Clear Sky Science · en
Claudin18.2 promote gastric cancer proliferation by activating MCM2/5
Why this matters for stomach cancer
Stomach cancer remains one of the deadliest cancers worldwide, in part because many patients are diagnosed late and existing treatments do not work for everyone. This study focuses on a little-known gatekeeper molecule on stomach cells, called claudin18.2, and asks two big questions: can drugs that recognize this molecule really help patients live longer, and what exactly does claudin18.2 do inside tumor cells to help them grow? The answers could sharpen how doctors choose patients for new targeted drugs and inspire novel combination therapies.
A surface marker with treatment promise
The researchers first revisited two large international clinical trials, named SPOTLIGHT and GLOW, which tested an antibody drug called zolbetuximab in patients with advanced stomach or gastroesophageal junction cancer whose tumors carried claudin18.2 but lacked another common marker, HER2. Pooling data from 1,072 patients, they found that adding zolbetuximab to standard chemotherapy clearly delayed cancer worsening compared with chemotherapy alone. The benefit was especially notable in Asian patients, in cancers that began in the main body of the stomach rather than the junction with the esophagus, and in tumors of the so-called intestinal type. These findings strengthen the case that claudin18.2 is not just a marker on the tumor surface, but a useful target for first-line therapy in selected patients.
A marker that tracks with more aggressive disease
To understand what claudin18.2 means for individual patients, the team examined tissue samples from 92 people who had surgery for stomach cancer. Using a highly specific antibody that recognizes only the claudin18.2 subtype, they scored how strongly tumor cell membranes were stained. About 46 percent of tumors were positive. Those positive tumors were more likely to have spread to nearby lymph nodes, and more patients with claudin18.2-positive cancers were already in advanced stages III or IV at diagnosis. When the scientists followed patient outcomes, they found that people whose tumors expressed claudin18.2 had shorter overall survival: only about 31 percent were alive at five years, compared with 52 percent among those whose tumors lacked the marker. This suggests that claudin18.2 may flag a more aggressive form of the disease.
How claudin18.2 drives tumor cell growth
Next, the investigators turned to cell and animal models to uncover what claudin18.2 does inside cancer cells. They focused on a stomach cancer cell line that naturally makes high levels of claudin18.2 and used genetic tools to switch this protein off. When claudin18.2 was reduced, the cells divided more slowly and many became stuck at a key checkpoint just before cell division, known as the G2/M phase. In mice, tumors grown from claudin18.2-deficient cells stayed much smaller and lighter than tumors from unmodified cells, even though the animals’ overall health and body weight were unchanged. Tumor samples from these mice also showed fewer cells actively cycling and lower levels of proteins linked to cell replication.
The link to the cell’s copying machines
To probe the mechanism, the team analyzed which genes changed when claudin18.2 was knocked down. They found broad shifts in pathways tied to the cell cycle and DNA copying. A family of proteins called minichromosome maintenance proteins, or MCMs, stood out. These proteins act like clamps that slide along DNA to help it unwind and duplicate. In claudin18.2-reduced cells, the levels of several MCM family members dropped, especially MCM2 and MCM5, which together form a critical entry gate for DNA into the replication machinery. Restoring claudin18.2 brought MCM2 and MCM5 back up and rescued the cells’ ability to proliferate. In mouse tumors, lowering claudin18.2 also lowered MCM2 and MCM5. This tight connection points to these MCM proteins as key middlemen through which claudin18.2 boosts tumor growth.
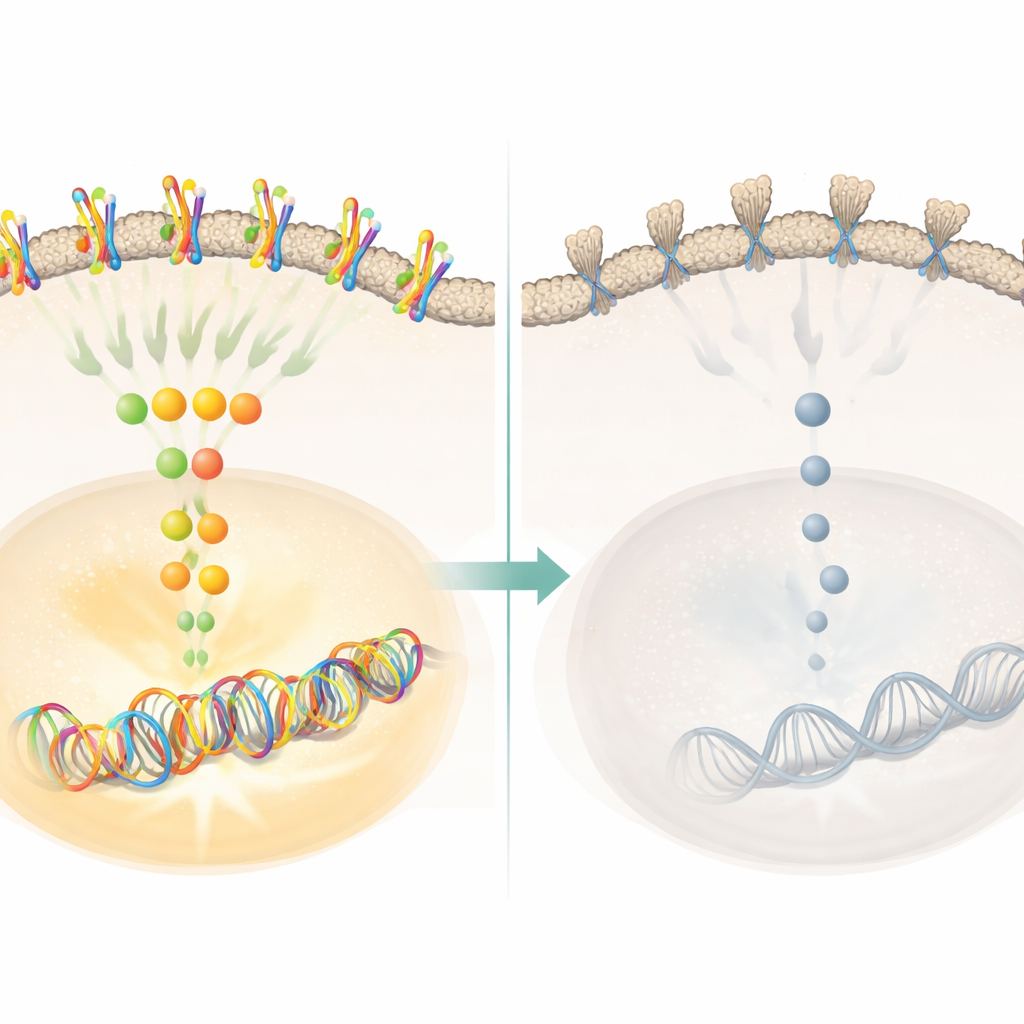
Signaling switches that control growth
The study further showed that claudin18.2 influences major growth-control pathways inside the cell. When claudin18.2 was silenced, activity dropped in receptor tyrosine kinases on the cell surface and in downstream molecules such as ERK and CDK1, which act as switches for cell-cycle progression. Phosphorylation, a chemical tag that turns these switches on, decreased on ERK, CDK1, and MCM2, suggesting that the entire signaling chain driving DNA replication was dialed down. When claudin18.2 levels were restored, signaling revived. Database analyses also suggested that tumors with high claudin18.2 might be especially sensitive to drugs that inhibit cell-cycle enzymes, hinting that pairing claudin18.2-targeted therapies with such agents could be fruitful.
What this means for patients
In everyday terms, this work shows that claudin18.2 is more than a tag on certain stomach cancers: it acts like a master switch that helps tumor cells keep copying their DNA and dividing, in part by revving up the MCM "copying machines" and key growth pathways. Blocking claudin18.2, whether with a drug like zolbetuximab or by dialing down its activity, slows tumors in cells, in mice, and in patients, and tumors that carry this marker tend to behave more aggressively. Together, these findings support using precise tests for claudin18.2 to select patients who are most likely to benefit from targeted therapy and to design smart combinations that shut off the tumor’s growth engine at multiple points.
Citation: Zheng, B., Fu, M., Lou, F. et al. Claudin18.2 promote gastric cancer proliferation by activating MCM2/5. Sci Rep 16, 10263 (2026). https://doi.org/10.1038/s41598-026-39540-1
Keywords: gastric cancer, claudin18.2, targeted therapy, cell cycle, DNA replication