Clear Sky Science · en
Androgen activity in the male embryonic hindbrain drives lethal PFA ependymoma
Why Boys’ Brains and a Rare Childhood Tumor Are Linked
Some brain tumors strike very young children and are often deadly, despite surgery and radiation. One of the worst is a tumor deep in the back of the brain called posterior fossa type A ependymoma. Doctors have long noticed that this cancer happens more often in boys and that boys tend to do worse than girls, but no one knew why. This study uncovers a surprising culprit: male sex hormones, or androgens, acting on immature brain cells before and shortly after birth.
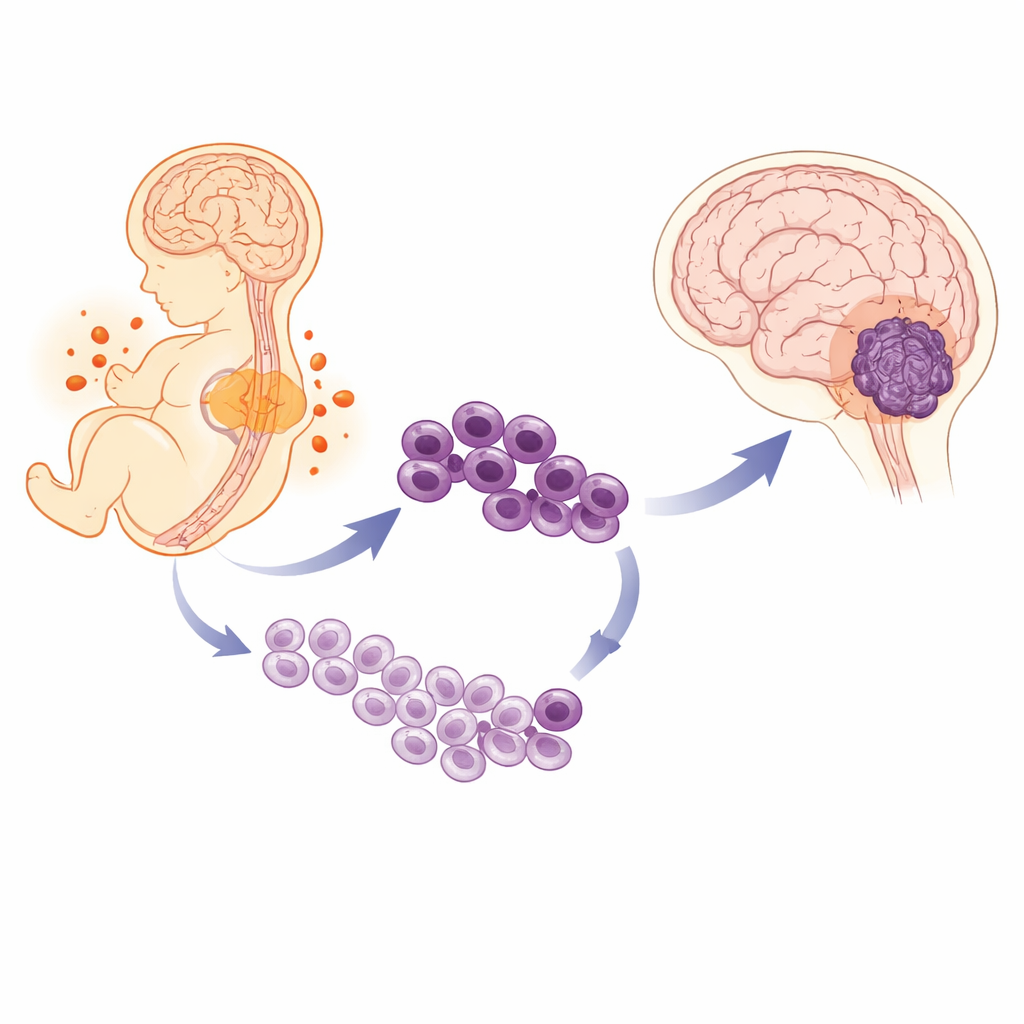
A Childhood Cancer with a Hidden Bias
The authors first examined large groups of children with this tumor and confirmed that boys not only get it more often, they also have poorer long-term survival than girls, even when they receive the same aggressive treatment. To understand what makes the tumors different, the team used single-cell RNA sequencing, a technique that reads which genes are active in thousands of cells one by one. In tumors from boys, a larger share of cells looked and behaved like very immature “stem-like” cells, whereas tumors from girls contained more cells that appeared further along the path toward becoming specialized brain cells. The more a boy’s tumor was enriched in these primitive cells, the worse his outcome tended to be.
Tracing the Tumor Back to the Developing Brain
Because these tumors appear in toddlers and resemble fetal brain tissue, the researchers suspected that something in normal brain development might set the stage for cancer. They built a detailed atlas of over 400,000 single cells from the hindbrain of mouse embryos at multiple stages, carefully balancing male and female samples. This map showed how early neural stem cells gradually turn into gliogenic progenitors—cells destined to become support cells such as astrocytes—and then into mature glia. When the team compared male and female embryos, they saw that male progenitor cells, especially in the hindbrain, tended to stay in a more primitive, fast-dividing state, while female cells showed stronger signs of maturing into their final forms.
Androgens Slow Maturation of Key Brain Cells
To tease apart whether this difference came from sex chromosomes or from hormones, the team used a “four-core genotype” mouse model that separates having testes (and thus making androgens) from having XY chromosomes. Across genotypes, what mattered most was the presence of testes and androgens, not the chromosomal sex. In embryos with testes, hindbrain progenitor cells showed gene activity patterns linked to stem cell maintenance, rapid division and blocked differentiation. In embryos with ovaries, the same cell types showed more genes tied to maturation and glial development. Many of the genes and control switches boosted by androgens in normal male embryos were also more active in the most aggressive, stem-like tumor cells from boys, tying the tumor’s behavior directly back to hormone-shaped developmental programs.
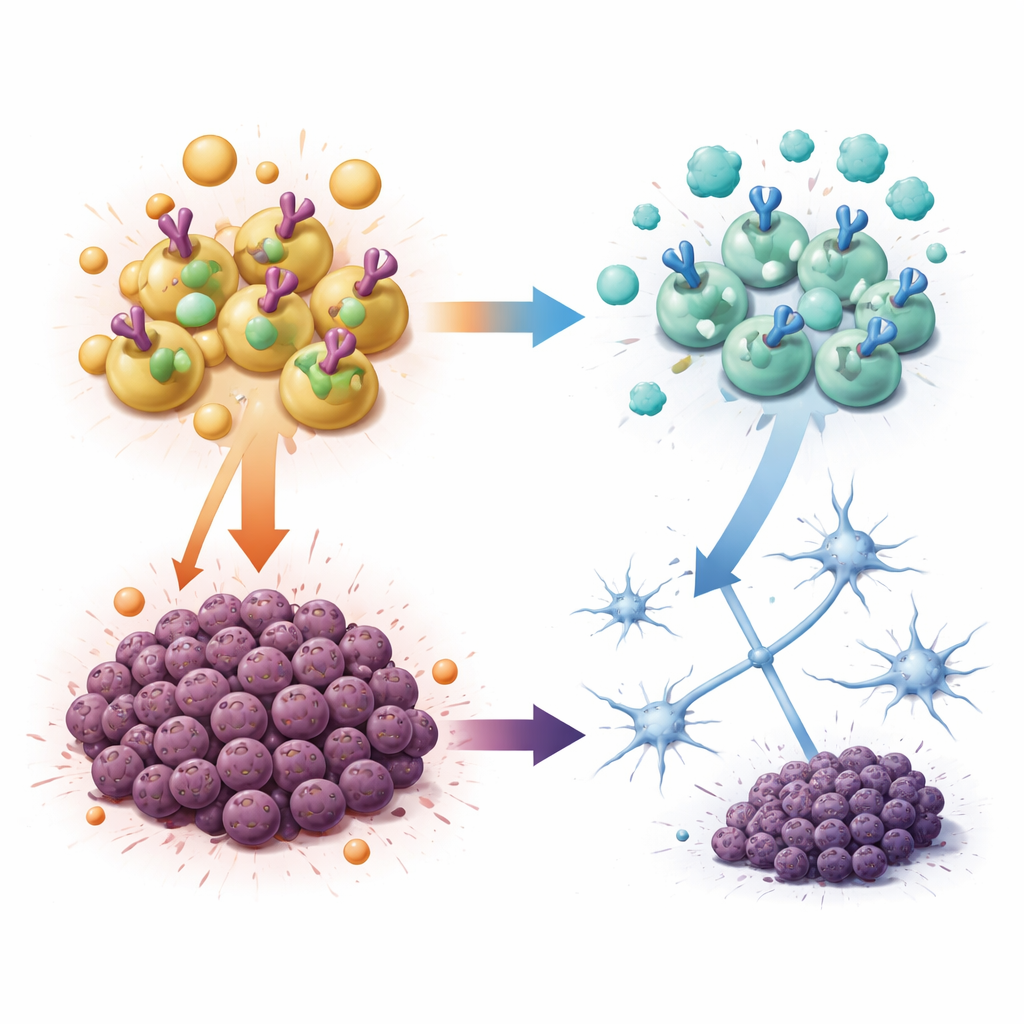
Hormones that Fuel – and Drugs that Starve – the Tumor
The researchers then asked whether androgens can directly change how tumor cells grow. In dishes, tumor cells from both boys and girls made more copies of themselves and formed more colonies when exposed to testosterone or its potent cousin dihydrotestosterone, but not when exposed to female hormones such as estrogen or progesterone. These tumor cells carried the androgen receptor, a protein that moves into the cell nucleus and turns on genes when androgens are present. Blocking this receptor with enzalutamide, a drug already used in prostate cancer, or degrading it with an experimental compound sharply reduced the stem-like capacity and growth of these tumor cells. Importantly, other pediatric brain cancer cell types did not respond much to androgens or to androgen-blocking drugs, suggesting that this vulnerability is specific to this hindbrain tumor.
What This Means for Children and Future Therapies
Taken together, the findings support a simple but powerful idea: in the developing hindbrain, androgens keep certain progenitor cells in an immature, fast-growing state for longer in males than in females. This extended window raises the chance that these cells will undergo malignant change and, once transformed, remain highly stem-like and aggressive, helping explain why boys are hit harder by this tumor. Because the same hormone signals that shape normal development also feed the cancer, treatments that block androgen signaling—carefully adapted for infants and young children—could offer the first rational targeted therapy for this devastating disease.
Citation: Zhang, J., Ong, W., Rasnitsyn, A. et al. Androgen activity in the male embryonic hindbrain drives lethal PFA ependymoma. Nature 652, 763–773 (2026). https://doi.org/10.1038/s41586-026-10264-6
Keywords: ependymoma, androgen signalling, pediatric brain cancer, hindbrain development, sex differences in cancer