Clear Sky Science · en
Genome-scale DNA methylome and transcriptome profiling of midgut of Bombyx mori infected with BmCPV
Why Silkworm Guts Matter to Us
Silkworms may seem like humble farm insects, but they sit at the heart of textile, agricultural, and even biomedical industries. When a gut-infecting virus strikes them, whole crops of cocoons can be lost. This study explores how one such virus subtly rewires the silkworm’s genetic control system, not by changing the DNA itself, but by adjusting chemical tags on it. Understanding this hidden layer of control can help protect an important economic insect and deepen our broader grasp of how viruses manipulate their hosts.
A Gut Virus with Big Economic Impact
The work focuses on Bombyx mori, the domesticated silkworm, and a common pathogen called Bombyx mori cytoplasmic polyhedrosis virus (BmCPV). This double-stranded RNA virus specifically targets cells in the silkworm midgut, the organ that digests food. Outbreaks of BmCPV can stunt growth and kill larvae, causing serious economic damage. Earlier research had shown that BmCPV infection changes how many silkworm genes are switched on or off and also alters marks on proteins that package DNA, known as histones. But how another key type of chemical tag on DNA itself, called DNA methylation, fits into this story was largely unknown.
The Hidden Code on DNA
DNA methylation is a small chemical change: a methyl group is added to specific building blocks in DNA, often at sites where a cytosine sits next to a guanine. Despite its small size, this tag can strongly influence whether nearby genes are active. In many animals, DNA methylation helps regulate development, silence repetitive DNA, and fine-tune when and where genes are used. Viruses infecting humans and other vertebrates are known to alter host DNA methylation to their advantage, shifting the host’s gene activity. In insects, overall methylation levels are much lower, but earlier work hinted that silkworm viruses can still exploit this mechanism. The authors set out to examine, across the entire genome, how BmCPV infection reshapes methylation patterns in the silkworm midgut and how these changes relate to gene activity.
Reading the Chemical Marks Genome-Wide
To do this, the team infected a well-studied silkworm strain and collected midgut tissue at two time points: 48 and 96 hours after infection. They also collected matching gut samples from uninfected larvae of the same age. From these tissues, they performed two large-scale measurements. First, they used whole-genome bisulfite sequencing, a method that reveals which cytosines across the genome carry methyl groups. Second, they used RNA sequencing to measure which genes were more or less active under each condition. They carefully filtered and aligned hundreds of millions of DNA reads, checked data quality, and calculated methylation levels at single sites as well as over broader genomic regions such as gene bodies, promoters, and untranslated regions.
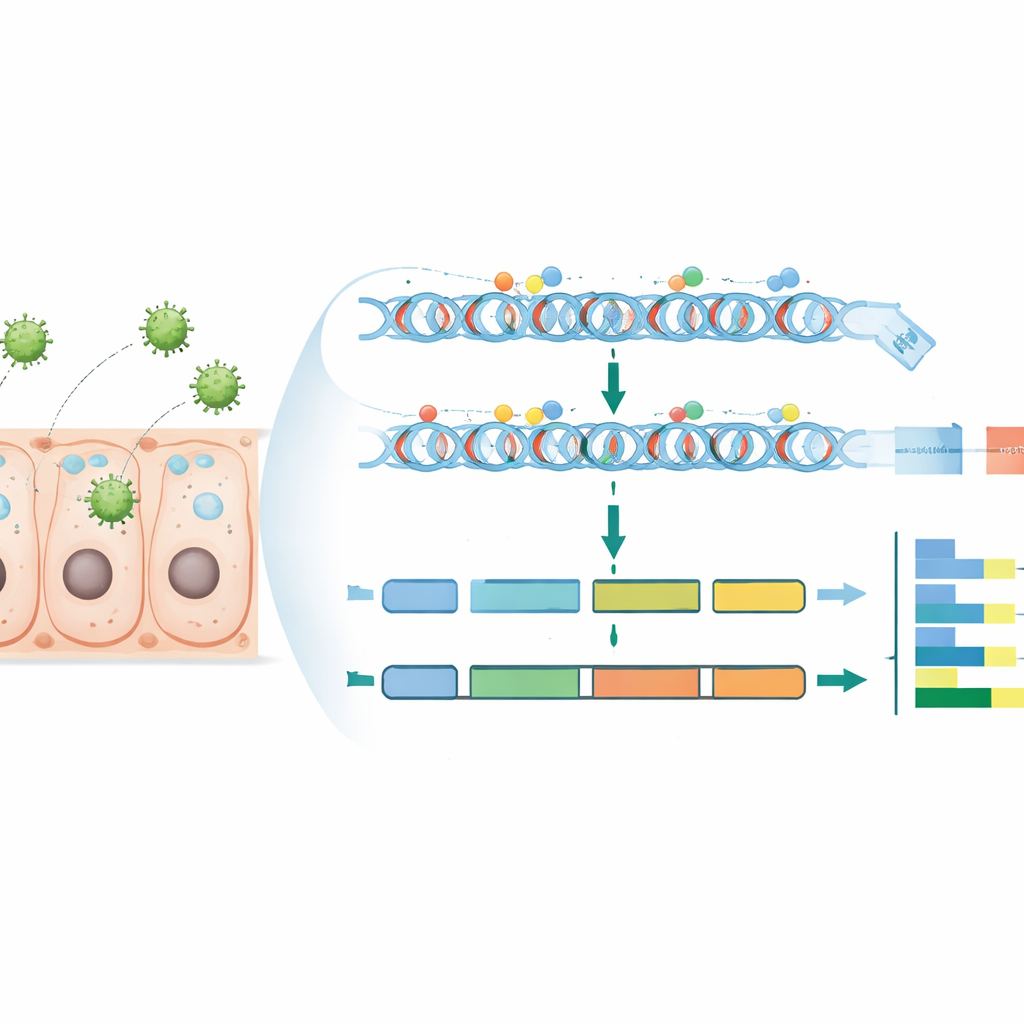
Where the Virus Tweaks the Controls
The researchers found that, as in many insects, overall DNA methylation in silkworm is low, and most methylated sites occur in the familiar CG context. Within the genome, methylation was not spread evenly: it tended to be higher in gene-related regions such as exons and untranslated regions, and lower in classic CpG-rich islands and repeated DNA. By comparing infected and uninfected samples at both time points, they pinpointed differentially methylated regions (DMRs) – stretches of DNA where methylation rose or fell during infection. They then linked these regions to nearby genes, especially when DMRs overlapped promoter regions just upstream of genes, which are crucial for turning genes on and off. Finally, by integrating methylation data with RNA sequencing, they identified genes whose activity shifts were closely associated with changes in promoter methylation.
Checking the Signals and Sharing the Data
To be sure these genome-wide patterns were real, the team validated selected regions using targeted methods. They employed methylation-specific PCR to confirm methylation changes at chosen sites and quantitative PCR to verify shifts in gene expression levels. In each case, the focused tests matched the large-scale sequencing results, strengthening confidence in the dataset. All sequencing reads, processed methylation tracks, and lists of methylated sites and differentially methylated regions have been deposited in public databases, providing a rich resource for other researchers studying insect immunity, virus–host interactions, or epigenetic regulation.
What This Means for Silkworm Health
In plain terms, this study shows that the gut virus BmCPV does not merely invade silkworm cells; it is associated with subtle but widespread retuning of the silkworm’s genetic control knobs via DNA methylation. Specific genes gain or lose these chemical tags near their start regions, and those same genes show corresponding increases or decreases in activity. While the work does not yet translate directly into a cure, it maps the control panel that the virus seems to touch. In the long run, such insight could help breeders and biotechnologists design silkworm strains that resist infection more effectively and may also shed light on common principles by which viruses manipulate the epigenetic machinery of their hosts.
Citation: Qiu, Q., Liu, Z., Huang, Y. et al. Genome-scale DNA methylome and transcriptome profiling of midgut of Bombyx mori infected with BmCPV. Sci Data 13, 568 (2026). https://doi.org/10.1038/s41597-026-06922-z
Keywords: silkworm virus, DNA methylation, epigenetics, host–virus interaction, Bombyx mori midgut