Clear Sky Science · en
Parallel but distinct adaptive routes in the budding and fission yeasts after 10,000 generations of experimental evolution
Why tiny yeasts can teach us big lessons
When we think about evolution, we often picture fossils and ancient forests, not microbes growing in plastic plates. Yet by watching yeast cells adapt in the lab over thousands of generations, scientists can replay evolution in fast‑forward. This study followed two distant cousins of yeast, raised in exactly the same low‑oxygen, sugar‑rich environment for about 10,000 generations, to ask a deceptively simple question: if you start with similar organisms and give them the same challenge, do they find the same genetic solutions—or does history push them down different paths?
Setting up evolution in a bottle
The researchers focused on a well‑known model, budding yeast, and its lesser‑known cousin, fission yeast. These species share most of their genes but differ in how their genomes are organized and how they normally live. Fission yeast, for example, relies heavily on its mitochondria to breathe oxygen and is naturally poor at fermentation, whereas budding yeast is a champion fermenter. The team grew 15 independent fission yeast populations in tiny wells filled with rich, high‑sugar liquid kept under lids and without shaking—conditions that limit oxygen and encourage fermentation. Every day, the cultures were diluted so only a fraction of cells survived to seed the next round, and this was repeated for roughly 10,000 generations. Along the way, they froze samples, sequenced DNA every 1,000 generations, and measured which populations grew faster than their ancestors.
Same environment, different evolutionary payoffs
All of the surviving fission yeast populations became fitter than their ancestor, meaning they grew better in the experimental conditions. But compared with budding yeast exposed to the same environment in a previous study, fission yeast gained fitness more slowly. This difference was not because fission yeast started out better suited to the conditions—in fact, ancestral budding yeast grew better at the outset. Instead, the authors suggest that budding yeast’s evolutionary past, including a duplicated genome and long experience with low‑oxygen niches, leaves it with more genetic “spare parts” and pathways it can tweak. Fission yeast, with more essential genes and a stricter reliance on respiration, has fewer easy knobs to turn and thus adapts through a different mix of changes.
Rewiring how cells handle sugar and oxygen
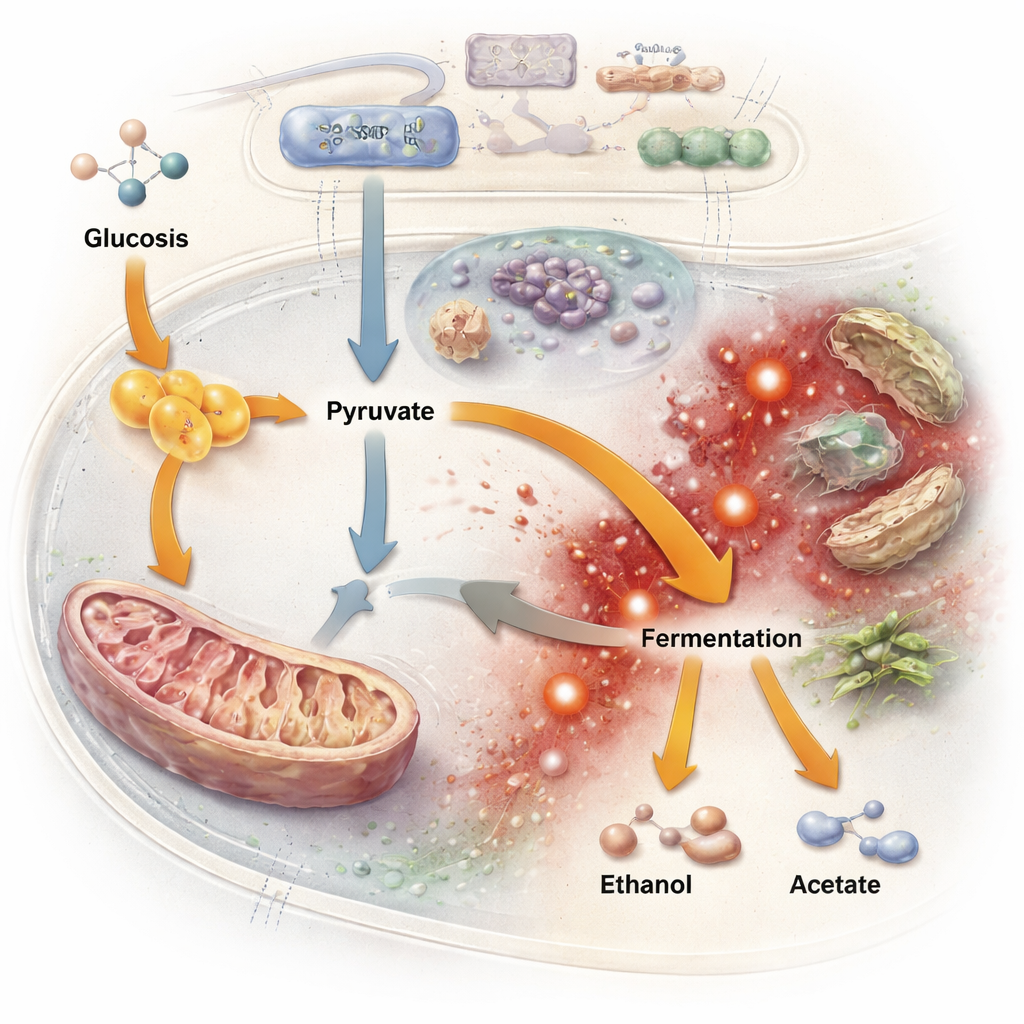
By tracking mutations over time, the team found that fission yeast rarely relied on the blunt tool of breaking genes entirely. Loss‑of‑function mutations that disrupt proteins—common in adapting budding yeast—were relatively scarce in fission yeast’s coding regions and were filtered by purifying selection. Instead, many important changes appeared in non‑coding stretches of DNA that help control when genes turn on or off, consistent with fission yeast’s genome being rich in regulatory regions. Across independent populations, evolution repeatedly hit a small set of “multi‑hit” genes tied to carbon metabolism and stress responses. A key target was the gene for pyruvate kinase, an enzyme that sits at a crucial branch point for sugar breakdown. Mutations around this gene, combined with altered activity of another enzyme that funnels carbon into the mitochondria, effectively shifted carbon flow away from respiration and toward fermentation. In other words, fission yeast rewired its internal plumbing to get by with less oxygen.
The hidden cost: fragile defenses against stress
This metabolic makeover came with a price. Fermentation in a low‑oxygen, high‑sugar world can lead to build‑up of ethanol, acetate, and other by‑products that damage mitochondria and generate reactive oxygen species—highly reactive molecules that harm cellular components. When the scientists challenged the evolved populations with hydrogen peroxide, a source of oxidative stress, most were more sensitive than the original strain. Genetic changes in iron transport, redox‑related enzymes, and a major oxidative‑stress regulator pointed to weakened protective systems. At the same time, RNA sequencing showed widespread shifts in gene activity across many pathways, including transport of key molecules, lipid handling, and autophagy (the cell’s recycling system). Strikingly, many of these expression changes occurred in genes that were not themselves mutated, implying that evolution often acted through “master switches” that control networks of genes rather than fine‑tuning each one directly.
What this tells us about evolution in general
Together, the findings paint a picture of “parallel but distinct” evolution. Both budding and fission yeasts, and even distant microbes like bacteria, tend to solve low‑oxygen challenges by redirecting carbon flow and tweaking central metabolism. Yet the exact genes, mutations, and side‑effects differ, shaped by each species’ genetic architecture and evolutionary past. For fission yeast, adapting to a hypoxic, sugar‑rich environment often means leaning harder on fermentation and accepting increased vulnerability to oxidative damage. This work suggests that to predict how organisms—from microbes to cancer cells—will evolve under stress, we must consider not only the environment but also the hidden constraints and histories written into their genomes.
Citation: N’Guessan, A., Wang, V., Bakerlee, C.W. et al. Parallel but distinct adaptive routes in the budding and fission yeasts after 10,000 generations of experimental evolution. Nat Ecol Evol 10, 765–778 (2026). https://doi.org/10.1038/s41559-026-03017-1
Keywords: experimental evolution, yeast adaptation, metabolism, hypoxia, oxidative stress