Clear Sky Science · en
Biallelic variants in RNU2-2 cause a remarkably frequent developmental and epileptic encephalopathy
Why a tiny gene matters for children’s brains
Neurodevelopmental disorders such as intellectual disability and epilepsy affect millions of children worldwide, yet for about half of affected families, genetic testing still fails to find a clear cause. This study reveals that changes in an unusually small piece of genetic material, a non‑coding RNA gene called RNU2-2, underlie a surprisingly common and severe childhood brain disorder marked by early developmental delay and hard‑to‑treat seizures.
From unsolved cases to a hidden culprit
The researchers began with tens of thousands of people who had undergone whole‑genome sequencing through large national and international projects, many of them children with unexplained learning problems or epilepsy. They focused on a special class of genes that do not make proteins but instead produce small RNA molecules that help assemble the cell’s splicing machinery—the system that edits raw RNA messages. By scanning 1,901 such small RNA genes for rare variants inherited from both parents, they found that one gene, RNU2-2, stood out. Children with unresolved neurodevelopmental disorders carried rare pairs of RNU2-2 variants far more often than expected when compared with tens of thousands of control genomes.
A new recessive syndrome takes shape
To confirm that this was not a statistical fluke, the team gathered additional cases from rare‑disease databases in the United Kingdom, Europe, the Middle East, and Asia. Altogether they identified 84 affected individuals from 67 families who had rare “biallelic” RNU2-2 variants—either the same change on both copies of the gene or two different damaging changes. Despite being collected from around the world, these children showed strikingly similar clinical pictures: profound developmental delay, serious intellectual disability, and seizures starting in infancy in many cases. A large fraction never learned to speak or walk independently, and brain scans commonly showed loss of brain tissue and enlarged fluid spaces, consistent with an encephalopathy, or global brain dysfunction.
How a tiny RNA upsets brain function
RNU2-2 produces a short RNA that forms part of the major spliceosome, the cellular machine that cuts and pastes pieces of RNA before they are translated into proteins. Humans also carry a highly similar “backup” version, U2-1. By analyzing RNA from blood samples, the scientists found that children with biallelic RNU2-2 variants had markedly reduced amounts of the U2-2 RNA, while U2-1 levels remained relatively stable. More importantly, the ratio of U2-2 to U2-1 was consistently much lower in affected individuals than in any controls, making this imbalance a promising diagnostic marker. The data suggest that many of the disease‑causing variants destabilize the U2-2 RNA, reducing its abundance to a level that the developing brain cannot tolerate, even if U2-1 partly compensates.
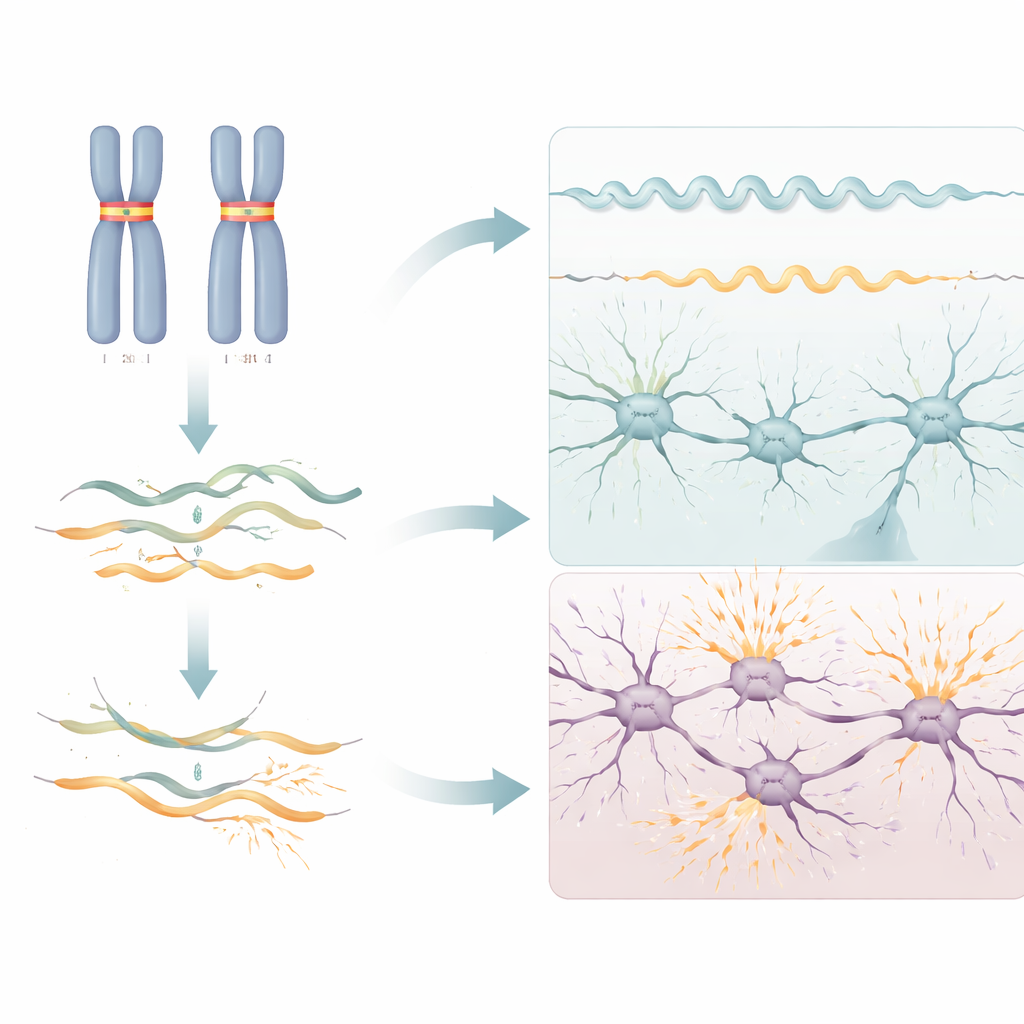
Distinct disorders from the same gene
RNU2-2 was already known to cause a different, dominant neurodevelopmental condition when only one copy of the gene carries certain specific mutations. The new work shows that the recessive syndrome uncovered here is not just a milder or earlier version of that disorder. The damaging variants occupy a broader range of positions in the RNA, the RNA changes differ—dominant cases show widespread splicing disruption in blood, whereas recessive cases mostly show RNA depletion—and the clinical features overlap only partly. For example, seizures in the first year of life and muscle stiffness (spasticity) are far more common in the recessive form, while distinctive hand movements and facial appearance are more typical of the dominant one.
How common is this hidden condition?
When the researchers compared RNU2-2 to all other known recessive genes in their large UK cohort of neurodevelopmental disorders, the recessive RNU2-2 syndrome emerged as the single most frequent recessive diagnosis, outnumbering the next most common gene by more than threefold. Together with related conditions caused by variants in other small RNA genes, this family of “RNU‑opathies” accounts for nearly 1.5% of previously unsolved neurodevelopmental cases in the 100,000 Genomes Project—an unexpectedly large contribution from genes that together occupy only a few hundred DNA letters.
What this means for families and future care
For families, the discovery of this recessive RNU2-2 syndrome means that a clear genetic answer is now within reach for many children with severe developmental delay and epilepsy. Because two altered copies are required, parents are usually healthy carriers, and accurate testing can guide reproductive decisions and identify at‑risk siblings. For clinicians and researchers, the work highlights the importance of tiny non‑coding genes that were once overlooked in standard tests. It also points to a measurable molecular signature—the lowered U2-2 to U2-1 RNA ratio—that may help confirm diagnoses and, in the longer term, could inform efforts to design therapies that restore the delicate RNA balance needed for healthy brain development.
Citation: Jackson, A., Blakes, A.J.M., Alhaddad, B. et al. Biallelic variants in RNU2-2 cause a remarkably frequent developmental and epileptic encephalopathy. Nat Genet 58, 798–809 (2026). https://doi.org/10.1038/s41588-026-02551-9
Keywords: neurodevelopmental disorders, epileptic encephalopathy, non-coding RNA, spliceosome, genome sequencing