Clear Sky Science · en
Genetic risk in Alzheimer’s disease
Why Our Genes Matter for Memory Loss
Alzheimer’s disease is often thought of as bad luck or a natural part of getting older, but decades of research show that our genes play a major role in who develops the condition and when. This review article pulls together the latest discoveries about Alzheimer’s genetic risk, why women are affected differently than men, and how modern computer tools can turn thousands of tiny DNA differences into early warning scores. For families worried about dementia, this work points toward a future where risk can be estimated more accurately and treatments can be tailored to the individual.
Many Small Genetic Nudges Toward Disease
The authors first explain that Alzheimer’s disease has a strong inherited component, with genes accounting for as much as four-fifths of the risk. A few rare gene changes can almost guarantee early, aggressive disease, but most people’s danger comes from the combined effect of many common DNA variants, each with only a small influence. Giant international studies that scan the whole genome have now uncovered more than 100 of these risk spots. Some sit in well-known genes such as APOE, TREM2, and ABCA7, which are involved in clearing sticky protein fragments, controlling inflammation in the brain, or handling fats and cholesterol. Others lie in stretches of DNA that do not code for proteins, making it harder to see how they act. New work in brain immune cells called microglia is beginning to show that some of these “silent” changes tweak the activity of nearby genes and the way cells respond to early damage.
Not One Alzheimer’s, but Many Flavors
Rather than being a single disease, Alzheimer’s increasingly appears to be a family of related brain disorders. The review describes studies that group patients into subtypes based on proteins in their spinal fluid. One subtype shows signs of overactive nerve-cell growth, another of an overzealous immune system, while others point to problems with the blood–brain barrier or the brain’s waste-clearing systems. Each pattern lines up with a different set of risk genes, suggesting that people arrive at memory loss through distinct biological routes. The authors also highlight important differences between racial and ethnic groups in how key gene variants, especially APOE, shape risk, and how other inherited traits like blood pressure and cholesterol can push people closer to or farther from disease.
How Sex Shapes Genetic Risk
A major theme of the article is that women and men do not share the same genetic landscape for Alzheimer’s. Women not only develop the disease more often, they also appear to move more quickly along harmful cellular pathways once brain changes begin. Studies of individual brain cells show that, in women, immune cells and supporting cells are more strongly switched on, while some nerve cells in men and women can even respond in opposite directions to the same pathology. When researchers build genetic risk scores separately for each sex, they find that some DNA variants matter more in women and others in men, and that sex-matched scores do a better job of predicting age of onset and the buildup of disease-related proteins. Machine learning methods trained on large genetic datasets further reveal sex-specific genes tied to DNA repair, cell division, stress responses, and other processes that may help explain women’s higher burden of disease.
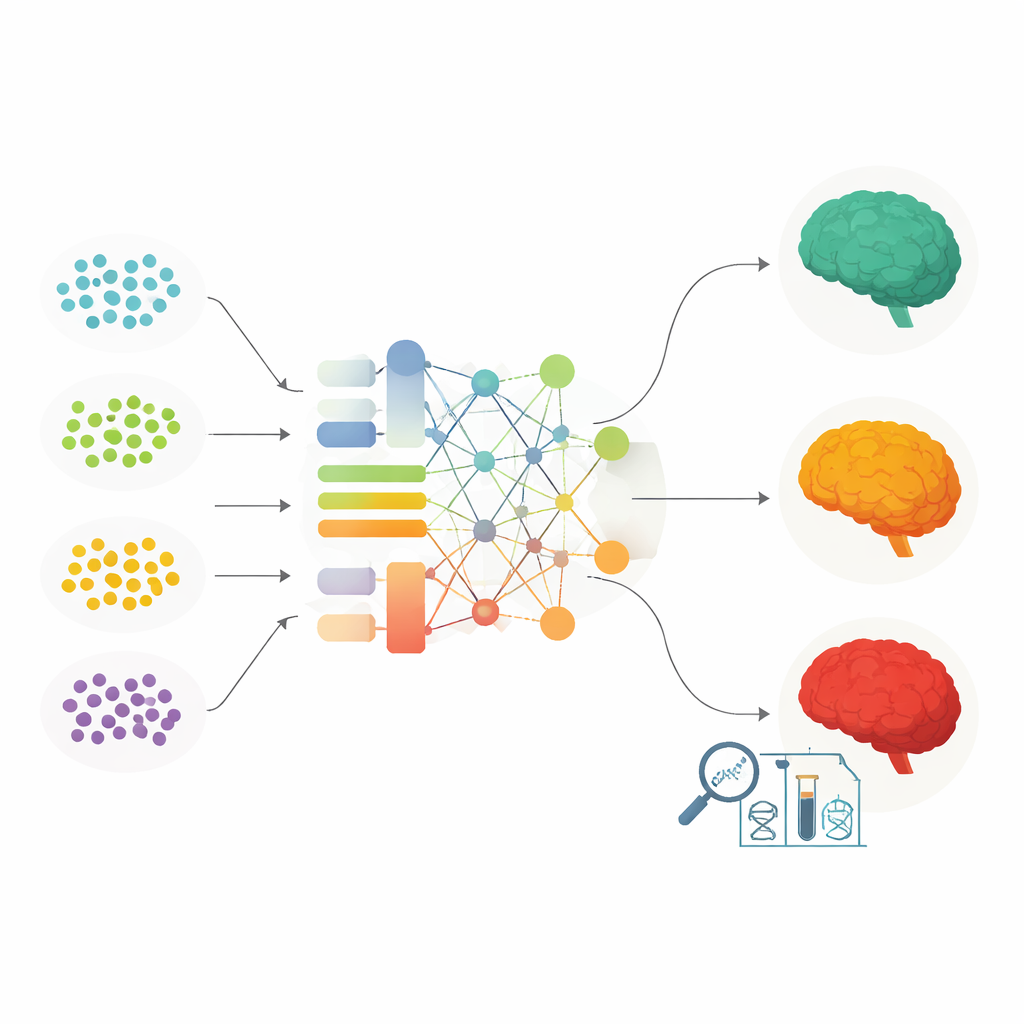
From Gene Lists to Personal Risk Scores
To turn all of this information into something potentially useful in the clinic, scientists use polygenic risk scores. These scores add up the combined push of many risk variants across the genome, sometimes giving extra weight to particularly important regions such as APOE. The review walks through different ways of building these scores and shows that carefully tuned versions can distinguish people who will go on to develop Alzheimer’s or progress from mild memory problems to full dementia. Newer approaches use machine learning algorithms that can capture complex interactions between variants, and even blend genetic data with brain scans and blood markers. These models generally outperform traditional scores, including in people with different APOE backgrounds, and can highlight which biological pathways are most important for prediction.
What This Means for Future Patients
Overall, the article concludes that genetics provides a powerful window into who is at risk for Alzheimer’s, why that risk differs by sex and ancestry, and which biological systems might be the best targets for treatment. At the same time, today’s risk scores work best in people of European descent and are not yet ready for routine screening. The authors argue that larger and more diverse genetic studies, deeper experiments to understand how non-coding DNA changes affect cells, and more transparent machine learning models are all needed before genetic prediction can safely guide prevention or therapy. If these challenges are met, future patients may receive personalized risk estimates that combine genes, sex, ancestry, and biomarkers, opening the door to earlier diagnosis and more precisely tailored interventions.
Citation: Pan, Y., Cho, H., Lou, Q. et al. Genetic risk in Alzheimer’s disease. npj Syst Biol Appl 12, 39 (2026). https://doi.org/10.1038/s41540-026-00665-8
Keywords: Alzheimer’s genetics, polygenic risk score, machine learning, sex differences, precision medicine