Clear Sky Science · en
Electrochemically optimized multi-component polyacrylonitrile nanofiber scaffolds as a platform for three-dimensional glioblastoma cell culture
Why a New Lab Model for Brain Cancer Matters
Glioblastoma is one of the deadliest brain cancers, and many treatments that look promising in the lab fail when they reach patients. One major reason is that most lab tests grow cancer cells as a flat sheet in a dish, which is very different from the tangled, three-dimensional structure of a real tumor in the brain. This study introduces a new lab platform that lets glioblastoma cells grow in 3D on a web of ultra-thin fibers while their behavior is monitored electrically in real time, potentially bringing lab experiments a step closer to what happens inside patients.
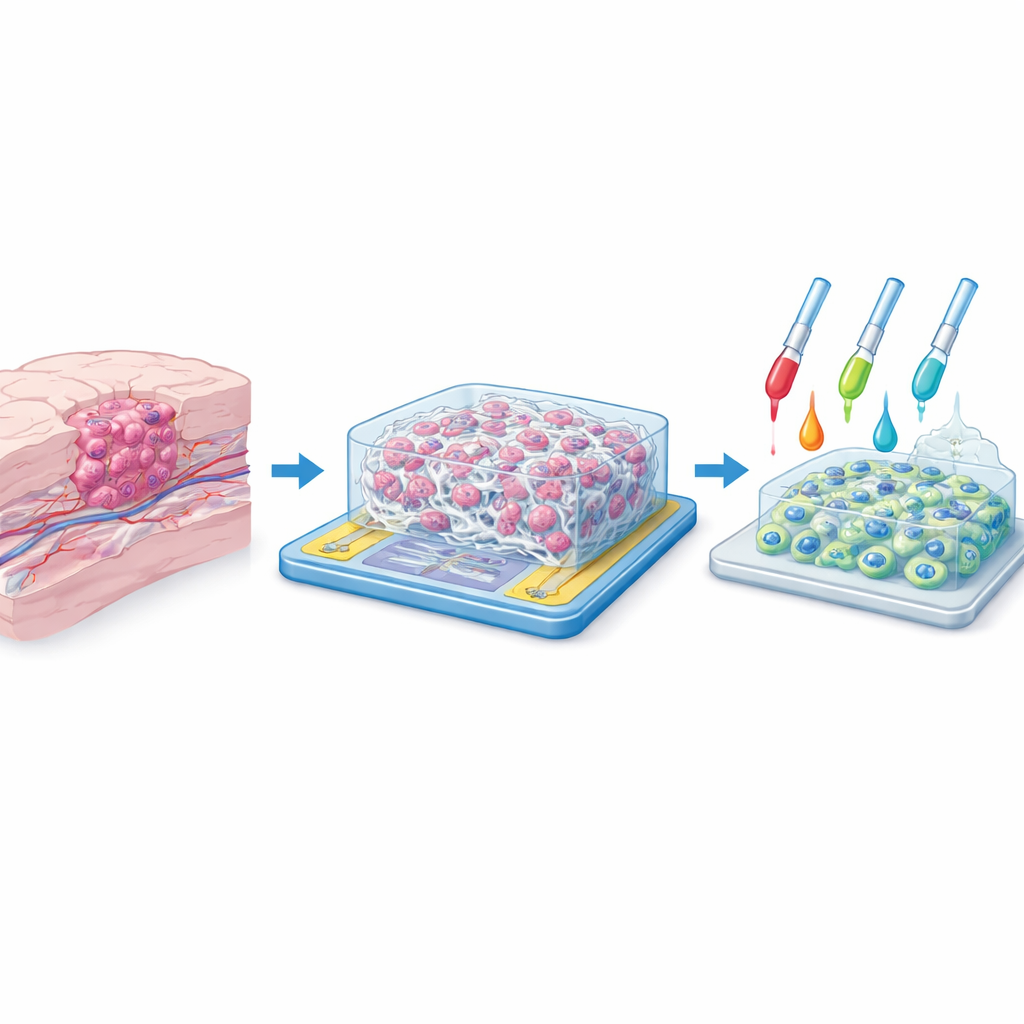
Recreating the Tumor’s Home
Inside the brain, tumor cells do not live on a flat surface. They weave between supportive tissues, cling to tiny protein fibers, and feel signals from neighboring cells in all directions. Traditional two-dimensional cultures flatten this complexity, often giving misleading results when scientists test new drugs. The researchers set out to build a more realistic “home” for glioblastoma cells: a three-dimensional scaffold made of synthetic fibers, with pore spaces large enough for cells to crawl into and around. Their goal was to mimic the physical feel of brain tissue while also allowing scientists to watch how the cells behave without disturbing them.
Spinning a Tiny Fiber Forest
To create this artificial microenvironment, the team used a technique called electrospinning, which draws liquid polymer into continuous strands thinner than a thousandth of a millimeter. The base material was polyacrylonitrile, a strong, stable plastic known for forming uniform nanofibers. By carefully controlling voltage, distance, and flow rate, they produced mats of overlapping fibers with diameters around 400–500 nanometers and pore openings of about 9–10 micrometers—just wide enough for individual cancer cells to infiltrate in three dimensions. Microscopy showed smooth, continuous fibers without bead defects and a consistent, highly porous network, suggesting that cells would encounter a realistic, maze-like environment similar to the natural tissue around a tumor.
Blending Multiple Ingredients into One Smart Scaffold
The innovation in this work goes beyond the basic fiber mat. The authors blended several functional ingredients into the fibers to fine-tune both cell behavior and electrical readout. They tested six combinations on transparent conducting glass: plain fibers; fibers with a light-responsive coumarin dye (C500); fibers with graphene oxide, a sheet-like carbon material; fibers with a holmium-based metal–organic framework; and two blends that combined graphene oxide with either C500 or the metal–organic framework. These additives were chosen to strengthen the fibers, tweak surface chemistry, and improve their ability to carry tiny electrical signals. Sophisticated electrical tests showed that some mixtures, particularly those with both graphene oxide and the metal–organic framework, allowed electrons to move very easily through the system.
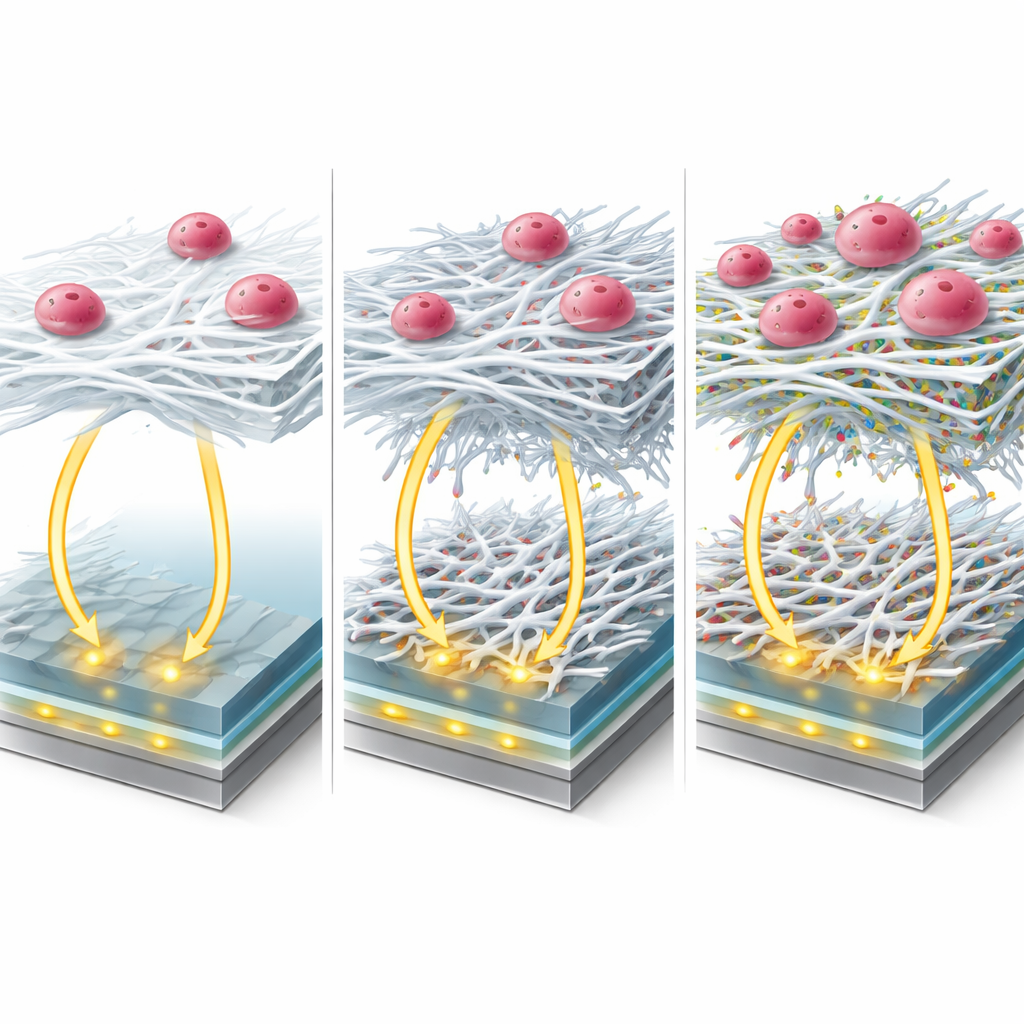
When Good Electricity Meets Poor Biology
However, what looks best electrically is not always best for living cells. When the researchers grew two human glioblastoma cell lines on the different fiber-coated electrodes, they found a striking mismatch. The configuration with graphene oxide and the metal–organic framework had the lowest electrical resistance but failed almost completely at supporting cell attachment. In contrast, fibers containing the coumarin dye C500 provided both low resistance and excellent cell health: more than 95 percent of the cells remained viable, and fluorescent staining revealed a dense, well-organized three-dimensional spread of nuclei throughout the scaffold. Electrical impedance measurements before and after cell seeding clearly changed as cells colonized the fibers, confirming that the platform could track cell growth without the need to remove or stain them each time.
What This Means for Future Brain Cancer Research
The study shows that it is possible to combine a realistic three-dimensional growth environment for glioblastoma cells with a built-in “stethoscope” that listens electrically to what the cells are doing. Among the tested recipes, the C500-enhanced fiber scaffold struck the best balance between being friendly to cells and sensitive to tiny electrical changes. For non-specialists, the key message is that this platform can host brain tumor cells in a way that more closely resembles real tumors, while letting scientists monitor how they grow and respond to potential drugs in real time. Such a system may help bridge the gap between over-simplified lab dishes and the complex reality of the human brain, improving the odds that therapies proven here will truly benefit patients.
Citation: Kurt, Ş., Bal Altuntaş, D., Sevim Nalkıran, H. et al. Electrochemically optimized multi-component polyacrylonitrile nanofiber scaffolds as a platform for three-dimensional glioblastoma cell culture. Sci Rep 16, 12644 (2026). https://doi.org/10.1038/s41598-026-39640-y
Keywords: glioblastoma, 3D cell culture, nanofiber scaffold, tumor microenvironment, electrochemical monitoring