, and 3c fit into the acetylcholinesterase from both target insects. All three were predicted to bind snugly in the active pocket through a network of hydrophobic contacts and hydrogen bonds, with binding strengths similar to or better than a widely used reference insecticide. Further molecular dynamics simulations, which follow the motions of atoms over time, showed that the complex between compound 3b and the enzyme remained stable and compact, suggesting a persistent blocking effect. At the same time, computer‑based ADMET screening (absorption, distribution, metabolism, excretion, and toxicity) indicated that these molecules meet common "drug‑likeness" rules and are unlikely to be strongly mutagenic or cancer‑causing, though their oily nature may limit simple water‑based formulations.
What the electronic calculations reveal
Density functional theory, a quantum‑level modeling approach, was used to probe why small structural changes make such big differences. The calculations showed that for some members of the series, especially compound 3c, the E form has a very small energy gap between key electron‑holding orbitals, making it highly reactive and a strong electron acceptor. Other members, like 3b, were predicted to prefer the Z form, which offered better alignment of electrons and larger dipole moments. These subtle electronic features influence how well a molecule can interact with the enzyme target or with surfaces, and help explain why certain variants bind more tightly even if they are not the most reactive on paper. In this way, the study links a molecule’s shape and charge distribution directly to its biological impact.
Where this work could lead
Overall, the study identifies a small set of benzofuran–pyrazole–acrylamide hybrids, especially compounds 3a, 3b, and 3c, as promising leads for new insecticides against two economically important pests. Lab assays show that they can kill caterpillars and beetles at relatively low concentrations, and computer models suggest they act by firmly blocking a vital nerve enzyme while having acceptable safety features. Although much more testing is needed in real‑world conditions and on non‑target organisms, these results outline a rational path for designing next‑generation pest control agents that are both powerful and more finely tuned to their targets.
Citation: El-Bana, G.G., Fouad, M.R., Deeb, A.D.H. et al. Design, characterization, DFT studies, and molecular docking of new benzofuran–pyrazol-acrylamide hybrids as insecticidal agents against Spodoptera littoralis and Tribolium castaneum. Sci Rep 16, 10344 (2026). https://doi.org/10.1038/s41598-026-39839-z
Keywords: insecticide design, acetylcholinesterase inhibitors, crop pest control, benzofuran pyrazole hybrids, computational toxicology