Clear Sky Science · en
Gonococcal outer membrane vesicle vaccines: bacterial population biology, clinical trials, immune profiling, and vaccine design
Why this sexually transmitted infection matters
Gonorrhoea is one of the most common sexually transmitted infections worldwide, and it is becoming harder to treat as the bacterium behind it evolves resistance to antibiotics. This article reviews a promising new line of defence: vaccines built from tiny bubbles naturally shed from related bacteria. It explains how the gonorrhoea bacterium dodges our immune system, why certain meningitis vaccines unexpectedly seem to offer some protection, and how scientists are now redesigning these bubble-based vaccines to more directly target gonorrhoea.
A shape-shifting germ that outsmarts immunity
The culprit, Neisseria gonorrhoeae, has spent thousands of years adapting to humans. It swaps DNA with its relatives and even other species, helping it quickly pick up antibiotic resistance. On its surface, key molecules constantly change their appearance or switch on and off, allowing the germ to hide from immune attack and re-infect the same person. Mapping this diversity with modern genetic tools shows that today’s gonorrhoea population is made up of many related but distinct lineages. That matters for vaccines: a shot based on only a few lab strains may miss much of what is circulating in real-world infections.
How the germ turns our defences to its advantage
Our first line of defence against bacteria includes blood proteins that punch holes in invaders and immune cells that coordinate longer-term protection. Gonorrhoea has learned to hijack both. It decorates its surface to recruit human “brakes” that shut down the hole-punching system and produces proteins that trigger antibodies which bind but do not kill, blocking more useful antibodies from doing their job. At mucosal surfaces such as the genital tract, it can even use complement proteins as a bridge to enter cells. Inside the tissues, it pushes immune responses toward a short-lived, inflammatory pattern dominated by a type of helper cell called Th17, while suppressing the memory-forming Th1 and Th2 responses. As a result, natural infection usually does not leave behind lasting protective immunity.
Surprising help from meningitis vaccines
An unexpected clue came from New Zealand, where a vaccine originally designed to control an outbreak of meningitis B was later linked to about a one-third reduction in cases of gonorrhoea. That vaccine, and a newer one called 4CMenB, both contain outer membrane vesicles (OMVs) — nanoscale spheres pinched off from the surface of meningitis bacteria — along with some added proteins. Because gonorrhoea and meningitis bacteria are close cousins, their OMVs carry overlapping sets of surface targets. Follow-up studies in several countries, as well as experiments in mice, confirmed that these meningitis vaccines can induce antibodies that recognise multiple gonorrhoea proteins and can shorten the duration of infection, although the protection is only partial and appears weaker in people with repeated exposures.
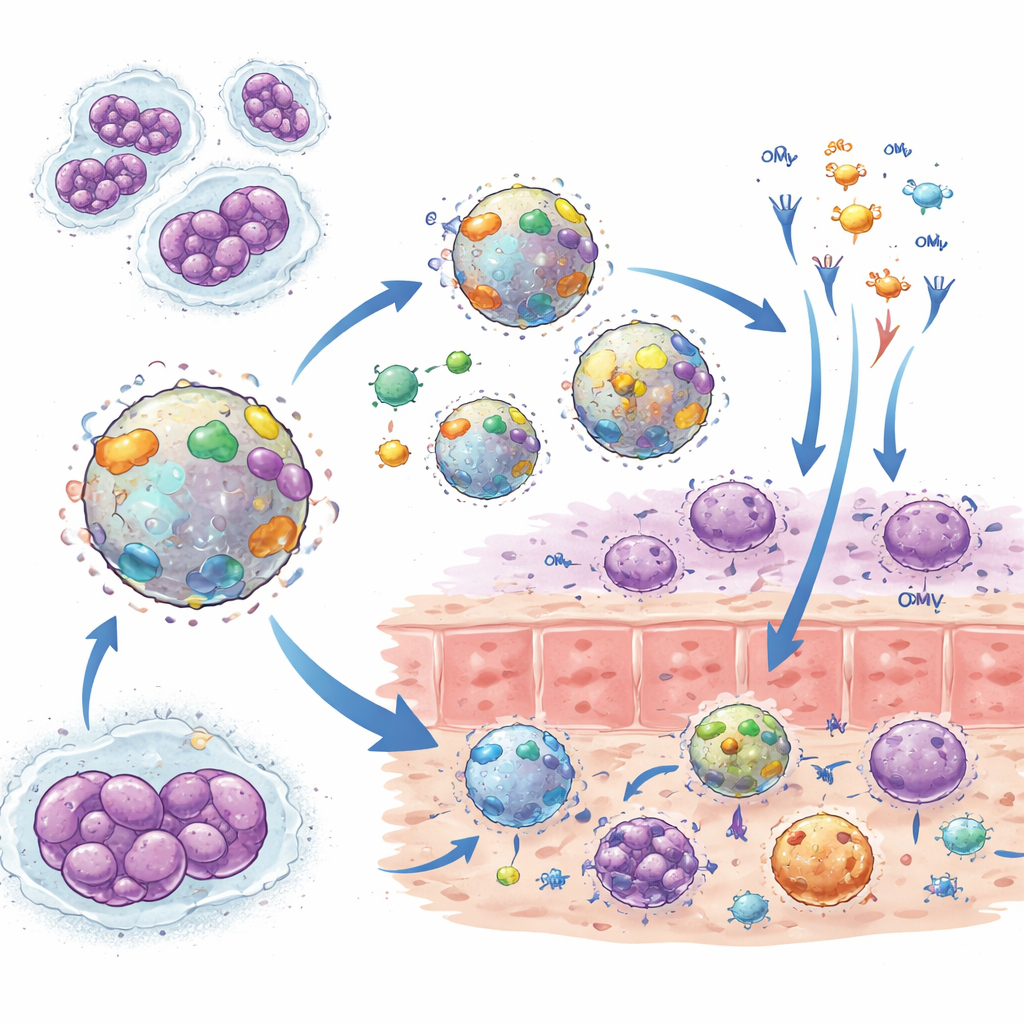
Designing smarter vesicle-based vaccines
Encouraged by these hints of cross-protection, researchers are now working on OMV vaccines purpose-built for gonorrhoea. One strategy keeps using meningitis bacteria but strips out some strongly variable or unhelpful components, or softens the toxic part of their surface, to focus the immune response and improve safety. Another uses OMVs made directly from gonorrhoea strains, combined with immune-boosting additives such as tiny particles that slowly release a Th1-promoting signal (the cytokine IL-12), or delivered by routes like the nose or vagina to stimulate strong local antibodies. Genetic tinkering can also swap in more helpful versions of certain proteins or remove those that suppress immunity, reshaping the vesicles so they display a richer, more protective set of targets.
Future directions for fighting gonorrhoea
The authors conclude that OMV-based vaccines remain one of the most promising paths to controlling gonorrhoea as antibiotics lose their edge. However, turning partial protection into reliable prevention will require carefully choosing which bacterial strains to base vaccines on, deeply cataloguing which vesicle components are most visible to the immune system, and re-engineering the bacterium so its vesicles no longer carry immune-dampening tricks. Future clinical studies, especially in high-risk groups and in both sexes, will be crucial for pinning down which antibody and cellular responses actually prevent infection. If successful, these rationally designed OMV vaccines could greatly reduce the global burden of gonorrhoea and its complications, protecting fertility and sexual health for millions.
Citation: Gu, Z., Unitt, A., Harrison, O.B. et al. Gonococcal outer membrane vesicle vaccines: bacterial population biology, clinical trials, immune profiling, and vaccine design. npj Vaccines 11, 85 (2026). https://doi.org/10.1038/s41541-026-01410-2
Keywords: gonorrhoea vaccines, outer membrane vesicles, antimicrobial resistance, Neisseria gonorrhoeae, sexually transmitted infections