Clear Sky Science · en
Dietary copper-driven colonic dysbiosis mediates oxidative stress and butyrate deficiency to facilitate the spread of resistome in pigs
Why Pig Feed Matters to Human Health
Farmers often add extra copper to pig feed to help young animals grow and fight off infections. But this helpful metal may have a hidden downside: it can encourage the rise and spread of bacteria that no longer respond to antibiotics. This study follows piglets raised on different forms of dietary copper and shows how an everyday farming practice can subtly reshape the gut, the microbes living there, and the pool of resistance genes that can eventually reach the wider environment and, potentially, people.
Two Kinds of Copper, Two Very Different Guts
The researchers compared three groups of weaned piglets: one with no added copper, one receiving a standard inorganic copper salt (copper sulfate), and one given an organic copper-peptide supplement. For four weeks, the pigs ate these “copper-rich” diets; afterward, all groups were switched to copper-free feed for another two weeks. By sequencing all the DNA in the animals’ feces, the team could track not only which bacteria were present, but also which carried genes that make them resistant to antibiotics or to metals such as copper and zinc.
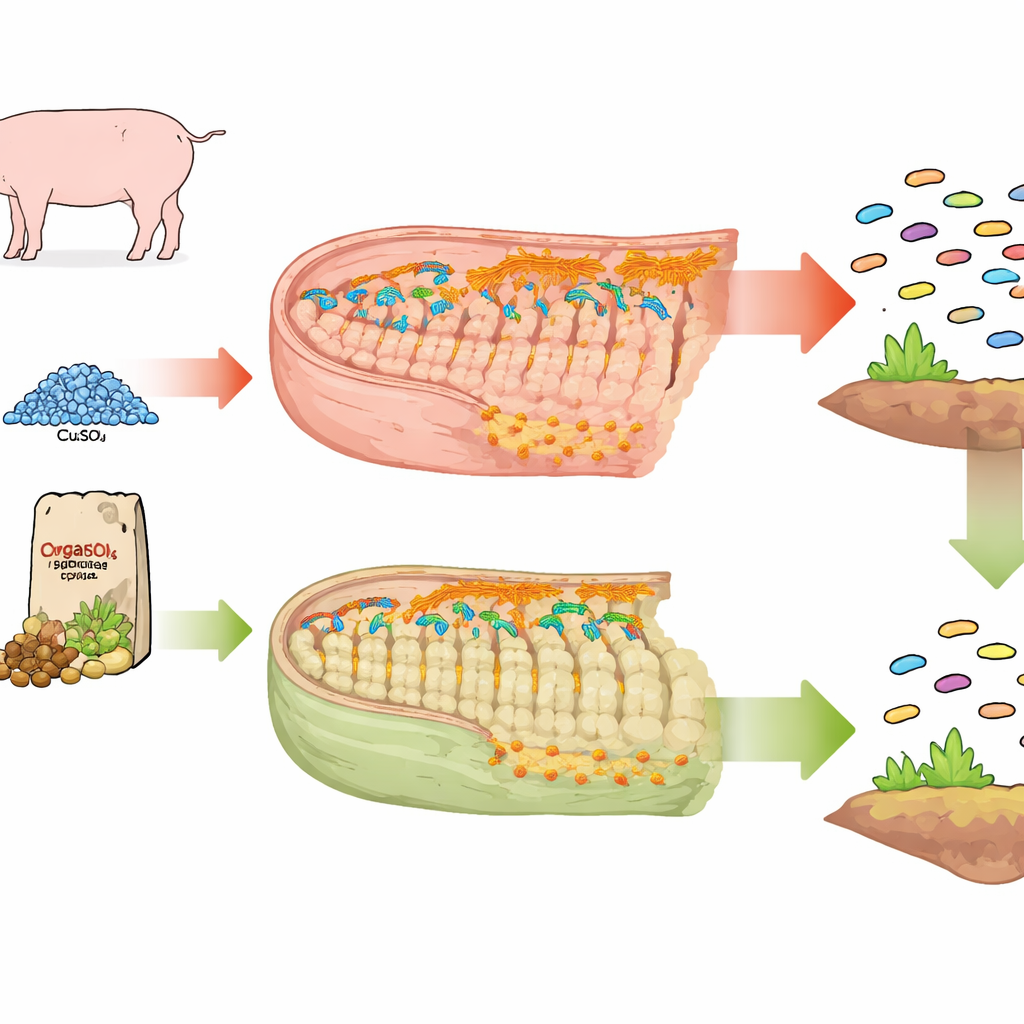
How Inorganic Copper Disturbs the Microbial Neighborhood
Piglets fed copper sulfate developed a much richer and denser collection of antibiotic resistance genes and metal resistance genes in their gut compared with pigs on a copper-free diet. Many of these genes were the kind that protect bacteria from multiple antibiotic families at once. They were also often linked to mobile genetic elements—tiny pieces of DNA, such as plasmids and transposons, that hop between microbes and shuttle resistance traits around. Disease-causing bacteria like Escherichia coli, Streptococcus suis, and Enterococcus species became more common hosts for these genes under copper sulfate feeding, turning the intestinal community into a larger and more dangerous reservoir of resistance.
From Healthy Barrier to Leaky, Stressed Colon
The copper sulfate diet did not just alter which microbes were present; it also damaged the gut’s physical defenses. Under the microscope, the colon lining of these pigs showed swelling, disorganized tissue, and signs of inflammation. Levels of protective proteins that help seal neighboring cells together were lower, indicating a leakier barrier. At the same time, the colon and blood contained more markers of oxidative stress—chemical by-products known as reactive oxygen species that can damage DNA. Beneficial bacteria that produce butyrate, a short-chain fatty acid that fuels colon cells and helps maintain a low-oxygen, stable environment, were reduced, and butyrate levels in the gut dropped.
Why Organic Copper Looks Safer
The organic copper-peptide supplement told a different story. Although it raised copper resistance genes—as any copper source might—it did not spur the same broad bloom of antibiotic resistance or mobile genetic elements. Pathogenic bacteria were not strongly favored, and damage to the intestinal barrier and butyrate levels was far milder. Because this form of copper is more easily absorbed earlier in the digestive tract, less appears to reach the colon to disturb the resident microbes. When all pigs were switched to copper-free feed, most metal resistance genes from the copper sulfate group declined, but antibiotic resistance genes remained elevated, pointing to lasting shifts in the gut ecosystem. In contrast, the organic copper group changed much less.
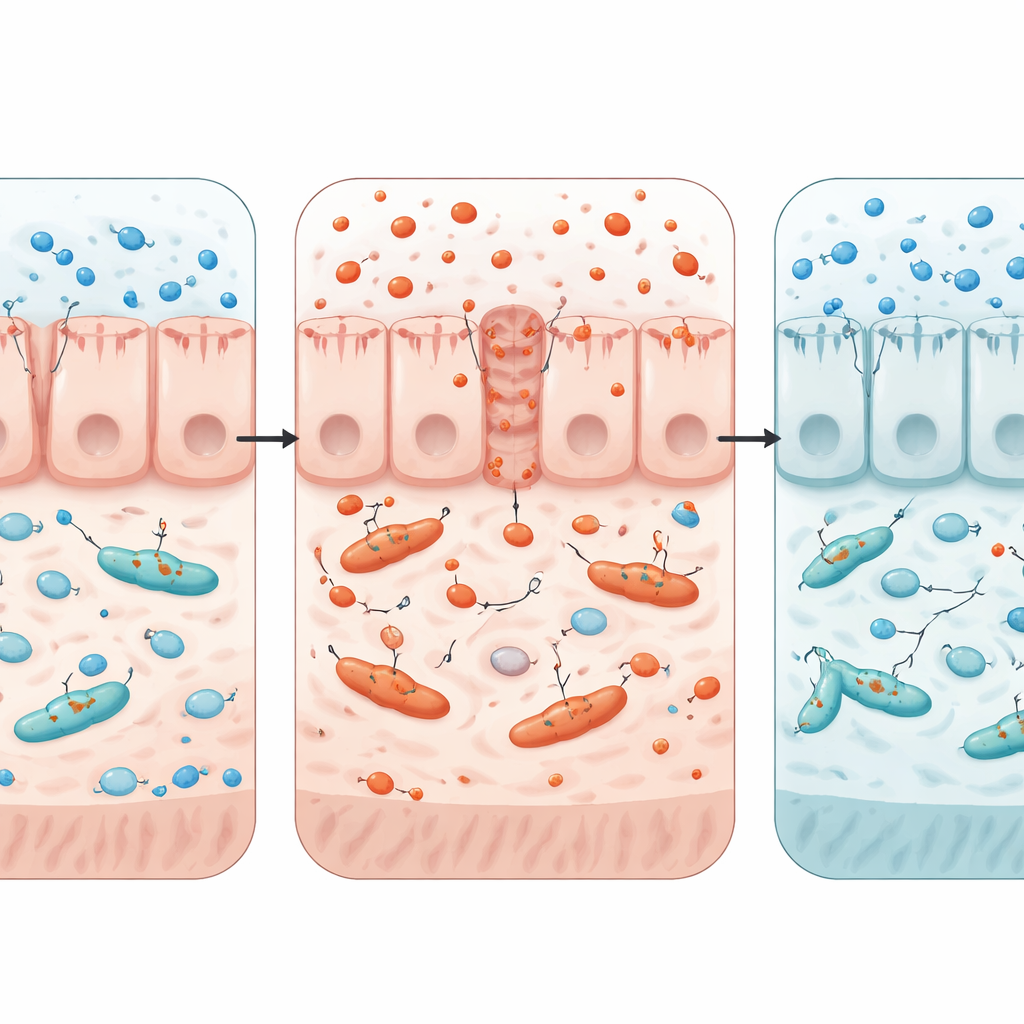
Zooming In on the Mechanism
To test how gut conditions influence resistance spread, the team built a laboratory model that combined a human-like intestinal cell layer with two strains of E. coli that could exchange a resistance-carrying plasmid. Adding copper ions increased oxidative stress in the cells, weakened the barrier, and boosted the rate at which the resistance plasmid moved between bacteria. When the researchers added butyrate or mimicked the low-oxygen conditions of a healthy colon, oxidative stress fell, the barrier strengthened, and gene transfer slowed. These tests supported the idea that it is not copper alone, but copper-triggered oxidative stress and loss of a stable, low-oxygen gut environment, that opens the door for resistance genes to spread.
What This Means Beyond the Barn
This work suggests that routine use of inorganic copper in pig feed can quietly enlarge and mobilize the collection of resistance genes in the animal gut by injuring the colon, depleting beneficial butyrate-producing microbes, and encouraging stressed, mobile DNA-rich pathogens. Organic copper sources, while not entirely risk-free, appear to deliver the growth benefits with far less disruption to intestinal balance and resistance spread. For farmers, veterinarians, and public health planners, the message is clear: choosing the form of a common mineral supplement can help curb the rise of hard-to-treat infections, even before any antibiotic is used.
Citation: Wen, Y., Gao, M., Wang, Z. et al. Dietary copper-driven colonic dysbiosis mediates oxidative stress and butyrate deficiency to facilitate the spread of resistome in pigs. npj Biofilms Microbiomes 12, 80 (2026). https://doi.org/10.1038/s41522-026-00949-1
Keywords: antimicrobial resistance, pig gut microbiome, dietary copper, butyrate and gut health, mobile resistance genes