Clear Sky Science · en
Dietary advanced glycation products combined with chronic restraint stress induced anxiety-like and depression-like behaviors in male mice
Why Everyday Food and Stress Matter to Mood
Many people today juggle high stress and grab-and-go meals packed with processed, browned, or fried foods. This study asks a simple but important question: when an unhealthy diet and chronic stress occur together, can they team up to worsen anxiety and depression? Using male mice, the researchers traced how certain chemicals from cooked foods, gut bacteria, and brain cells interact, revealing a chain of events that links what we eat and how stressed we feel to changes in the brain that resemble mood disorders.
Burnt Bits in Food and a Stressed Body
The work centers on compounds called advanced glycation end products, or AGEs, which form when foods are cooked at high temperatures, such as grilling or frying. These compounds are abundant in many ultra-processed foods. The team exposed mice to an AGE-rich diet for 12 weeks and added daily restraint stress during the final month, mimicking a lifestyle of chronic tension and highly processed meals. Compared with control animals, stressed mice already showed signs of anxiety- and depression-like behavior, such as less pleasure from sweet solutions and more immobility in standard tests. Adding AGEs on top of stress made these behaviors noticeably worse, and brain tissue from key mood areas showed more damage and reduced levels of proteins that support healthy nerve connections.
The Gut’s Hidden Role in Mood
To understand how food and stress communicate with the brain, the researchers turned to the gut. They found that stress and AGEs together disturbed the community of gut microbes, reducing overall diversity and boosting strains previously linked to inflammation and chemical imbalance. At the same time, the intestinal barrier—normally a tight wall that controls what passes into the bloodstream—became leaky. Microscopy revealed inflamed tissue and loss of protective mucus-secreting cells, while key sealing proteins in the gut and in the blood–brain barrier were reduced. These changes allowed inflammatory signals and certain small molecules to circulate more freely and influence the brain, setting the stage for deeper chemical shifts. 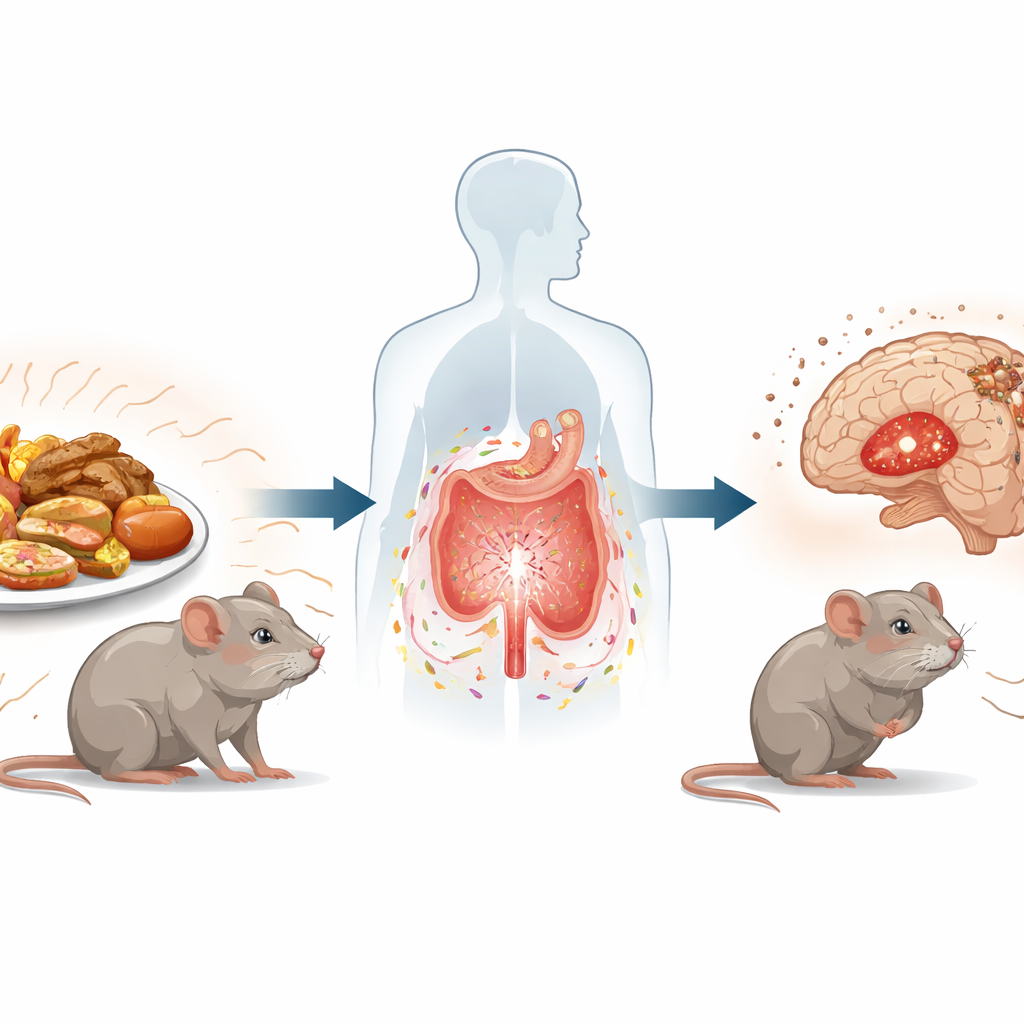
A Traffic Jam in Tryptophan Chemistry
The study focused on tryptophan, an amino acid best known as a building block for serotonin, a mood-related messenger. In healthy conditions, tryptophan feeds both serotonin production and another route called the kynurenine pathway. Under combined AGEs and stress, that balance tipped sharply. Less tryptophan and serotonin were found in the brain and body, while more of it was shunted into kynurenine and downstream products such as 3-hydroxykynurenine. These metabolites can cross into the brain and are known to promote oxidative stress, essentially chemical rusting inside cells. When the researchers blocked the key enzyme that drives this shift using a drug called 1-methyltryptophan, levels of these toxic by-products dropped, brain chemistry improved, and the mice showed fewer anxiety- and depression-like behaviors.
When Brain Cells Rust from Within
Digging even deeper, the team uncovered a specific kind of cell death in the hippocampus, a brain region tied to memory and emotion. This process, called ferroptosis, is driven by iron build-up and runaway lipid oxidation. Mice exposed to both AGEs and stress showed higher iron levels, weaker antioxidant defenses, and mitochondria—the cell’s power plants—with shrunken, damaged structures characteristic of ferroptosis. Inhibiting this cell-death pathway with a compound called ferrostatin-1 protected neurons and eased behavioral symptoms, even though it did not fix the upstream tryptophan imbalance. In cultured nerve-like cells, adding kynurenine or 3-hydroxykynurenine alone was enough to trigger ferroptosis, firmly linking these diet- and stress-related metabolites to iron-driven brain cell loss. 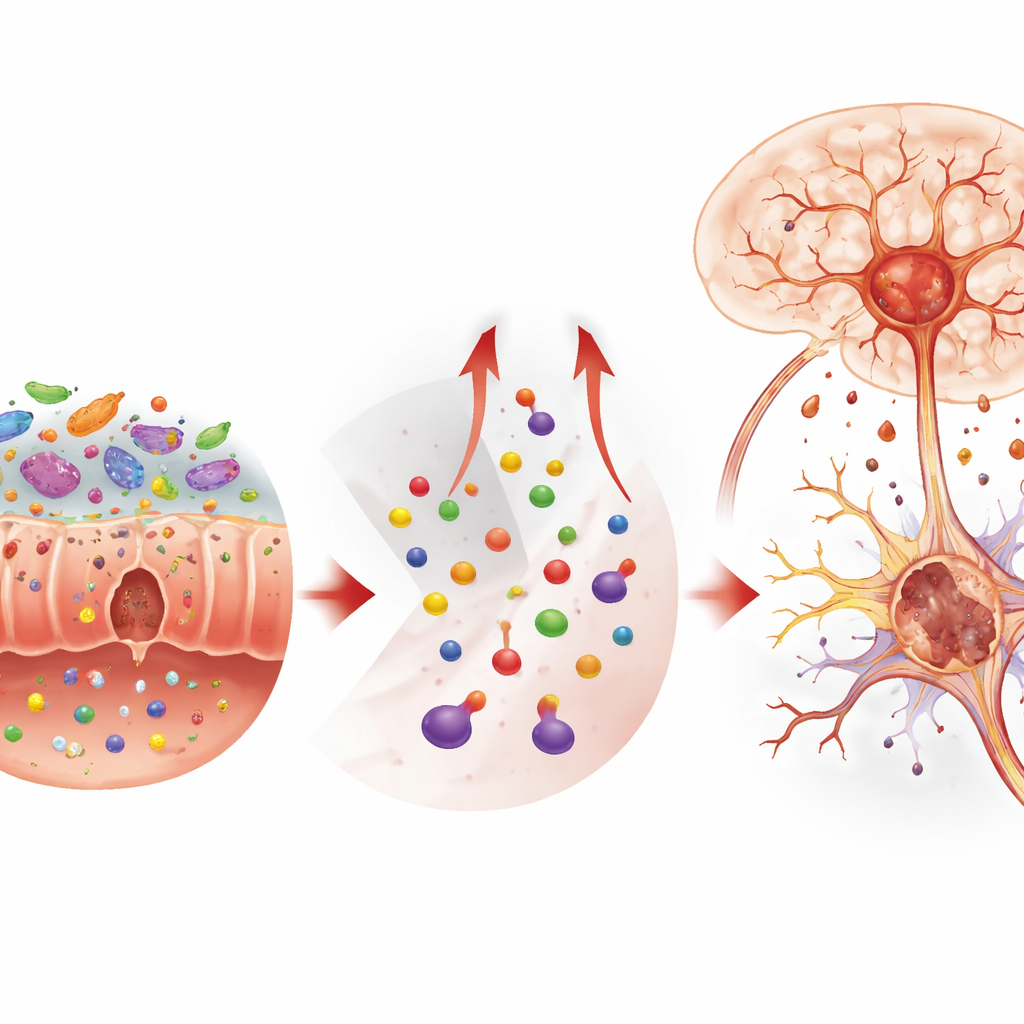
What This Means for Everyday Life
Taken together, the findings sketch a stepwise story: frequent intake of heavily cooked, AGE-rich foods combined with ongoing stress first unsettles the gut microbiome and weakens intestinal and brain barriers. This environment pushes tryptophan metabolism away from serotonin and toward toxic kynurenine products, which then accumulate in neurons and drive iron-dependent cell death. In mice, this chain ends in anxiety- and depression-like behaviors. While more work is needed in humans, the study suggests that managing stress, limiting ultra-processed and heavily browned foods, and targeting this gut–tryptophan–brain axis could become powerful strategies for preventing or treating mood disorders.
Citation: Zhang, Y., Gu, K., Xin, L. et al. Dietary advanced glycation products combined with chronic restraint stress induced anxiety-like and depression-like behaviors in male mice. npj Sci Food 10, 135 (2026). https://doi.org/10.1038/s41538-026-00789-5
Keywords: gut–brain axis, processed food, tryptophan metabolism, ferroptosis, anxiety and depression